1 Name of Medicine
Vinorelbine tartrate.
2 Qualitative and Quantitative Composition
Capsules contain 20 mg, 30 mg and 80 mg vinorelbine (as tartrate).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Soft capsule.
20 mg soft capsule.
Oval-shaped light brown soft capsule.
30 mg soft capsule.
Oblong-shaped pink soft capsule.
80 mg soft capsule.
Oblong-shaped pale yellow soft capsule*.
* Not marketed.4.1 Therapeutic Indications
Non-small cell lung cancer.
Velabine is indicated for the first line treatment of advanced non-small cell lung cancer, as a single agent or in combination.
Breast cancer.
Velabine is indicated for the treatment of advanced breast cancer after failure of standard therapy as a single agent or in combination.4.2 Dose and Method of Administration
Adults.
Single agent. The recommended regimen is:
First three administrations.
60 mg/m2, administered once weekly.
Subsequent administrations.
Beyond the third administration, increasing the dose of vinorelbine soft capsules to 80 mg/m2 once weekly is recommended, except in those patients for whom the neutrophil count has dropped once below 500/mm3 or more than once between 500 and 1000/mm3 during the first 3 administrations at 60 mg/m2.
Dose modifications according to haematological status. If the neutrophil count is below 1500/mm3 and/or the platelet count is below 100,000/mm3, then treatment should be delayed until recovery. See Table 1.
 For any administration planned at the 80 mg/m2/week dose, if the neutrophil count falls below 500/mm3 or more than once between 500 and 1000/mm3, the dose must be delayed until recovery and reduced from 80 to 60 mg/m2 per week during the 3 subsequent administrations. See Table 2.
For any administration planned at the 80 mg/m2/week dose, if the neutrophil count falls below 500/mm3 or more than once between 500 and 1000/mm3, the dose must be delayed until recovery and reduced from 80 to 60 mg/m2 per week during the 3 subsequent administrations. See Table 2.
 It is possible to re-escalate the dose from 60 to 80 mg/m2/week if the neutrophil count does not drop below 500/mm3, or more than once between 500 and 1000/mm3 during the three administrations given at the 60 mg/m2 dose.
It is possible to re-escalate the dose from 60 to 80 mg/m2/week if the neutrophil count does not drop below 500/mm3, or more than once between 500 and 1000/mm3 during the three administrations given at the 60 mg/m2 dose.
Dose modification for hepatic impairment. Vinorelbine is contraindicated in patients with severe hepatic impairment. In patients with moderate hepatic impairment (serum bilirubin between 1.5 and 3 x ULN, independent of ALT and AST concentrations), vinorelbine capsules monotherapy should be administered at a dose of 50 mg/m2/week. In patients with mild hepatic impairment (serum bilirubin < 1.5 x ULN and ALT and/or AST between 1.5 and 2.5 x ULN), vinorelbine capsules may be administered at the standard dose of 60 mg/m2/week. Haematological toxicity should be closely monitored. Hepatic impairment has not been studied in combination chemotherapy.
Combination chemotherapy. General.
The use of oral vinorelbine in combination regimens has not been extensively studied. However, based on pharmacokinetic studies, the oral dose of 80 mg/m2 was demonstrated to correspond to 30 mg/m2 of the IV* form and 60 mg/m2 orally to 25 mg/m2 IV.
In combination regimens, intravenous vinorelbine dosing may be replaced with oral vinorelbine therapy. The recommended dose is 60 mg/m2. The safety of higher doses of oral vinorelbine (e.g. 80 mg/m2) in combination regimens has not been established, except for use with capecitabine in advanced breast cancer (see below).
* Vinorelbine injection for intravenous administration is available in other brands.
Combination with capecitabine in advanced breast cancer.
In combination with capecitabine for the treatment of advanced breast cancer, the administration of vinorelbine capsules is recommended on days 1 and 8 for three week cycles at 60 mg/m2 for the first cycle, then increased to 80 mg/m2 for subsequent cycles, except in patients for whom the neutrophil count has dropped below 500/mm3 or more than once between 500 and 1000/mm3 during the first administration at 60 mg/m2. Capecitabine should be administered at a dose of 1000 mg/m2, twice daily on Days 1-14 of the three week cycle.
Table 3 gives the dose required for appropriate ranges of body surface area (BSA).
 Even for patients with a body surface area (BSA) ≥ 2 m2, the dose should never exceed 120 mg per week at 60 mg/m2 and 160 mg per week at 80 mg/m2.
Even for patients with a body surface area (BSA) ≥ 2 m2, the dose should never exceed 120 mg per week at 60 mg/m2 and 160 mg per week at 80 mg/m2.
Procedures for proper handling and disposal of anticancer drugs should be used. Several guidelines on this subject have been published.
Method of administration.
Velabine soft capsules must be given strictly by the oral route. They should be swallowed whole with water and should not be chewed or sucked. It is recommended that the capsule be taken with food.4.3 Contraindications
Known hypersensitivity to vinorelbine or to other vinca alkaloids or to any of the excipients listed in Section 6.1 List of Excipients.
Disease significantly affecting absorption.
Previous significant surgical resection of stomach or small bowel.
Neutrophil counts < 1500 cells/mm3, or current or recent severe infection due to neutropenia (within 2 weeks).
Platelet count < 100,000 cells/mm3.
Pregnancy.
