1 Name of Medicine
Melatonin.
2 Qualitative and Quantitative Composition
Voquily Jet Lag Relief capsules contain 2 mg melatonin.
Excipients with known effect.
Gelatin - may contain traces of sulfites.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
2 mg.
Opaque hard gelatin capsules with a white body and light blue cap.4.1 Therapeutic Indications
Short-term treatment of jet lag in adults aged 18 and over.
4.2 Dose and Method of Administration
Dosage.
The recommended dose is 2 mg once daily taken between 8 pm and 4 am (at the preferred local sleep time of the travel destination). Taking prior to departure may cause unwanted drowsiness and may be no more effective.
Capsules should be swallowed whole on an empty stomach, at least 1 hour after food.
Voquily Jet Lag Relief should be taken for the shortest period of time, with a maximum duration of 5 days.
Method of administration.
Food can enhance the increase in plasma melatonin concentration (see Section 5.2). Intake of melatonin with carbohydrate-rich meals may impair blood glucose control for several hours. It is recommended that Voquily Jet Lag Relief is administered on an empty stomach and food is not consumed 1 hour before and 1 hour after intake of Voquily Jet Lag Relief.
Voquily Jet Lag Relief capsules should be swallowed whole with fluid.4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed, see Section 6.1.
4.4 Special Warnings and Precautions for Use
Drowsiness.
Melatonin may cause daytime drowsiness. Therefore, Voquily Jet Lag Relief should be used with caution if the effects of drowsiness are likely to be associated with a risk to patient safety.
Autoimmune diseases.
There is no data concerning the use of melatonin in patients with autoimmune diseases. Therefore, Voquily Jet Lag Relief is not recommended in patients with autoimmune diseases.
Seizure risk.
Melatonin may increase seizure frequency in patients experiencing seizures (e.g. epileptic patients). Patients suffering from seizures must be informed about this possibility before using melatonin. Melatonin may promote or increase the incidence of seizures in children and adolescents with multiple neurological defects.
Use in hepatic impairment.
Limited data indicates that daytime endogenous blood melatonin concentration is markedly elevated in patients with hepatic impairment due to reduced clearance. Therefore, Voquily Jet Lag Relief is not recommended for patients with hepatic impairment.
Use in renal impairment.
Limited data is available for the use of melatonin in patient with renal impairment. Therefore, Voquily Jet Lag Relief is not recommended for patients with renal impairment.
Use in the elderly.
Melatonin metabolism is known to decline with age. Across a range of doses, higher AUC and Cmax levels have been reported in older subjects compared to younger subjects, reflecting the lower metabolism in the elderly. Also, see Section 5.2 Pharmacokinetic Properties, Elderly.
Paediatric use.
Efficacy and safety of Voquily Jet Lag Relief in children for the treatment of jetlag has not been established.
Effects on laboratory tests.
No information is available on the effect of melatonin on laboratory tests.4.5 Interactions with Other Medicines and Other Forms of Interactions
Melatonin is metabolised primarily through the CYP1A1 and CYP1A2 enzymes. As a result, drugs that affect CYP enzymes which are involved in melatonin's metabolism could potentially affect melatonin's exposure with or without potential clinical consequences. However, there is a limited data available in terms of melatonin's potential for pharmacokinetic interactions.
Hepatic enzymes.
Melatonin has been observed to induce CYP3A in vitro at supra-therapeutic concentrations. The clinical relevance of the finding is unknown. If induction occurs, plasma concentrations of concomitantly administered drugs can be reduced.
Melatonin does not appear to induce CYP1A enzymes in vitro at supra-therapeutic concentrations. Therefore, interactions between melatonin and other active substances as a consequence of melatonin's effect on CYP1A enzymes are not likely to be significant.
Melatonin's metabolism is mainly mediated by CYP1A enzymes. Therefore, interactions between melatonin and other active substances as a consequence of their effect on CYP1A enzymes is possible:
Quinolones.
CYP1A2 inhibitors such as quinolones may give rise to increased melatonin exposure.
Carbamazepine and rifampicin.
CYP1A2 inducers such as carbamazepine and rifampicin may give rise to reduced plasma concentrations of melatonin.
Fluvoxamine.
Caution should be exercised in patients on fluvoxamine, which increases melatonin levels (17-fold higher AUC and 12-fold higher serum Cmax) by inhibiting its metabolism by hepatic cytochrome P450 (CYP) isozymes CYP1A2 and CYP2C19. The combination should be avoided.
