1 Name of Medicine
Calcifediol monohydrate.
2 Qualitative and Quantitative Composition
Each Vistella capsule contains 255 micrograms of calcifediol (present as calcifediol monohydrate).
For the full list of excipients, see Section 6.1.
3 Pharmaceutical Form
Vistella is an orange, soft gelatin capsule containing a clear, low viscous liquid and free from particles.
4.1 Therapeutic Indications
Treatment of vitamin D deficiency in adults, and maintenance treatment as required.
4.2 Dose and Method of Administration
Dosage.
Treatment of vitamin D deficiency in adults, and maintenance treatment as required: one capsule (255 micrograms of calcifediol) once a month.
Higher doses may be necessary in some patients after analytical verification of the extent of vitamin D deficiency. In these cases, the maximum dose administered should not exceed one capsule per fortnight.
Once the plasma levels of 25(OH)D are stabilised within the desired range, the treatment should be discontinued, or the frequency of administration lowered.
Dietary intake of vitamin D and sun exposure vary among patients and should be taken into account when calculating the appropriate dose of vitamin D analogue drugs such as calcifediol.
The dose, frequency and duration of Vistella treatment will be determined by the prescriber taking into account the plasma levels of 25(OH)D and other clinically relevant factors including comorbidities. Serum concentrations of 25(OH)D should be monitored after initiation of the treatment, usually after 3-4 months or as per doctor's recommendations.
Vistella must not be administered daily.
The potency of Vistella is sometimes expressed in international units. These units are not interchangeable with the units used to express the potency of cholecalciferol (Vitamin D) preparations (see Section 4.4).
Method of administration.
Oral administration.
Paediatric population.
The efficacy and safety of Vistella have not been established in the paediatric population. Use of this product is not recommended in the paediatric population.4.3 Contraindications
Hypersensitivity to the active ingredient or to any of the excipients listed in Section 6.1.
Hypercalcemia (serum calcium > 2.620 mmol/L) or hypercalciuria.
Calcium lithiasis.
Hypervitaminosis D.
4.4 Special Warnings and Precautions for Use
In order to obtain an adequate clinical response to oral administration of calcifediol, an appropriate dietary calcium intake is also required. Therefore, to control the therapeutic effects, the following parameters should be monitored, in addition to 25(OH)D: serum calcium, phosphorus and alkaline phosphatase as well as urinary calcium and phosphorus in 24 hours.
A decrease in serum levels of alkaline phosphatase normally precedes the onset of hypercalcemia.
Once parameters are stabilised and the patient is under maintenance treatment, the above mentioned tests should be performed regularly, especially for serum levels of 25(OH)D and calcium.
Use in renal impairment.
To be administered with caution. Use of Vistella in patients with chronic kidney disease should be accompanied by periodic monitoring of serum calcium and phosphorus, and hypercalcemia prevention. Transformation to calcitriol takes place in the kidney; thus, in case of severe renal impairment (creatinine clearance of less than 30 mL/min) a very significant reduction in the pharmacological effects may occur.
Heart failure.
Special caution is required. The patient's serum calcium should be monitored constantly, especially in patients on digitalis, because hypercalcemia may occur and arrhythmias appear. Twice weekly tests are recommended at the beginning of treatment.
Hypoparathyroidism.
1-alpha-hydroxylase is activated by parathyroid hormone. As a result, in case of parathyroid insufficiency the activity of calcifediol may decrease.
Primary hyperparathyroidism.
Use of vitamin D by people with primary hyperparathyroidism increases the risk of hypercalcaemia and hypercalciuria. Serum calcium should be monitored closely.
Kidney stones.
Calcemia should be monitored, since vitamin D increases absorption of calcium and may aggravate the situation. In these patients supplements of vitamin D should be administered only if the benefits outweigh the risks.
In patients with prolonged immobilisation it may be necessary to reduce the dose in order to avoid hypercalcemia.
Patients with sarcoidosis, tuberculosis or other granulomatous diseases: to be administered with caution, since these conditions lead to a greater sensitivity to the effect of vitamin D as well as to an increase of the risk of adverse effects at doses lower than the recommended dose. It is necessary to monitor serum and urinary calcium concentrations in these patients.
Patients and their families and/or caregivers should be informed of the importance of complying with the prescribed dosage and with recommendations about diet and concomitant intake of calcium supplements in order to prevent overdosing.
Effects on laboratory tests.
Patients should be warned that this drug contains a component that can alter the results of laboratory tests:
Determination of cholesterol.
Calcifediol may interfere with Zlatkis-Zak method, leading to false increases in serum cholesterol levels.
Warnings on excipients.
This medicine contains 1% ethanol (alcohol), which corresponds to 4.98 mg/capsule.
This medicine contains 31.3 mg sorbitol in each capsule. Patients with hereditary fructose intolerance should not take this medicinal product.