Lactation (see Section 4.6 Fertility, Pregnancy and Lactation).
Patients requiring long-term oxygen therapy.
Severe hepatic insufficiency.
In combination with yellow fever vaccine (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
4.4 Special Warnings and Precautions for Use
Administration.
Vinorelbine soft capsules should be administered under the supervision of a physician experienced in the use of cancer chemotherapeutic agents with facilities for monitoring cytotoxic drugs.
If the patient chews or sucks the capsule by mistake, rinse mouth with water or preferably a normal saline solution. In the event of the capsule being cut or damaged, the liquid content is an irritant, and so may cause damage if in contact with skin, mucosa or eyes. Damaged capsules should not be swallowed and should be returned to the pharmacy or to the physician in order to be properly destroyed. If any contact occurs, immediate thorough washing with water or preferably with normal saline solution should be undertaken.
If vomiting occurs within a few hours of drug intake, administration of the dose should not be repeated. Prophylactic treatment such as 5HT3 antagonists (e.g. ondansetron, granisetron) may reduce the incidence. Vinorelbine capsules are associated with a higher incidence of nausea/vomiting than the IV formulation. A primary prophylaxis with antiemetics is recommended. It is recommended that the capsule be taken with food.
Constipation is a very common gastrointestinal disorder. The prescription of laxatives may be appropriate for patients with a prior history of constipation and/or are receiving concomitant treatment with opiates.
Myelosuppression.
Neutropenia is dose-limiting. Complete blood counts with differentials should be performed and results reviewed prior to administering each dose of vinorelbine capsules. Patients treated with vinorelbine capsules should be frequently monitored for myelosuppression both during and after therapy. Vinorelbine capsules should not be administered to patients with neutrophil counts < 1500 cells/mm3 and/or platelet counts below 100,000 cells/mm3. Patients developing severe neutropenia should be monitored carefully for evidence of infection and/or fever. If patients present signs or symptoms suggestive of infection, a prompt investigation should be carried out (see Section 4.2 Dose and Method of Administration, Dose modifications according to haematological status).
Vinorelbine capsules should be used with extreme caution in patients whose bone marrow reserve may have been compromised by prior irradiation or chemotherapy, or whose marrow function is recovering from the effects of previous chemotherapy (see Section 4.2 Dose and Method of Administration).
During clinical trials, where treatment was initiated at a weekly dose of 80 mg/m2 (corresponding to an IV dose of 30 mg/m2 in terms of systemic exposure), febrile neutropenia, in some cases fatal, was encountered in about 15% of patients. Therefore, it is recommended that the starting dose should be 60 mg/m2 and increased to 80 mg/m2 only if the dose is tolerated (see Section 4.2 Dose and Method of Administration).
General.
Most drug-related adverse events of vinorelbine capsules are reversible. If severe adverse events occur, vinorelbine capsules should be reduced in dosage or discontinued and appropriate corrective measures taken. Reinstitution of therapy with vinorelbine capsules should be carried out with caution and alertness as to possible recurrence of toxicity.
Special care should be taken when prescribing for patients: with a history of ischaemic heart disease; with poor performance status.
Patients presenting with ischaemic cardiac disease should be carefully monitored (see Section 4.8 Adverse Effects (Undesirable Effects)).
This product is specifically contraindicated with yellow fever vaccine. Its concomitant use with other live attenuated vaccines is not recommended.
Acute shortness of breath and severe bronchospasm have been reported infrequently along with rare cases of interstitial pneumopathy following the administration of vinorelbine and other vinca alkaloids, most commonly when the vinca alkaloid was used in combination with mitomycin. These adverse events may require treatment with supplemental oxygen, bronchodilators and/or corticosteroids, particularly when there is a pre-existing pulmonary dysfunction.
Due to the presence of sorbitol, patients with the rare hereditary problem of fructose intolerance should not take this medicine.
Vinorelbine capsules contain small amounts of ethanol (alcohol), less than 100 mg per dose. The small amount of alcohol in this medicine will not have any noticeable effects.
Vinorelbine capsules should not be given concomitantly with radiotherapy if the treatment field includes the liver.
Use in hepatic impairment.
Vinorelbine is contraindicated in patients with severe hepatic impairment. In patients with moderate hepatic impairment (serum bilirubin between 1.5 and 3 x ULN, independent of ALT and AST concentrations), dose reduction is recommended (see Section 4.2 Dose and Method of Administration). In patients with mild hepatic impairment (serum bilirubin < 1.5 x ULN and ALT and/or AST between 1.5 and 2.5 x ULN), no dose reduction is required. Haematological toxicity should be closely monitored.
Use in renal impairment.
Because of the low level of renal excretion, no dose modification is necessary in patients with renal impairment.
Use in the elderly.
Clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Paediatric use.
Safety and effectiveness have not been established. Use in children and adolescents aged < 18 years is therefore not recommended.
Effects on laboratory tests.
Since dose-limiting clinical toxicity is the result of depression of the white blood cell count, it is imperative that complete blood counts with differentials be obtained and reviewed on the day of treatment prior to each dose of vinorelbine capsules. If the neutrophil count is below 1500/mm3 and/or the platelet count is below 100,000/mm3, then treatment should be delayed until recovery.4.5 Interactions with Other Medicines and Other Forms of Interactions
Interactions common to all cytotoxics.