5- or 8-methoxypsoralen.
Caution should be exercised in patients on 5- or 8-methoxypsoralen (5 and 8-MOP), which increases melatonin levels by inhibiting its metabolism.
Cimetidine.
Caution should be exercised in patients on cimetidine, a CYP2D inhibitor which increases plasma melatonin levels by inhibiting its metabolism.
Oestrogens.
Caution should be exercised in patients on oestrogens (e.g. contraceptives or hormone replacement therapy), which increase melatonin levels by inhibiting its metabolism by CYP1A1 and CYP1A2.
Thioridazine and imipramine.
Melatonin has been co administered in studies with thioridazine and imipramine, active substances which affect the central nervous system. No clinically significant pharmacokinetic interactions were found in each case. However, melatonin co administration resulted in increased feelings of tranquillity and difficulty in performing tasks compared to imipramine alone, and increased feelings of "muzzy-headedness" compared to thioridazine alone.
Hypnotics.
Melatonin may enhance the sedative properties of benzodiazepines and nonbenzodiazepine hypnotics, such as zaleplon, zolpidem and zopiclone. In a study of jet lag therapy the combination of melatonin and zolpidem resulted in a higher incidence of morning sleepiness, nausea, and confusion, increased impairment of attention, memory and coordination as well as reduced activity during the first hour after getting up, compared to zolpidem alone. The use of melatonin in combination with these drugs is not recommended.
Warfarin.
Melatonin may increase the anticoagulation activity of warfarin. The combination of warfarin or other vitamin K antagonists with melatonin may require dose adjustment of the anticoagulant drugs and should be avoided.
Cigarette smoking.
Cigarette smoking may decrease melatonin levels due to induction of CYP1A2.
Alcohol.
Alcohol is a sedative with the ability to alter physical and mental functions. There is a potential for patients to have enhanced drowsiness when alcohol is co-administered with melatonin.
Other.
There is a large amount of data in the literature regarding the effect of adrenergic agonists/antagonists, opiate agonists/antagonists, antidepressant medicinal products, prostaglandin inhibitors, benzodiazepines, tryptophan and alcohol, on endogenous melatonin secretion. Whether or not these active substances interfere with the dynamic or kinetic effects of melatonin or vice versa has not been studied.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No significant effects on fertility or reproductive performance were observed in rats given oral melatonin prior to mating through to early gestation at doses over 900-fold the recommended clinical dose, based on surface area.
Voquily Jet Lag Relief is not recommended in women and men planning pregnancy.
(Category B3)
No clinical data on exposed pregnancies are available. In view of the lack of clinical data, use in pregnant women and by women intending to become pregnant is not recommended.
Endogenous melatonin has been detected in human breastmilk, thus exogenous melatonin is likely excreted into human milk. The effects of melatonin on the nursing infant have not been established. Therefore, breast-feeding is not recommended in women under treatment with melatonin.4.7 Effects on Ability to Drive and Use Machines
No studies were identified which evaluated the impact of melatonin on the ability to drive or operate machinery. However, as melatonin may cause drowsiness, Voquily Jet Lag Relief is not recommended prior to driving and using machines until the effects are known.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile.
A review of published clinical trials indicated that the most common adverse events in adults are somnolence, headache, dizziness, nausea and stomach cramps.
The adverse reactions reported below are those generally reported for melatonin in published clinical trials and spontaneous case reports.
The adverse reactions are reported according to MedDRA system organ classification and frequencies. Frequencies are defined as: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000), not known (cannot be established from the available data).
Blood and lymphatic system disorders.
Rare: leucopoenia, thrombocytopenia.
Cardiac disorders.
Rare: palpitations, angina pectoris.
Not known: tachycardia.
Ear and labyrinth disorders.
Not known: vertigo.
Eye disorders.
Rare: visual acuity reduced, vision blurred, lacrimation increased.
Gastrointestinal disorders.
Common: nausea.
Uncommon: abdominal pain, upper abdominal pain, dyspepsia, dry mouth, vomiting.
Rare: flatulence, salivary hypersecretion, halitosis, gastro-oesophageal reflux disease, oral ulcers, gastritis.
Not known: diarrhoea, constipation.
General disorders and administration site conditions.