This medicine may cause allergic reactions because it contains sunset yellow (E-110). It can cause asthma, especially in patients allergic to acetylsalicylic acid.
International Units (IU) should not be used for determination of the dose of calcifediol as this could lead to overdosing. Instead, the dosing recommendation in Section 4.2 should be followed.4.5 Interactions with Other Medicines and Other Forms of Interactions
Phenytoin, phenobarbital, primidone and other enzyme inducers.
Enzyme inducers may reduce plasma concentrations of calcifediol and inhibit its effects by inducing its hepatic metabolism. For this reason, it is generally recommended to monitor plasma 25-OH-D levels when calcifediol is administered with antiepileptics that are CYP3A4 inducers in order to consider supplementation.
Cardiac glycosides.
Calcifediol can cause hypercalcemia, which can, in turn, enhance the inotropic effects of digoxin and its toxicity, producing cardiac arrhythmias.
Drugs that decrease the absorption of calcifediol such as cholestyramine, colestipol or orlistat, which can result in decreased effects. It is recommended to space doses of these medicines and vitamin D supplements for at least 2 hours.
Paraffin and mineral oil.
Due to liposolubility of calcifediol, the product can dissolve in paraffin and intestinal absorption may decrease. Using other types of laxatives or at least spacing doses is recommended.
Thiazide diuretics.
Co-administration of a thiazide diuretic (hydrochlorothiazide) with vitamin D supplements in patients with hypoparathyroidism may lead to hypercalcemia, which may be temporary or require the interruption of the treatment with the vitamin D analogue.
Some antibiotics, such as penicillin, neomycin and chloramphenicol can increase calcium absorption.
Phosphate-binding agents such as magnesium salts.
Since vitamin D has an effect on phosphate transport in the intestine, kidney and bone, hypermagnesemia may occur. The dosage of agents that bind to phosphate shall be adjusted according to phosphate concentrations in serum.
Verapamil.
Some studies show potential inhibition of antianginal action, due to antagonism of their actions.
Vitamin D.
Co-administration of any vitamin D analogue should be avoided as additive effects and hypercalcemia can occur.
Calcium supplements.
Uncontrolled intake of additional preparations containing calcium should be avoided.
Corticosteroids.
They counteract the effects of vitamin D analogue drugs such as calcifediol.
Interaction with food and drinks.
Food supplemented with vitamin D should be taken into account, since additive effects may occur.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
Oral calcifediol had no effect on male or female fertility in rats at doses up to 40 microgram/kg/day (42 times the clinical dose on a body surface area basis).
(Category B3)
Do not use calcifediol 255 micrograms soft capsules during pregnancy.
No controlled studies with calcifediol in pregnant women have been performed.
There are few studies in humans with vitamin D, including calcifediol. Vitamin D can cause hypercalcemia in pregnant women, which could lead to a syndrome of supravalvar aortic stenosis, retinopathy and intellectual disability in infants and newborn. An increased incidence of fetal skeletal abnormalities was seen in rats and rabbits when calcifediol was administered orally at ≥ 12 microgram/kg/day and 25 microgram/kg/day (12 and 52 times the clinical dose on a body surface area basis), respectively during the period of organogenesis.
Calcifediol is excreted into breast milk and should not be used during breast-feeding.
The risk in newborns/infants cannot be excluded. Maternal ingestion of high doses of calcifediol can produce high levels of calcitriol in milk and cause hypercalcemia in infants.4.7 Effects on Ability to Drive and Use Machines
Calcifediol capsules have no or negligible influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
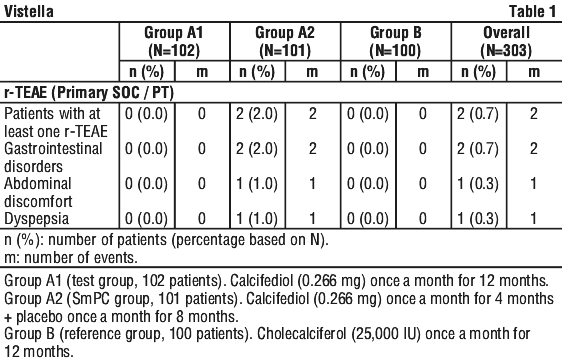
Adverse reactions to calcifediol are generally uncommon (≥ 1/1,000 to < 1/100) but sometimes they can be common (≥ 1/100 to < 1/10).
There were no adverse events with a frequency of very common (≥ 1/10) and common (≥ 1/100 to < 1/10) related to Vistella during the clinical development in the randomized, double-blind phase 3 study (n = 303).
Other adverse reactions at least possibly related to Vistella and reported at a frequency of uncommon (≥ 1/1,000 to < 1/100) during the clinical development include:
Gastrointestinal disorders.