Due to the increase of thrombotic risk in the case of tumoral diseases, the use of anticoagulative treatment is frequent. As the intra-individual variability of the coagulability during diseases is high and there is the risk of interaction between oral anticoagulants and anticancer therapy, if the patient is treated with oral anticoagulants, increasing the frequency of INR (International Normalised Ratio) monitoring is recommended.
Concomitant use contraindicated. Yellow fever vaccine.
Risk of fatal generalised vaccine disease.
Concomitant use not recommended. Live attenuated vaccines.
Risk of generalised vaccine disease, possibly fatal. This risk is increased in patients already immunodepressed by their underlying disease. The use of an inactivated vaccine where one exists is recommended (e.g. poliomyelitis).
Phenytoin.
Risk of exacerbation of convulsions resulting from the decrease in phenytoin absorption by the cytotoxic drug or loss of efficacy of the cytotoxic drug due to increased hepatic metabolism by phenytoin.
Concomitant use with caution. Ciclosporin, tacrolimus.
Excessive immunodepression with risk of lymphoproliferation.
Interactions specific to vinca alkaloids.
Concomitant use not recommended. Itraconazole.
Increased neurotoxicity of vinca alkaloids due to the decrease in their hepatic metabolism.
Concomitant use with caution. Mitomycin.
Acute pulmonary reactions have been reported with vinorelbine and other vinca alkaloids used in conjunction with mitomycin: risk of bronchospasm and dyspnoea are increased; in a rare case, an interstitial pneumonitis was observed. Vinorelbine capsules should be administered with caution in combination with mitomycin.
As vinca alkaloids are known as substrates for P-glycoprotein, and in the absence of a specific study, caution should be exercised when combining vinorelbine with strong modulators of this membrane transporter.
Interactions specific to vinorelbine.
The combination of vinorelbine capsules and other drugs with known bone marrow toxicity is likely to exacerbate the myelosuppressive adverse effects.
In the absence of specific studies evaluating drug-drug interaction with warfarin, the patient should be cautiously monitored when vinorelbine is given in combination with warfarin.
Although the pharmacokinetics of vinorelbine are not influenced by the concurrent administration of cisplatin, the incidence of toxicities, specifically granulocytopenia, with the combination of vinorelbine capsules and cisplatin is significantly higher than with single-agent vinorelbine capsules.
In studies with rats, the anticoagulant effect of phenindione was potentiated when given in combination with a high dose of vinorelbine (30 mg/m2/day for 4 consecutive days or 15 mg/m2/day for 5 consecutive days) but combination treatment with sodium valproate did not cause any increase in anticonvulsant activity.
Vinorelbine is metabolised by cytochrome CYP3A4. Although interaction studies have not been performed, it is expected that inhibitors of CYP3A4 such as ketoconazole, itraconazole, ritonavir etc. would result in elevated blood concentrations of vinorelbine. Inducers of CYP3A4 such as rifampicin and phenytoin may reduce concentrations of vinorelbine. Since the magnitude of the inducing or inhibiting effects is unknown, such drug combinations should be avoided.
An increased incidence of grade 3/4 neutropenia has been suggested when intravenous vinorelbine and lapatinib were associated in one clinical phase I study. In this study, the recommended dose of the intravenous form of vinorelbine in a 3-weekly schedule on Day 1 and Day 8 was 22.5 mg/m2 when combined with daily lapatinib 1000 mg. This type of combination should be administered with caution.
Food.
Simultaneous intake of a low fat standard meal does not modify exposure to vinorelbine.4.6 Fertility, Pregnancy and Lactation
Contraception in males and females.
Women of childbearing potential should be advised to avoid becoming pregnant during therapy with vinorelbine capsules. Effective contraception must be used during treatment and for 7 months after the treatment.
Men being treated with vinorelbine capsules are advised not to father a child during and for a minimum of 4 months after treatment. Men must use effective contraception during treatment and for 4 months after treatment.
Effects on fertility.
Adverse effects on the male reproductive system were observed in repeat-dose toxicity studies in animals, including decreased spermatogenesis in rats dosed twice weekly at 2.1 - 7.2 mg/m2 for 13 weeks, reduced prostate/seminal vesicle secretion in rats dosed twice weekly at 3 mg/m2 for 26 weeks, reduced testicular weight in mice dosed at 19 mg/m2/day for three 5-day cycles and reduced epididymal weight in dogs dosed at 5 mg/m2 for 26 weeks. Vinorelbine tartrate did not affect fertility when administered to male and female rats prior to and during mating; however, the doses used in these studies (9 mg/m2 once weekly or up to 4.2 mg/m2 at 3-day intervals) were lower than the human dose.
Prior to treatment, advice should be sought for conserving sperm due to the chance of irreversible infertility as a consequence of treatment with oral vinorelbine.
(Category D)
Vinorelbine capsules may cause foetal harm if administered to a pregnant woman. When given every three days during organogenesis, vinorelbine tartrate has been shown to be teratogenic in rats and rabbits at doses of 3 and 7.7 mg/m2, respectively. A single 9 mg/m2 dose of vinorelbine tartrate caused embryonic deaths in mice. Doses causing adverse foetal effects in animals were lower than the human dose. There are no studies in pregnant women. If vinorelbine capsules are used during pregnancy, or if the patient becomes pregnant while receiving this drug, the patient should be apprised of the potential hazard to the foetus.
