What is in this leaflet
This leaflet answers some common questions about VAQTA. It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines and vaccines have risks and benefits. Your doctor has weighed the risks of you being given VAQTA against the benefits they expect it will have for you.
If you have any concerns about being given this vaccine, ask your doctor or pharmacist.
Keep this leaflet. You may need to read it again.
What VAQTA is used for
VAQTA is a vaccine used to help prevent hepatitis A. It can be given to children 12 months of age and older, teenagers and adults.
Hepatitis A is an infection of the liver caused by the hepatitis A virus. It can be caught by coming into contact with an infected person who has poor sanitary habits, eating or drinking foods prepared by an infected food handler, or, while uncommon, by blood transfusion from an infected donor. Other circumstances that can increase the risk of infection include:
- travelling to areas where hepatitis A is common
- living in a community with one or more recorded outbreaks within the last five years
- being around groups of other children, for example, daycare centres
- living in the same house as someone who is infected
- having chronic liver disease or having had a liver transplant
- sexual contact with someone who is infected
- being infected with Human Immunodeficiency Virus (HIV)
- working in the healthcare field
- eating raw shellfish
- sharing needles for injecting drugs
- having a blood disorder requiring transfusion of blood products.
Symptoms of hepatitis A usually begin 2 to 8 weeks after coming into contact with the virus. These include loss of appetite, feeling sick (nausea), vomiting, fever, chills, tiredness, yellowing of the skin and/or eyes and dark urine. Most people recover completely from hepatitis A disease. However, occasionally cases develop into severe disease and death.
Hepatitis A occurs in Australia and New Zealand, but is not common. However, it is very common in many parts of the world and the risk of infection is greatest in those areas where hygiene and sanitation are poor. Immunisation against hepatitis A is suggested for travellers to such areas, including Asia, India, Africa, Central and South America.
The vaccine contains inactivated virus and is injected into the body. The body then produces its own protection by making disease-fighting substances (antibodies) to fight the virus. The vaccine itself cannot cause the infection. If a vaccinated person comes into contact with live virus the body is usually ready, and produces antibodies to destroy it. However, as with all vaccines, 100% protection against hepatitis A cannot be guaranteed.
Because hepatitis A infection can go undetected for a long period of time, it is possible that an individual may already be infected at the time the vaccine is given. The vaccine may not prevent hepatitis A in these individuals.
VAQTA will not protect against hepatitis caused by other agents or viruses (such as hepatitis B, hepatitis C, hepatitis D, hepatitis E).
Before you are given VAQTA
When you or your child must not be given it
Do not have VAQTA if:
- you or your child have an allergy to VAQTA or any of the ingredients listed at the end of this leaflet
- the expiry date on the pack has passed.
If the vaccine is used after the expiry date has passed, it may not work.
If you are not sure whether you or your child should be given VAQTA, talk to your doctor.
Do not give VAQTA to children under 12 months of age. The safety and effectiveness of VAQTA in these children have not been established.
Before you or your child are given it
Tell your doctor if:
- you are pregnant or intend to become pregnant
It is not known whether the vaccine is harmful to an unborn baby when administered to a pregnant woman. Your doctor will give you VAQTA only if it is clearly needed.
- you are breast-feeding
It is not known whether VAQTA passes into breast milk. Your doctor will discuss the possible risks and benefits of you being given VAQTA while breast-feeding.
- you or your child have any medical conditions, especially the following:
- bleeding problems
- diseases which decrease the immune system, for example, AIDS
- cancer
VAQTA may not work as well as it should if you or your child have diseases or conditions which decrease the body's immune defence system. Your doctor will decide whether or not to give the vaccine.
- you or your child have an acute infection or a high temperature
Your doctor may decide to delay your injection of VAQTA.
- you or your child have any allergies to any other medicines or any other substances, such as foods, preservatives, latex rubber or dyes.
If you have not told your doctor about any of the above, tell them before you or your child are given an injection of VAQTA.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop. VAQTA may not work as well as it should if you or your child are taking medicines that decrease the immune system, such as corticosteroids (eg. prednisone) or cyclosporin.