Abdominal discomfort and dyspepsia. See Table 1.
 The most significant adverse effects are related to excessive intake of vitamin D, i.e. they are often associated with overdose or prolonged treatment, especially when associated with high doses of calcium.
The most significant adverse effects are related to excessive intake of vitamin D, i.e. they are often associated with overdose or prolonged treatment, especially when associated with high doses of calcium.
The doses of vitamin D analogues required for hypervitaminosis vary considerably from one subject to another. The most common adverse reactions are due to the hypercalcemia which can occur initially or at a later stage:
Endocrine disorders.
Pancreatitis, among the late symptoms of hypercalcemia.
Metabolism and nutrition disorders.
Elevation of blood urea nitrogen, albuminuria, hypercholesterolemia, hypercalcemia.
Nervous system disorders.
In case of moderate hypercalcemia the following symptoms may appear: weakness, fatigue, drowsiness, headache, irritability.
Eye disorders.
Rarely (≥ 1/10,000 to < 1/1,000), at very high doses photophobia and conjunctivitis with corneal calcifications may occur.
Cardiac disorders.
In case of hypercalcemia cardiac arrhythmias may occur.
Gastrointestinal disorders.
Nausea, vomiting, dry mouth, constipation, taste disturbances, with a metallic taste, abdominal cramps. If hypercalcemia progresses anorexia may occur.
Hepatobiliary disorders.
High calcemia levels can lead to increased transaminase (AST and ALT).
Musculoskeletal and connective tissue disorders.
Bone and muscle pain may occur in early stages of hypercalcemia, calcification in soft tissues.
Renal and urinary disorders.
Manifestations of hypercalcemia are: nephrocalcinosis and deterioration of kidney function (with polyuria, polydipsia, nocturia and proteinuria).
General disorders and alterations in the place of administration.
Later symptoms of hypercalcemia include: rhinorrhea, pruritus, hyperthermia, decreased libido.
Reporting suspected adverse events.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Symptoms.
Administration of vitamin D in high doses or for long periods of time may cause hypercalcemia, hypercalciuria, hyperphosphatemia and renal failure. As early symptoms of overdose, weakness, fatigue, drowsiness, headache, anorexia, dry mouth, metallic taste, nausea, vomiting, abdominal cramps, polyuria, polydipsia, nocturia, constipation or diarrhea, dizziness, tinnitus, ataxia, rash, hypotonia (especially in children), muscle or bone pain and irritability may appear.
Among later symptoms of hypercalcemia the following are included: runny nose, itching, decreased libido, nephrocalcinosis, renal failure, osteoporosis in adults, growth retardation in children, weight loss, anaemia, conjunctivitis with calcification, photophobia, pancreatitis, elevated blood urea nitrogen, albuminuria, hypercholesterolemia, increased transaminases (AST and ALT), hyperthermia, generalised vascular calcification, convulsions, soft tissue calcification. Rarely, patients may develop hypertension or psychotic symptoms; serum alkaline phosphatase may decrease; electrolyte imbalances together with moderate acidosis can lead to cardiac arrhythmias.
In the most serious cases, where serum calcium exceeds 2.994 mmol/L, syncope, metabolic acidosis and coma may happen. Although symptoms of overdose are usually reversible an overdose might lead to kidney or heart failure.
It is accepted that serum levels of 25-OH-cholecalciferol above 150 nanogram/mL may be associated with an increased incidence of adverse effects.
Increased calcium, phosphate, albumin and urea nitrogen in blood as well as cholesterol and blood transaminases are typical of this kind of overdose.
Treatment.
Treatment of calcifediol overdose consists of:
1. Withdrawal of treatment (with calcifediol) and with any calcium supplement being administered.
2. Follow a diet low in calcium. Administration of large volumes of liquids, both orally and parenterally, is advisable to increase calcium excretion. If necessary, administer steroids and induced forced diuresis with loop diuretics such as furosemide.
3. If intake has occurred in the previous 2 hours, gastric emptying and forced emesis are advisable. If vitamin D has already passed through the stomach, a laxative (paraffin or mineral oil) can be administered. If vitamin D has already been absorbed, hemodialysis or peritoneal dialysis with a dialysis solution free of calcium can be performed.
Hypercalcemia derived from prolonged administration of calcifediol persists for approximately 4 weeks after discontinuation of treatment. Signs and symptoms of hypercalcemia are usually reversible. However, metastatic calcification can cause serious kidney or heart failure and death.5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Vitamin D and analogues, ATC code: A11CC06.
Mechanism of action.