It is not known whether vinorelbine is excreted in milk of animals or humans. A study in rats showed that growth of the offspring was suppressed when vinorelbine tartrate was administered to lactating dams at 6 mg/m2 every three days. Because many drugs are excreted in human milk and because of the potential serious adverse reactions in nursing infants from vinorelbine capsules, breastfeeding must be discontinued in women before starting treatment with vinorelbine capsules.4.7 Effects on Ability to Drive and Use Machines
The effect of vinorelbine on the ability to drive and use machines has not been studied. However, patients should be advised not to drive or operate machinery if they experience any adverse reactions with a potential impact on their ability to perform these activities (e.g. dizziness and fatigue are common).
4.8 Adverse Effects (Undesirable Effects)
The overall reported incidence of adverse effects was determined from clinical studies in 316 patients (132 patients with non-small cell lung cancer and 184 patients with breast cancer) who received the recommended regimen of vinorelbine (first three administrations at 60 mg/m2/week followed by 80 mg/m2/week).
Adverse reactions reported are listed below by MedDRA body system organ class and the frequency convention.
Frequency of adverse reactions is defined as: very common (≥ 1/10); common (≥ 1/100 and < 1/10); uncommon (≥ 1/1000 and < 1/100); rare (≥ 1/10,000 and < 1/1000) and very rare (< 1/10,000). Additional adverse reactions pooled from post-marketing experience and clinical trials have been added according to the MedDRA classification with the frequency 'Not known'.
The reactions were described using the Common Terminology Criteria for Adverse Events (CTCAE) classification which provides a terminology for adverse events (Aes) and a grading scale the severity of AEs (grade 1=G1; grade 2=G2; grade 3=G3; grade 4=G4; grade 1-4=G1-4; grade 1-2=G1-2; grade 3-4=G3-4).
Adverse effects reported with oral vinorelbine.
Pre-marketing experience. The most commonly reported adverse drug reactions are bone marrow depression with neutropenia, anaemia and thrombocytopenia, gastrointestinal toxicity with nausea, vomiting, diarrhoea, stomatitis and constipation. Gastrointestinal adverse events occur more commonly with oral vinorelbine than with intravenous administration. Fatigue and fever were also very commonly reported.
Post-marketing experience. Vinorelbine capsules are used as a single agent or in combination with other chemotherapeutic agents or targeted therapy agents such as cisplatin or capecitabine. The most common system organ classes involved from post-marketing experience are: 'blood and lymphatic system disorders', 'gastrointestinal disorders', 'infections and infestations' and 'general disorders and administration site conditions'. This information is consistent with pre-marketing experience. See Table 4.

Adverse effects with vinorelbine IV.
In addition, some adverse effects have been observed with vinorelbine IV from pre- and post-marketing experience which were not reported with vinorelbine oral. It cannot be ruled out that these effects may also be experienced with the use of vinorelbine oral as with other vinca alkaloids. See Table 5.

Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
There is no known antidote for overdoses of oral vinorelbine. No case of overdosage has been reported with oral vinorelbine, however the primary anticipated complications of overdosage would consist of bone marrow suppression and peripheral neurotoxicity. If overdosage occurs, general supportive measures together with appropriate blood transfusions, growth factors, and antibiotics should be instituted as deemed necessary by the physician. A close monitoring of hepatic function is recommended.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Vinorelbine injection for intravenous administration is unavailable in this brand, however this dosage form is available in other brands. Clinical trial and pharmacokinetic information obtained using a vinorelbine intravenous formulation are also included in the following sections for prescriber information.
Mechanism of action.
Vinorelbine is an antineoplastic drug. It is a semi-synthetic member of the vinca alkaloid family that interferes with microtubule assembly. The vinca alkaloids are structurally similar compounds comprised of two multiringed units, vindoline and catharanthine. Unlike other vinca alkaloids, the catharanthine unit is the site of structural modification for vinorelbine. The antitumor activity of vinorelbine is thought to be due primarily to inhibition of mitosis at metaphase through its interaction with tubulin. In intact tectal plates from mouse embryos, vinorelbine, vincristine and vinblastine inhibited mitotic microtubule formation at the same concentration (2 microM), including a blockade of cells at metaphase. Vincristine produced depolymerisation of axonal tubules at 5 microM, but vinblastine and vinorelbine did not have this effect until concentrations of 30 microM and 40 microM, respectively. These data suggest relative selectivity of vinorelbine for mitotic microtubules.
Clinical trials.
Vinorelbine IV. Non-small cell lung cancer.
The activity of vinorelbine was investigated in a series of phase II trials. The overall response rate to vinorelbine single agent in NSCLC patients ranged from 8% to 33% in previously untreated patients. In the two major phase II trials with more than 60 evaluable patients, the overall response rate was over 30% in chemotherapy-naive patients. The high activity of vinorelbine as single agent in nonsmall cell lung cancer which was observed in non-controlled phase II studies has also been confirmed in three randomised phase III trials. In one prospective randomised study with 216 stage IV patients, vinorelbine was compared to 5-fluorouracil with leucovorin (considered equivalent to best supportive care for the purposes of the study).
The median survival time of patients who received vinorelbine was 30 weeks compared to 22 weeks for those on the 5-fluorouracil/leucovorin arm (log-rank p=0.03). The response rates were 12% for the vinorelbine arm and 3% for the fluorouracil/leucovorin arm.
