What is in this leaflet
This leaflet answers some common questions about VOSEVI. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
This leaflet was last updated on the date at the end of this leaflet. More recent information may be available. The latest Consumer Medicine Information is available from your pharmacist or doctor and may contain important information about the medicine and its use of which you should be aware.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking VOSEVI against the benefits it is expected to have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
This medicine has been prescribed for you personally and you should not pass it on to others. It may harm them, even if their symptoms are the same as yours.
What VOSEVI is used for
VOSEVI is used to treat hepatitis C virus (HCV) infection in adults 18 years and older.
Hepatitis C is a virus that infects the liver.
How VOSEVI works
VOSEVI is one tablet that contains three active substances, sofosbuvir, velpatasvir and voxilaprevir. This medicine works by lowering the amount of HCV in your body and may lead to a cure of your HCV infection over a number of weeks.
Cure means the HCV virus is cleared from your blood (remains at an undetectable level) when measured 3 months after finishing all treatment.
VOSEVI does not protect against re-infection with the HCV virus if cure has been achieved.
Ask your doctor if you have any questions about why VOSEVI has been prescribed for you.
Use in children
VOSEVI is for adults. VOSEVI has not been studied in children under the age of 18.
Before you take VOSEVI
When you must not take it
Together with your doctor, you need to decide whether VOSEVI is right for you.
Do not take VOSEVI if you also take any medicines that contain rifampicin.
Do not take VOSEVI if you are allergic to:
- sofosbuvir,
- velpatasvir,
- voxilaprevir, or
- any of the other ingredients of VOSEVI. The ingredients of VOSEVI are listed in the product description section of this leaflet.
Some people are allergic to medicines. If you have any of the following symptoms soon after taking your medicine, DO NOT TAKE ANY MORE VOSEVI and tell your doctor IMMEDIATELY or go to the accident and emergency department at your nearest hospital:
- Skin troubles such as lumpy skin rash or “hives”
- Swelling of the face, lips, mouth, or throat which may cause difficulty in swallowing or breathing
- Wheezing, chest pain, or tightness
- Fainting
These are very serious effects. If you have them, you may have a serious allergic reaction. You may need urgent medical attention or hospitalisation. Hypersensitivity reactions are very rare.
Ask your doctor or pharmacist if you don’t understand anything in this list.
If you are pregnant, think you may be pregnant, or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine. You should not breast feed during treatment with VOSEVI.
Do not take VOSEVI after the expiry or “use by” date (EXP) printed on the bottle. If you take it after the expiry date has passed, it may not work as well.
Do not take VOSEVI if the packaging is torn or shows signs of tampering.
Before you start to take it
Tell your doctor if you are allergic to foods, dyes, preservatives or any other medicines.
Tell your doctor if you have, or have had, any of the following medical conditions:
- Have liver problems, other than hepatitis C.
- Have a current or previous infection with the hepatitis B virus, since your doctor may want to monitor you more closely.
- Have severe kidney problems or if you are on haemodialysis as the effects of VOSEVI on patients with severe kidney problems have not been fully tested.
- Have any other medical condition.
Tell your doctor if you:
- Are pregnant or planning to become pregnant
- Are breastfeeding or plan to breastfeed. It is not known whether sofosbuvir, velpatasvir or voxilaprevir, the three active substances of VOSEVI, pass into human breast milk.
If you have not told your doctor or pharmacist about any of the above, tell them before you start taking VOSEVI.