Uncommon: malaise, chest pain, asthenia.
Rare: pain, thirst, fatigue.
Not known: pyrexia.
Immune system disorders.
Not known: hypersensitivity reaction.
Infections and infestations.
Rare: herpes zoster.
Investigations.
Uncommon: weight increased.
Rare: blood electrolytes abnormal, elevated alkaline phosphatase level.
Not known: weight decreased, electrocardiogram QT interval abnormal.
Metabolism and nutrition disorders.
Rare: hypertriglyceridaemia.
Not known: decreased appetite.
Musculoskeletal and connective tissue disorders.
Rare: arthritis, muscle spasms.
Not known: muscular weakness, joint swelling, myalgia, arthralgia.
Nervous system disorders.
Very common: somnolence.
Common: headache, dizziness.
Uncommon: paraesthesia, hyperactivity, lethargy, migraine.
Rare: syncope (fainting), disorientation, memory impairment, restless legs syndrome.
Not known: tremor, light headedness, seizure.
Psychiatric disorders.
Common: irritability.
Uncommon: nervousness, abnormal dreams, nightmares, anxiety, insomnia, restlessness.
Rare: mood altered, aggressive behaviour, libido increased, depression.
Not known: suicide attempt/ideation, libido decreased, hypnotic effects, confusion, visual disturbances.
Renal and urinary disorders.
Uncommon: glycosuria, proteinuria.
Rare: polyuria, haematuria, nocturnal enuresis.
Reproductive system and breast disorders.
Rare: priapism, prostatitis.
Very rare: galactorrhoea.
Respiratory, thoracic and mediastinal disorders.
Not known: cough.
Skin and subcutaneous tissue disorders.
Uncommon: dermatitis, dry skin, rash, pruritus.
Rare: nail disorder.
Very rare: tongue oedema, oedema of the oral mucosa.
Not known: hyperhidrosis, night sweats.
Vascular disorders.
Uncommon: hypertension.
Rare: Hot flushes.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important.
It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Ingestion of daily doses of up to 300 mg of melatonin did not cause clinically significant adverse reactions.
Drowsiness, headache, dizziness, and nausea are the most commonly reported signs and symptoms of high doses with oral melatonin. Flushes, abdominal cramps, diarrhoea, headache, and scotoma lucidum have been reported after ingestion of extremely high melatonin doses (3000 - 6600 mg) for several weeks.
In cases of overdose, general supportive measures should be employed. Gastric lavage and administration of activated charcoal can be considered.
Clearance of the active substance is expected within 12 hours of ingestion.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: melatonin receptor agonists, ATC code: N05CH01.
Melatonin is a hormone and antioxidant. Melatonin secreted by the pineal gland is involved in the synchronisation of circadian rhythms to the diurnal light-dark cycle. Physiologically, melatonin secretion increases shortly after the onset of darkness, peaks at 2-4 and diminishes during the second half of the night. Peak melatonin secretion is almost diametrically opposite peak daylight intensity, with daylight being the primary stimulus for maintaining the circadian rhythmicity of melatonin secretion.
Melatonin has a hypnotic/sedative effect and increases propensity for sleep. Melatonin administered earlier or later than the nocturnal peak in melatonin secretion can, respectively, advance or delay the circadian rhythmicity of melatonin secretion.
Mechanism of action.
The activity of melatonin at the MT1 and MT2 receptors is believed to contribute to its sleep-promoting properties via their distinct actions on the circadian clock. The MT1 receptors are thought to inhibit neuronal firing, while the MT2 receptors have been implicated in the phase-shifting response.
Clinical trials.