The activity of vinorelbine in combination with cisplatin has been investigated in two randomised phase III trials in a total of 782 patients. In a two arm trial, vinorelbine was compared to vinorelbine with cisplatin. The overall response rate to vinorelbine as single agent was 16% while that of the combination vinorelbine/cisplatin was 43%. The median survival time for patients receiving vinorelbine as single agent was similar to that observed with vinorelbine and cisplatin.
In a large European clinical trial, 612 patients with Stage III or IV non-small cell lung cancer, no prior chemotherapy and WHO performance status of 0, 1 or 2 were randomised to treatment with single-agent vinorelbine (30 mg/m2/week), vinorelbine (30 mg/m2/week), cisplatin (120 mg/m2 days 1 and 29 then every 6 weeks), and vindesine (3 mg/m2/week for 7 weeks, then every second week) plus cisplatin (120 mg/m2 days 1 and 29 then every 6 weeks). Vinorelbine plus cisplatin produced longer survival times than vindesine plus cisplatin (median survival 40 weeks vs 32 weeks, p=0.03). The median survival time for patients receiving single-agent vinorelbine was similar to that observed with vindesine plus cisplatin (31 weeks vs 32 weeks). The 1-year survival rates were 36% for vinorelbine plus cisplatin, 27% for vindesine plus cisplatin and 30% for single-agent vinorelbine. The overall objective response rate (all partial responses) was significantly higher in patients treated with vinorelbine plus cisplatin (28%) than in those treated with vindesine plus cisplatin (19%, p=0.03) and in those treated with single-agent vinorelbine (14%, p < 0.001). The response rates reported for vindesine plus cisplatin and single-agent vinorelbine were not significantly different. Significantly, less nausea, vomiting, alopecia and neurotoxicity were observed in patients receiving single-agent vinorelbine compared to those receiving the combination of vindesine and cisplatin.
Advanced breast cancer - second line.
Twenty phase II studies of IV vinorelbine monotherapy have been performed as second line or subsequent treatment of advanced breast cancer patients. The response rate and duration of response to chemotherapy declines as patients progress through first, second and third line chemotherapy. Thirteen of these phase II studies were in mixed anthracycline-pretreated and anthracycline-naive populations, entering 494 patients and reporting overall response rates of 14 - 45% (patients weighted average = 29.2%) and median survival times of 58 - 69 weeks.
The remaining seven phase II studies were in anthracycline-pretreated patients, entering a total of 339 patients, reporting response rates of 16 - 64% (patient weighted average = 30.9%) and median survival was 24 - 82 weeks.
In a randomised phase III study conducted to investigate efficacy in anthracycline-refractory advanced breast cancer, 115 patients received IV vinorelbine as a single agent versus sixty four patients who received intravenous melphalan. The median dose, number of doses and duration of treatment for vinorelbine were 27.5 mg/m2, 9 doses and 12 weeks, respectively and for melphalan, 25 mg/m2, 2 doses and 8 weeks, respectively. Of those receiving vinorelbine, thirteen of 84 (15.5%) patients with measurable disease achieved an objective response compared with four of 46 (8.7%) receiving melphalan. Overall survival was 35 weeks for patients receiving vinorelbine compared with 31 weeks for those receiving melphalan (log-rank p=0.023). Neither treatment had an adverse effect on quality of life.
IV vinorelbine has also been studied in combination with other agents in the second-line treatment of advanced breast cancer. Results from trials are summarised in Table 6.
 Vinorelbine oral. Oral vinorelbine has been developed as a line extension of the IV dosage form. Hence, the primary objective of the clinical program was to demonstrate bioequivalence between the oral and intravenous formulations on the basis of pharmacokinetic studies. An oral dose of 80 mg/m2 was demonstrated to correspond to 30 mg/m2 of the IV formulation and 60 mg/m2 orally to 25 mg/m2 given by the IV route. Subsequent phase II studies were conducted to examine the efficacy and tolerance of oral vinorelbine.
Vinorelbine oral. Oral vinorelbine has been developed as a line extension of the IV dosage form. Hence, the primary objective of the clinical program was to demonstrate bioequivalence between the oral and intravenous formulations on the basis of pharmacokinetic studies. An oral dose of 80 mg/m2 was demonstrated to correspond to 30 mg/m2 of the IV formulation and 60 mg/m2 orally to 25 mg/m2 given by the IV route. Subsequent phase II studies were conducted to examine the efficacy and tolerance of oral vinorelbine.
Non-small cell lung cancer.
One randomised phase II study (97 CA 205) with the recommended oral dose regimen was conducted, comparing oral and IV vinorelbine in patients with advanced or metastatic NSCLC who had not previously been treated with cytotoxic chemotherapy. The results are summarised in Table 7.
 In a multicentre, phase II study of 56 patients in combination with cisplatin 100 mg/m2 (day 1 q 4 weeks), the weekly administration of vinorelbine (IV vinorelbine 25 mg/m2 day 1, oral vinorelbine 60 mg/m2 days 8, 15 and 22) produced a response rate of 30% for all registered patients and 33% for evaluable patients in the first line treatment of unresectable, localised or metastatic NSCLC. Median progression-free survival and survival were 5.5 and 8.9 months, respectively.