Your doctor will advise you if you are taking any of these or other medicines that decrease the immune system. Your doctor will decide whether or not to give the vaccine.
Use with other vaccines
VAQTA may be given at the same time as yellow fever, typhoid, measles, mumps, rubella, varicella and pneumococcal 7-valent conjugate vaccines. VAQTA may also be given at the same time as an immune globulin injection. The injections should be given at different places on the body and using separate syringes.
There is limited information available regarding the administration of other vaccines at the same time as VAQTA. Your doctor will decide if VAQTA should be given with other vaccines.
How VAQTA is given
How much is given
Your doctor will decide on the dose of VAQTA that you will be given This depends on your age.
The usual dose for children and teenagers (12 months through 17 years of age) is 0.5 mL (approx. 25U).
The usual dose for adults (18 years and older) is 1.0 mL (approx. 50U).
How it is given
VAQTA is given as an injection, usually into your upper arm muscle by a doctor or trained nurse.
The vaccine should not be injected directly into veins (intravenously).
Vaccination schedule
VAQTA is generally given as a total of two doses. Each dose is given on a separate visit.
The schedule for children and teenagers (12 months to 17 years) is:
- 1st dose: at elected date
- 2nd dose: 6 to 18 months later
The schedule for adults is:
- 1st dose: at elected date
- 2nd dose: 6 to 18 months later
The schedule for adults infected with HIV is:
- 1st dose: at elected date
- 2nd dose: 6 months later
It is important to return at the scheduled date for the follow-up dose.
If you have been given another hepatitis A vaccine as your 1st dose, VAQTA may be given as the 2nd dose, 6 to 12 months later.
If you miss a dose
If you miss a scheduled dose, talk to your doctor and arrange another visit as soon as possible.
After you have been given VAQTA
Things you must do
Keep your follow-up appointment with your doctor or clinic. It is important to have your follow-up dose of VAQTA at the appropriate time to make sure the vaccine has the best chance of providing protection against the hepatitis A virus.
Things to be careful of
Be careful driving or operating machinery until you know whether VAQTA has affected you. VAQTA should not normally interfere with your ability to drive a car or operate machinery. However, VAQTA may cause tiredness or weakness in some people. Make sure you know how you react to VAQTA before you drive a car, operate machinery, or do anything else that could be dangerous if you are tired or weak.
Side Effects
Tell your doctor or pharmacist as soon as possible if you do not feel well during or after having had an injection of VAQTA. VAQTA helps protect most people from hepatitis A, but it may have unwanted side ffects in a few people. All medicines and vaccines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
In all children, teenagers and adults:
Tell your doctor if you or your child has any of the following and they are troublesome or ongoing:
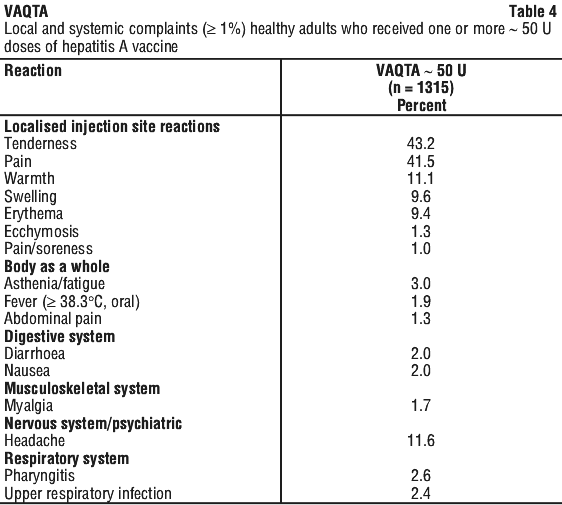
- local reaction around the injection site such as pain, soreness, tenderness, warmth, redness or swelling
- fever, sore throat, runny nose, cough
- stomach pain, feeling sick (nausea), vomiting, diarrhoea
- headache
These are the more common side effects of VAQTA that may occur in children, teenagers and adults. For the most part these have been mild.