Vitamin D has two main forms: D2 (ergocalciferol) and D3 (cholecalciferol). Vitamin D3 is synthesised in the skin by exposure to sunlight (ultraviolet radiation) and is obtained from the diet. Vitamin D3 must undergo a two-step metabolic process to be active; the first step occurs in the microsomal fraction of the liver where vitamin D is hydroxylated at position 25 (25-hydroxycholecalciferol or calcifediol); the second step takes place in the kidney where 1,25-dihydroxycholecalciferol or calcitriol is formed due to the activity of enzyme 25-hydroxycholecalciferol 1-hydroxylase; conversion to 1,25-dihydroxycholecalciferol is regulated by its own concentration, by parathyroid hormone (PTH) and by serum calcium and phosphate concentration. Other metabolites with unknown function exist. 1,25-dihydroxycholecalciferol is transported from the kidney to target tissues (intestine, bone and possibly kidney and parathyroid gland) by binding to specific plasma proteins.
Calcifediol is a precursor of the active form of vitamin D (1,25-dihydroxycholecalciferol or calcitriol) and its pharmacodynamic effects are expected to be compatible with known biological roles of vitamin D.
Pharmacodynamic effects.
Vitamin D increases absorption of calcium and phosphorus in the intestine and improves normal bone formation and mineralization and acts on three levels:
Intestine: vitamin D enhances absorption of calcium and phosphorus in the small intestine. Bone: calcitriol enhances bone formation by increasing levels of calcium and phosphate and stimulates action of osteoblasts.
Kidney: calcitriol enhances tubular reabsorption of calcium.
Parathyroid glands: vitamin D inhibits the secretion of parathyroid hormone.
Clinical efficacy and safety.
The efficacy and safety of calcifediol 266 micrograms soft capsules was evaluated in a randomised, double blind study in post-menopausal women with vitamin D deficiency (25(OH)D levels < 50 nmol/L). 303 subjects were randomised and 298 conformed the intention to treat population. Patients were treated with calcifediol 266 micrograms/month or cholecalciferol at recommended therapeutic guidelines dose (25,000 IU/month). After 1 month, 13.5% of patients treated with calcifediol achieved 25(OH)D levels greater than 30 nanogram/mL (75 nmol/L) and 59.1% achieved levels greater than 20 nanogram/mL (50 nmol/L). After 4 months 35% of patients treated with calcifediol achieved 25(OH)D levels greater than 30 nanogram/mL (75 nmol/L) and 81% achieved levels greater than 20 nanogram/mL (50 nmol/L). The mean (SD) increase in 25(OH)D concentrations with respect to baseline levels was 9.7 (6.7) nanogram/mL (24.3 (16.8) nmol/L) after 1 month and 14.9 (8.1) nanogram/mL (37.3 (20.3) nmol/L) after 4 months of treatment. Highest 25(OH)D levels with calcifediol were achieved after 4 months of treatment and remained stable for 12 months in the long term, indicating a non-accumulative effect. Calcifediol was safe and well tolerated.
5.2 Pharmacokinetic Properties
Absorption.
Calcifediol monohydrate is well absorbed in the intestine, not being dependent on the presence of bile acids and micelle formation as is cholecalciferol. Following oral administration of calcifediol, the maximum serum concentration of 25-hydroxyvitamin D is reached approximately after 5.5 hours.
Distribution.
Calcifediol circulates in the blood bound to a specific α-globulin (DBP). It is stored in adipose tissue and muscle for prolonged periods. Due to its lower lipophilicity, storage of calcifediol in adipose tissue is lower than for vitamin D.
Metabolism.
Both calcifediol and calcitriol are inactivated in all target tissues by hydroxylation to 24,25-OH2D via CYP24A1. This is followed by a number of oxidation steps, eventually producing calcitroic acid in humans.
Excretion.
Calcifediol monohydrate half-life is around 12 to 21 days, and it is primarily excreted in the bile.
5.3 Preclinical Safety Data
Genotoxicity.
Calcifediol was not genotoxic in bacterial reverse mutation assays, in vitro clastogenicity assay in mouse lymphoma cells or human lymphocytes and an in vivo micronucleus test in rats.
Carcinogenicity.
No carcinogenicity studies were conducted. The weight of evidence indicates that the carcinogenic potential of calcifediol monohydrate is unlikely.6 Pharmaceutical Particulars
6.1 List of Excipients
Ethanol, anhydrous; medium chain triglycerides; gelatin; glycerol; sorbitol (70%); titanium dioxide (E171); sunset yellow (E-110).
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
This medicine is packed in PVC/PVDC-Al blisters containing 1 (starter pack), 3, 4, 5, 6, 10 and 12 capsules. Please note that not all pack sizes are available in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
CAS number.
19356-17-3.7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes


 The most significant adverse effects are related to excessive intake of vitamin D, i.e. they are often associated with overdose or prolonged treatment, especially when associated with high doses of calcium.
The most significant adverse effects are related to excessive intake of vitamin D, i.e. they are often associated with overdose or prolonged treatment, especially when associated with high doses of calcium.