In a multicentre, phase II study of 56 patients in combination with cisplatin 100 mg/m2 (day 1 q 4 weeks), the weekly administration of vinorelbine (IV vinorelbine 25 mg/m2 day 1, oral vinorelbine 60 mg/m2 days 8, 15 and 22) produced a response rate of 30% for all registered patients and 33% for evaluable patients in the first line treatment of unresectable, localised or metastatic NSCLC. Median progression-free survival and survival were 5.5 and 8.9 months, respectively.
Advanced breast cancer. As a single agent.
Two non-comparative multi-centre phase II studies (96 CA 201 and 97 CA 206) of oral vinorelbine given weekly at a dose of 60 mg/m2 for the first 3 weeks and then increased to 80 mg/m2 as a single agent, were conducted in the first line treatment of advanced breast cancer. A total of 184 patients were enrolled.
The majority of patients had metastatic disease at study entry, visceral lesions and had received prior hormonotherapy. The proportion of patients having received prior neo/adjuvant chemotherapy was 49.2% (96 CA 201) and 26.5% (97 CA 206). No patient had received prior chemotherapy for advanced/metastatic disease.
The efficacy results are summarised in Table 8.
 A response rate of 30% was reported in Study 97 CA 206 and 21% in Study 96 CA 201. The lower response rate observed in the latter study could be explained by the very poor prognosis features in the patients having received no prior adjuvant chemotherapy: 57% of patients had stage IIIB - IV disease, 67% had a disease-free interval less than 2 years and 62% had at least 3 organs involved. In this subset of patients, the response rate was only 14%.
A response rate of 30% was reported in Study 97 CA 206 and 21% in Study 96 CA 201. The lower response rate observed in the latter study could be explained by the very poor prognosis features in the patients having received no prior adjuvant chemotherapy: 57% of patients had stage IIIB - IV disease, 67% had a disease-free interval less than 2 years and 62% had at least 3 organs involved. In this subset of patients, the response rate was only 14%.
Median durations of progression-free survival and overall survival were consistent in the 2 studies: 4.6 and 4.2 months; and 19.3 and 23.9 months, respectively.
A randomised phase II trial (CA221) assessed in parallel the efficacy and safety of oral vinorelbine at 60 mg/m2 on days 1 and 8 every 3 weeks for the first cycle then increased to 80 mg/m2 on days 1 and 8 every 3 weeks for subsequent cycles; and IV vinorelbine at 25 mg/m2 on days 1 and 8 every 3 weeks for the first cycle then increased to 30 mg/m2 on days 1 and 8 every 3 weeks for subsequent cycles. The study was closed 2.5 years after its initiation due to low accrual (85 patients enrolled out of 230 planned). Premature closure of the study did not allow for the accurate assessment of the efficacy of oral vinorelbine. The following results were observed: response rate of 7% and disease control rate of 47.4% in the oral arm; response rate of 22% and disease control rate of 51.9% in the IV arm. Median overall survival was similar in the 2 study arms: 9.4 months and 10.2 months, respectively.
In combination.
A randomised phase II study (CA 222) of the combination of oral vinorelbine with capecitabine versus a sequential regimen of oral vinorelbine and capecitabine versus the combination of docetaxel and capecitabine was carried out in 139 metastatic breast cancer patients previously treated with anthracyclines.
In combination with capecitabine, oral vinorelbine was given on days 1 and 8 of three-week cycles at 60 mg/m2 for the first cycle and then at 80 mg/m2. In the sequential regimen, patients received oral vinorelbine 60 then 80 mg/m2/week for a total of 3 three-week cycles and then 3 cycles of capecitabine. In the control arm, the standard regimen of docetaxel 75 mg/m2 on day 1 and capecitabine every 3 weeks was used. In the 3 study arms, the dose of capecitabine was 2000 mg/m2/day administered from days 1 to 14 every 3 weeks.
Efficacy results in the 3 study arms are summarised in Table 9.
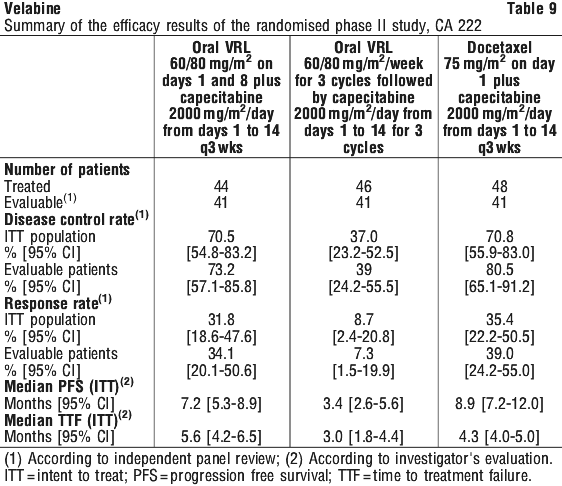 The two combination arms, oral vinorelbine plus capecitabine and docetaxel plus capecitabine, produced similar disease control rates (70.5% vs 70.8% in the ITT population), similar response rates (31.8% vs 35.4%), similar progression-free survival (7.2 months vs 8.9 months) and similar time-to-treatment failure (5.6 months vs 4.3 months). In comparison, the sequential regimen of oral vinorelbine followed by capecitabine was inferior to the combination regimens for all the efficacy parameters considered.