In children - 12 months to 23 months of age:
Tell your doctor if your child experiences these and they are troublesome or ongoing:
- skin rash
- irritability, crying
- loss of appetite
- discharge with itching of the eyes and crusty eyelids
- infection of the ear, which may cause temporary hearing loss and pain
These are the more common side effects of VAQTA that have been reported in children aged 12 months to 23 months.
In adults:
Tell your doctor if you notice any of the following and they worry you:
- tiredness, generalised weakness
- aching muscles, muscle tenderness, arm pain
These are other mild side effects of VAQTA that have been reported in adults.
In children/teenagers 2 years to 17 years of age and adults 18 years of age and older:
Tell your doctor immediately if you or your child experience any of the following:
- muscle weakness
- loss of coordination, unsteadiness when walking
- bleeding or bruising more easily than normal
- headache and fever, progressing to hallucinations, confusion, paralysis of part or all of the body, disturbances of behaviour, speech and eye movements, stiff neck and sensitivity to light
These may be serious side effects. You may need urgent medical attention.
Tell your doctor immediately if you or your child notice any of the following after being given the injection:
- skin rash, itching
- hives or nettlerash (pinkish, itchy swellings on the skin)
These may be signs of an allergic reaction to VAQTA. These side effects are rare.
Other side effects not listed above may also occur in some patients. Tell your doctor if you notice any other effects. Do not be alarmed by this list of possible side effects. You may not experience any of them.
Storage
VAQTA is usually stored in the doctor's surgery or clinic, or at the pharmacy. However if you need to store VAQTA:
- Keep it where children cannot reach it.
- Keep it in the refrigerator, but not in the door compartment.
- Do not put VAQTA in the freezer, as freezing destroys the vaccine.
- Keep the injection in the original pack until it is time for it to be given.
Product description
What it looks like
VAQTA comes in glass vials and prefilled syringes. It is a white milky liquid. Two different vaccine doses are available:
- approximately 25 Units in 0.5 mL of liquid
- approximately 50 Units in 1 mL of liquid
In the injection vial products, the vial stopper contains dry natural latex rubber.
In the injection syringe products, the syringe plunger stopper and tip cap contain dry natural latex rubber.
Ingredients
Active ingredient:
The active ingredient of VAQTA is the hepatitis A virus protein. The vaccine is not infectious, and will not give you the hepatitis A virus.
Inactive ingredients:
- aluminium (as amorphous aluminium hydroxyphosphate sulfate)
- borax
- sodium chloride
VAQTA is made without any human blood or blood products. It does not contain any infectious material.
The manufacture of this product includes exposure to bovine derived materials. No evidence exists that any case of vCJD (considered to be the human form of bovine spongiform encephalopathy) has resulted from the administration of any vaccine product.
Supplier
Who sponsors VAQTA
Merck Sharp & Dohme (Australia) Pty Ltd
Level 1, Building A, 26 Talavera Road
Macquarie Park NSW 2113
http://www.msd-australia.com.au
Tel: (+61) 02 8988 8000
Who distributes VAQTA
Seqirus (Australia) Pty Ltd
Melbourne, Victoria
Australia
http://www.seqirus.com.au
Tel: 1800 642 865
This leaflet was prepared in May 2024
Australian Register Numbers:
25U Vial - AUST R 58534
50U Vial - AUST R 58536
25U Injection syringe - AUST R 58539
50U Injection syringe - AUST R 58540
WPC-VAQ-I-122010S
Copyright © 2024 Merck & Co., Inc., Rahway, NJ, USA, and its affiliates. All rights reserved.
Published by MIMS July 2024




 Local and/or systemic hypersensitivity reactions occurred in < 1% of children, adolescents or adults in clinical trials and included the following regardless of causality: pruritus, urticaria and rash.
Local and/or systemic hypersensitivity reactions occurred in < 1% of children, adolescents or adults in clinical trials and included the following regardless of causality: pruritus, urticaria and rash.