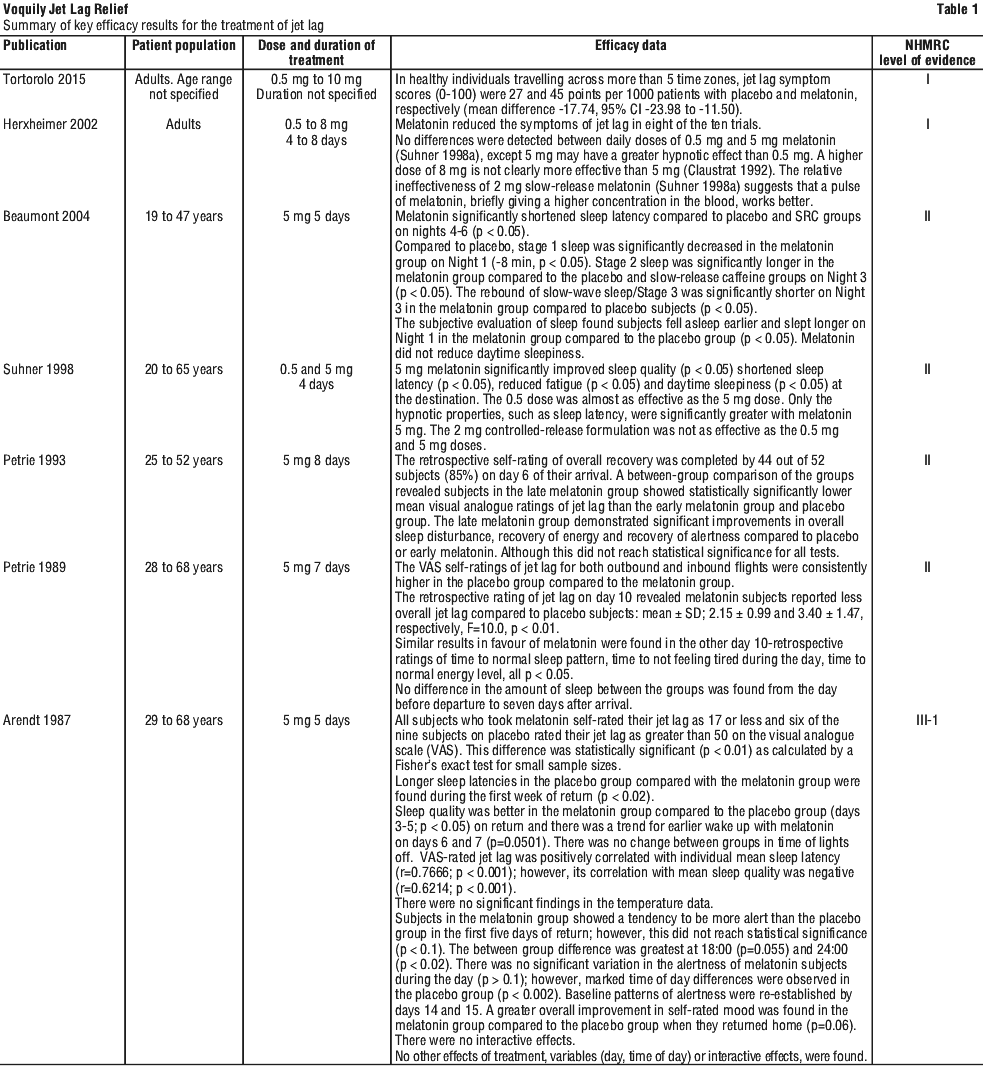
Clinical evidence to support the use of Voquily Jet Lag Relief for the short-term treatment of jet lag in adults aged 18 and over is derived from 19 published reports. Table 1 summarises the patient populations, dosage regimens, efficacy data for the pivotal studies.
5.2 Pharmacokinetic Properties
Melatonin is a small, amphiphilic molecule (molecular weight 232 g/mol) active in its parent form. Melatonin is synthesised in the human body from tryptophan via serotonin. Small quantities are obtained via diet. Data summarised below are from studies that generally involved healthy men and women, primarily young and middle-aged adults.
Absorption.
Orally administered melatonin is almost completely absorbed. Oral bioavailability is ~ 15%, owing to first-pass metabolism of ~ 85%. Plasma Tmax is ~ 50 minutes. A 3 mg dose of immediate-release melatonin raises plasma melatonin Cmax to ~ 3400 picogram/mL, which is ~ 60 times the nocturnal (endogenous) plasma melatonin Cmax, though both endogenous- and exogenous Cmax show considerable inter-individual variation.
Data on the effect of intake of food at or around the time of intake of melatonin on its pharmacokinetics are limited, though suggest that concomitant food intake may increase bioavailability almost 2-fold. Food appears to have a limited effect on Tmax for immediate-release melatonin. This is not expected to affect the efficacy or safety of Voquily Jet Lag Relief.
Plasma melatonin Cmax and AUC increase in a directly proportional, linear manner for oral doses of immediate-release melatonin in the range 1 - 6 mg whereas Tmax and plasma T½ remain constant.
Distribution.