The two combination arms, oral vinorelbine plus capecitabine and docetaxel plus capecitabine, produced similar disease control rates (70.5% vs 70.8% in the ITT population), similar response rates (31.8% vs 35.4%), similar progression-free survival (7.2 months vs 8.9 months) and similar time-to-treatment failure (5.6 months vs 4.3 months). In comparison, the sequential regimen of oral vinorelbine followed by capecitabine was inferior to the combination regimens for all the efficacy parameters considered.
Oral vinorelbine in combination with other cytotoxics was assessed in three phase I/II studies: with epirubicin (Study CA 205); docetaxel (CA 101); and paclitaxel (CA 102).
The efficacy results of these studies are summarised in Table 10.
 The two regimens alternating oral and IV vinorelbine in combination with epirubicin or docetaxel gave similar response rates of approximately 50%. Median durations of overall survival tended to be longer for the taxane combinations than for the combination with epirubicin.
The two regimens alternating oral and IV vinorelbine in combination with epirubicin or docetaxel gave similar response rates of approximately 50%. Median durations of overall survival tended to be longer for the taxane combinations than for the combination with epirubicin.
5.2 Pharmacokinetic Properties
Absorption.
Following oral administration, vinorelbine is promptly absorbed and the Tmax is reached within 1.5 to 3 hours with a blood concentration peak (Cmax) of approximately 130 nanogram/mL after dosing at 80 mg/m2. The absolute bioavailability is about 40% and simultaneous intake of a low fat standard meal does not modify the area under the concentration-time curve (AUC). The effect of a high fat meal on absorption has not been studied.
Vinorelbine oral 60 and 80 mg/m2 leads to a comparable AUC to that obtained from 25 and 30 mg/m2 of the IV formulation, respectively. Interindividual variability of the AUC is similar after administration by both the IV and oral routes. There is a proportional increase between the AUC and dose.
Distribution.
The mean pharmacokinetic parameters were evaluated in blood. After intravenous administration, the terminal half-life averaged 38 hours. Blood clearance was high, approached liver blood flow and averaged 0.72 L/hr/kg (range: 0.32 - 1.26 L/hr/kg), while steady state volume of distribution was large, averaged 21.2 L/kg (range: 7.5 - 39.7 L/kg), and indicated extensive tissue distribution.
Vinorelbine binds extensively to blood cells and especially platelets (70 - 80%), but less extensively (about 15%) to plasma proteins. There is a significant uptake of vinorelbine in lungs, as assessed by pulmonary surgical biopsies showing up to a 300 fold greater concentration than in serum. Vinorelbine has not been detected in the central nervous system.
Metabolism.
Vinorelbine is mostly metabolised by the CYP 3A4 isoform of the cytochrome P450. All the metabolites have been identified and none are active except 4-O-deacetylvinorelbine which is the main metabolite in blood. No sulfo- or glucurono- conjugates are observed.
Excretion.
Renal elimination is low (< 20% of the dose) and consists mostly of the parent compound. Biliary excretion is the predominant elimination route of both metabolites and unchanged vinorelbine, which is the main recovered compound.
Renal impairment.
The effect of renal dysfunction on vinorelbine disposition has not been assessed, however, dose reduction, in the presence of renal insufficiency is not indicated with vinorelbine due to its low renal elimination.
Hepatic impairment.
Vinorelbine is cleared from the circulation primarily by the liver, and therefore elevated blood concentrations may be expected in patients with hepatic impairment. In a phase I pharmacokinetic study, 6 subjects with severe hepatic impairment were treated with 20 mg/m2 intravenously. Blood concentrations were elevated compared to historical data from patients with normal hepatic function. Oral vinorelbine is contraindicated in patients with severe hepatic impairment. In studies in patients with mild and moderate hepatic impairment, the pharmacokinetics of orally administered vinorelbine were not modified after administration of: 60 mg/m2 in patients with mild hepatic impairment (bilirubin < 1.5 x ULN and ALT and/or AST between 1.5 and 2.5 x ULN) and 50 mg/m2 in patients with moderate hepatic impairment (bilirubin between 1.5 and 3 x ULN, independent of ALT and AST levels). Haematological toxicity should be closely monitored.
Pharmacokinetics/pharmacodynamic relationships.
A strong relationship was demonstrated between AUC and leucocyte or PMN decreases.
5.3 Preclinical Safety Data
Genotoxicity.
Vinorelbine tartrate has been shown to affect chromosome number and possibly structure in vivo (polyploidy in bone marrow cells from Chinese hamsters and a positive micronucleus test in mice).
It was not mutagenic or cytotoxic in a reverse histidine mutation (Ames) test but showed mutagenic potential in a mouse forward mutation (TK locus) test.
Carcinogenicity.
Carcinogenicity studies in mice and rats showed no tumorigenic activity at dose levels up to 2.4 mg/m2 given by IV injection every two weeks for 18 months or two years, respectively. However, the positive findings in genetic toxicity assays suggest that the drug may have carcinogenic potential at the higher dose level used in humans.6 Pharmaceutical Particulars
6.1 List of Excipients
Velabine soft capsules also contain the following excipients: ethanol, medium chain triglycerides, purified water, polysorbate 80 and macrogol 400. The soft capsule shell contains: gelatin, sorbitol, titanium dioxide, iron oxide yellow (20 mg and 80 mg capsules only), and iron oxide red (30 mg capsules only).
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2 to 8°C (Refrigerate. Do not freeze) in the original container.