Taking other medicines
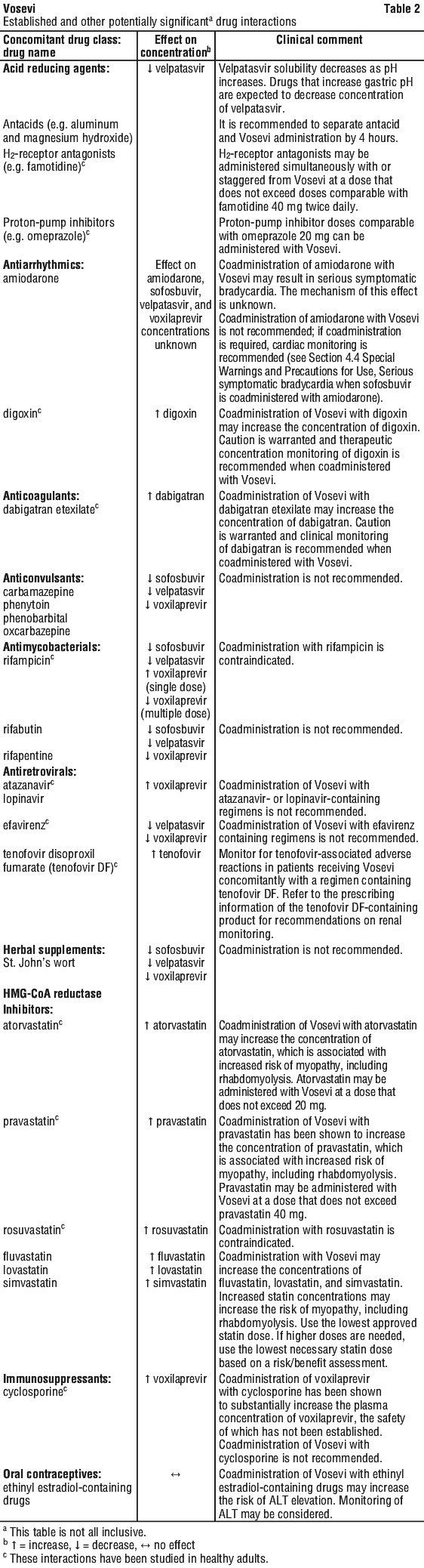
Tell your doctor or pharmacist if you are taking any other medicines including any that you buy without a prescription from your pharmacy, supermarket or health food shop. Some medicines and VOSEVI may interfere with each other.
Tell your doctor if you take any of the following medicines:
- amiodarone used to treat heart conditions
- digoxin used to treat heart conditions
- rosuvastatin, simvastatin, atorvastatin or other statins used to treat high cholesterol
- tenofovir disoproxil fumarate used to treat HIV infection
- atazanavir, efavirenz or lopinavir used to treat HIV infection
- rifampicin, rifapentine, rifabutin (antibiotics used to treat infections, including tuberculosis)
- St. John’s Wort (Hypericum perforatum – herbal medicine used to treat depression)
- carbamazepine, phenytoin, phenobarbital, oxcarbazepine (medicines used to treat epilepsy and prevent seizures)
- dabigatran used to thin the blood
- cyclosporin used to suppress the immune system
- Warfarin or other similar medicines called vitamin K antagonists used to thin the blood.
- ethinyl estradiol, a drug in some oral contraceptives used to prevent pregnancy
VOSEVI may interact with these medicines. As a result, the amounts of VOSEVI or other medicines in your blood may be affected. This may stop your medicines from working properly, or make any side effects worse. In some cases your doctor may need to give you a different medicine or adjust the dose of medicine you are taking.
- Take any medicines used to treat stomach ulcers, heartburn or acid reflux, such as:
- antacids (e.g. aluminium hydroxide or magnesium hydroxide)
- Proton pump inhibitors (e.g. omeprazole)
- H2-antagonists (e.g. famotidine)
These medicines can decrease the amount of velpatasvir in your blood. If you are taking one of these medicines your doctor will either give you a different medicine for stomach ulcers, heartburn, or acid reflux, or recommend how and when you take that medicine.
- If you are taking an antacid, take it at least 4 hours before or at least 4 hours after VOSEVI.
- If you are taking a proton pump inhibitor, your doctor may give you a different medicine or adjust the dose of medicine you are taking.
- If you are taking an H2-antagonist, your doctor may give you a different medicine or adjust the dose of the medicine you are taking.
It is very important to let your doctor or pharmacist know what medications, herbal supplements, or vitamins you are taking.
Know the medicines you take. Keep a list of medicines and show it to your doctor and pharmacist when you get a new medicine. Your doctor and your pharmacist can tell you if you can take these medicines with VOSEVI. Do not start any new medicines while you are taking VOSEVI without first talking with your doctor or pharmacist.