The in vitro plasma protein binding of melatonin is approximately 60%. Melatonin is mainly bound to albumin, alpha-1-acid glycoprotein and high density lipoprotein. The binding to the other serum proteins is insignificant. The melatonin binding was constant over the range of the studied concentrations in serum. Literature data indicates that melatonin is distributed in all body fluids and is accessible at all tissues.
Metabolism.
Melatonin is mainly metabolised by the liver. Experimental data suggest that the cytochrome P450 enzymes CYP1A1 and CYP1A2 are primarily responsible for melatonin metabolism, with CYP2C19 of minor importance. Melatonin is primarily metabolised to 6-hydroxymelatonin (constituting ~ 80 - 90% of melatonin metabolites recovered in the urine). N-acetylserotonin appears to be the primary minor metabolite (constituting ~ 10% of melatonin metabolites recovered in the urine). Melatonin metabolism is very rapid, with plasma 6-hydroxymelatonin level rising within minutes of exogenous melatonin entering the systemic circulation. 6-hydroxymelatonin undergoes sulphate conjugation (~ 70%) and glucuronide conjugation (~ 30%) prior to excretion.
Excretion.
Plasma elimination half-life (T½) is ~ 45 minutes (normal range ~ 30 - 60 minutes) in healthy adults. Dosage once daily in combination with the short half-life means minimal accumulation of melatonin during regular treatment. Melatonin metabolites are mainly eliminated by the urine, ~ 90% as sulphate and glucuronide conjugates of 6-hydroxymelatonin. Less than ~ 1% of a melatonin dose is excreted unchanged in urine.
Gender.
Limited data suggest that Cmax and AUC following ingestion of immediate-release melatonin may be higher (potentially roughly double) in women compared to men, however a large variability in the pharmacokinetics is observed. Plasma melatonin half-life does not appear to be significantly different in men and women.
Elderly.
Data from other formulations of melatonin indicate that the absorption of orally ingested melatonin may be decreased up to 50% in the elderly.
Melatonin metabolism is known to decline with age. Across a range of doses, higher AUC and Cmax levels have been reported in older subjects compared to younger subjects, reflecting the lower metabolism of melatonin in the elderly. Cmax levels around 500 picogram/mL in adults (18-45) versus 1200 picogram/mL in the elderly (55-65); AUC levels around 3,000 picogram.h/mL in adults versus 6000 picogram.h/mL in the elderly.
Hepatic impairment.
The liver is the primary site of melatonin metabolism and therefore, hepatic impairment results in higher endogenous melatonin levels.
Limited data indicates that daytime endogenous blood melatonin concentration is markedly elevated in patients with cirrhosis due to reduced clearance.
Renal impairment.
Decreased renal function is not expected to influence the elimination of melatonin as < 1% of the dose is excreted unchanged in the urine. Melatonin is primarily excreted as metabolites; plasma levels of melatonin metabolites can be expected increase in patients with more advanced renal impairment.
5.3 Preclinical Safety Data
Genotoxicity.
Results from a standard battery of in vitro and in vivo assays showed no evidence of a genotoxic potential for melatonin.
Carcinogenicity.
An oral lifetime carcinogenicity study with melatonin in rats showed an increased incidence of thyroid follicular cell adenomas in males at doses around 700-fold the recommended clinical dose, based on body surface area. No neoplastic tissue histopathology was examined at lower doses and therefore the no-effect dose could not be determined. These effects were associated with liver enzyme induction in this species and are unlikely to be relevant to humans.6 Pharmaceutical Particulars
6.1 List of Excipients
Microcrystalline cellulose, povidone, maltodextrin, magnesium stearate, titanium dioxide, indigo carmine (2 mg) gelatin.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Store in outer carton to protect from light.
6.5 Nature and Contents of Container
PVC/PVDC/Al blister pack with 10 capsules.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
Chemical name: N-[2-(5-methyloxy-1H-indol-3-yl)ethyl]acetamide.
Chemical structure:
 Molecular formula: C13H16N2O2.
Molecular formula: C13H16N2O2.
Molecular weight: 232.27.
CAS number.
73-31-4.7 Medicine Schedule (Poisons Standard)
Schedule 3: Pharmacist Only Medicine.
Summary Table of Changes




 Molecular formula: C13H16N2O2.
Molecular formula: C13H16N2O2.