6.5 Nature and Contents of Container
PVC/PVDC/Al blister packs.
Pack size: 1 capsule.
The blister packs are child-resistant and comply with European Standard EN 14375:2003 Child-resistant Non-reclosable Packaging for Pharmaceutical Products - Requirements and Testing.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Vinorelbine tartrate is a semi-synthetic vinca alkaloid with antitumor activity. The chemical name is 3',4'-didehydro-4'-deoxy-C'-norvincaleukoblastine [R-(R*,R*) - 2,3 dihydroxybutanedioate (1:2) (salt)]. Vinorelbine tartrate is a white to yellow or light brown amorphous powder with the molecular formula C45H54N4O8.2C4H6O6 and a molecular weight of 1079.12. The aqueous solubility is > 1000 mg/mL in distilled water.
Chemical structure.

CAS number.
125317-39-7.7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Summary Table of Changes


 For any administration planned at the 80 mg/m2/week dose, if the neutrophil count falls below 500/mm3 or more than once between 500 and 1000/mm3, the dose must be delayed until recovery and reduced from 80 to 60 mg/m2 per week during the 3 subsequent administrations. See Table 2.
For any administration planned at the 80 mg/m2/week dose, if the neutrophil count falls below 500/mm3 or more than once between 500 and 1000/mm3, the dose must be delayed until recovery and reduced from 80 to 60 mg/m2 per week during the 3 subsequent administrations. See Table 2. It is possible to re-escalate the dose from 60 to 80 mg/m2/week if the neutrophil count does not drop below 500/mm3, or more than once between 500 and 1000/mm3 during the three administrations given at the 60 mg/m2 dose.
It is possible to re-escalate the dose from 60 to 80 mg/m2/week if the neutrophil count does not drop below 500/mm3, or more than once between 500 and 1000/mm3 during the three administrations given at the 60 mg/m2 dose. Even for patients with a body surface area (BSA) ≥ 2 m2, the dose should never exceed 120 mg per week at 60 mg/m2 and 160 mg per week at 80 mg/m2.
Even for patients with a body surface area (BSA) ≥ 2 m2, the dose should never exceed 120 mg per week at 60 mg/m2 and 160 mg per week at 80 mg/m2.


 In a multicentre, phase II study of 56 patients in combination with cisplatin 100 mg/m2 (day 1 q 4 weeks), the weekly administration of vinorelbine (IV vinorelbine 25 mg/m2 day 1, oral vinorelbine 60 mg/m2 days 8, 15 and 22) produced a response rate of 30% for all registered patients and 33% for evaluable patients in the first line treatment of unresectable, localised or metastatic NSCLC. Median progression-free survival and survival were 5.5 and 8.9 months, respectively.
In a multicentre, phase II study of 56 patients in combination with cisplatin 100 mg/m2 (day 1 q 4 weeks), the weekly administration of vinorelbine (IV vinorelbine 25 mg/m2 day 1, oral vinorelbine 60 mg/m2 days 8, 15 and 22) produced a response rate of 30% for all registered patients and 33% for evaluable patients in the first line treatment of unresectable, localised or metastatic NSCLC. Median progression-free survival and survival were 5.5 and 8.9 months, respectively. A response rate of 30% was reported in Study 97 CA 206 and 21% in Study 96 CA 201. The lower response rate observed in the latter study could be explained by the very poor prognosis features in the patients having received no prior adjuvant chemotherapy: 57% of patients had stage IIIB - IV disease, 67% had a disease-free interval less than 2 years and 62% had at least 3 organs involved. In this subset of patients, the response rate was only 14%.
A response rate of 30% was reported in Study 97 CA 206 and 21% in Study 96 CA 201. The lower response rate observed in the latter study could be explained by the very poor prognosis features in the patients having received no prior adjuvant chemotherapy: 57% of patients had stage IIIB - IV disease, 67% had a disease-free interval less than 2 years and 62% had at least 3 organs involved. In this subset of patients, the response rate was only 14%. The two combination arms, oral vinorelbine plus capecitabine and docetaxel plus capecitabine, produced similar disease control rates (70.5% vs 70.8% in the ITT population), similar response rates (31.8% vs 35.4%), similar progression-free survival (7.2 months vs 8.9 months) and similar time-to-treatment failure (5.6 months vs 4.3 months). In comparison, the sequential regimen of oral vinorelbine followed by capecitabine was inferior to the combination regimens for all the efficacy parameters considered.
The two combination arms, oral vinorelbine plus capecitabine and docetaxel plus capecitabine, produced similar disease control rates (70.5% vs 70.8% in the ITT population), similar response rates (31.8% vs 35.4%), similar progression-free survival (7.2 months vs 8.9 months) and similar time-to-treatment failure (5.6 months vs 4.3 months). In comparison, the sequential regimen of oral vinorelbine followed by capecitabine was inferior to the combination regimens for all the efficacy parameters considered. The two regimens alternating oral and IV vinorelbine in combination with epirubicin or docetaxel gave similar response rates of approximately 50%. Median durations of overall survival tended to be longer for the taxane combinations than for the combination with epirubicin.
The two regimens alternating oral and IV vinorelbine in combination with epirubicin or docetaxel gave similar response rates of approximately 50%. Median durations of overall survival tended to be longer for the taxane combinations than for the combination with epirubicin.