Your doctor and pharmacist may have more information on medicines to be careful with or to avoid while taking VOSEVI.
How to take VOSEVI
Follow all directions given to you by your doctor or pharmacist carefully.
How much to take
The usual dose is one VOSEVI tablet orally, once daily.
Never change the dose on your own. Do not stop this medicine unless your doctor tells you to stop.
How to take it
VOSEVI tablets must be taken with food.
When to take it
Take VOSEVI at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
If you forget to take it
Do not miss a dose of VOSEVI. If you forget to take VOSEVI, take your missed dose right away unless it is almost time for your next dose.
Do not take a double dose to make up for a forgotten dose. Continue with your regular dosing schedule.
Do not stop taking VOSEVI unless your doctor tells you to. It is very important that you complete the full course of treatment to give the medicines the best chance to cure your HCV infection.
Do not change your dose or stop taking VOSEVI without first talking to your doctor.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to use your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or Poisons Information Centre: 13 11 26 (Australia) and 0800 764 766 (New Zealand) or go to the Accident and Emergency department at your nearest hospital if you think you or anyone else may have taken too many VOSEVI tablets. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are taking VOSEVI
Things you must do
Tell all doctors and pharmacists who are treating you that you are taking VOSEVI.
Tell your doctor as soon as possible if there is any worsening of your condition.
Things you must not do
Do not give VOSEVI to anyone else, even if they have the same condition as you.
Do not stop taking VOSEVI or change the dose without checking with your doctor. It is important not to stop taking your VOSEVI tablets, unless advised to do so by your doctor.
Do not breastfeed. See “Before you take it”.
Things to be careful of
Be careful driving or operating machinery until you know how VOSEVI affects you.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking VOSEVI.
VOSEVI helps most people with HCV infection, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects. Do not be alarmed by these lists of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- headache
- fatigue
- diarrhoea
- nausea
Check with your doctor as soon as possible if you have any problems while taking VOSEVI, even if you do not think the problems are connected with the medicine or are not listed in this leaflet.
Tell your doctor immediately if you or your family notice any of the following side effects:
- signs of allergy such as rash or hives on the skin; swelling of the face, lips, tongue or other parts of the body; wheezing or difficulty breathing
Tell your doctor if you notice anything else that is making you feel unwell. Other side effects not listed above may also occur in some people.
After taking VOSEVI
Storage
Keep VOSEVI tablets where children cannot reach them. A locked cupboard at least one-and-a half metres above the ground is a good place to store them.
Keep VOSEVI tablets in a cool, dry place where it stays below 30 °C.
Do not store VOSEVI or any other medicine in a bathroom or near a sink.
Do not leave VOSEVI in the car or on a window sill – heat and dampness can destroy some medicines.
Keep your VOSEVI tablets in the bottle with the cap tightly closed until you take them. If you take VOSEVI tablets out of their pack they may not keep well.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
VOSEVI tablets are capsule shaped and beige in colour. Each tablet has “GSI” on one side and  on the other side of the tablet.
on the other side of the tablet.
VOSEVI tablets are supplied in bottles containing 28 tablets.
Ingredients
Active Ingredients: sofosbuvir, velpatasvir and voxilaprevir
Inactive Ingredients: silicon dioxide, copovidone, croscarmellose sodium, lactose monohydrate, magnesium stearate, and microcrystalline cellulose.
Film-coating: ferrosoferric oxide, iron oxide red, iron oxide yellow, polyethylene glycol, polyvinyl alcohol, purified talc, and titanium dioxide.
Sponsor
Australia
Gilead Sciences Pty Ltd
Level 6, 417 St Kilda Road
Melbourne, Victoria 3004
New Zealand
c/- Grant Thornton New Zealand
Limited, L4, 152 Fanshawe Street
Auckland 1010
This leaflet was prepared on 08 March 2018.VOSEVI and GSI are trademarks of Gilead Sciences, Inc. or one of its related companies. Other brands listed are trademarks of their respective owners and are not trademarks of Gilead Sciences, Inc.
AUST R 286358
Published by MIMS June 2018



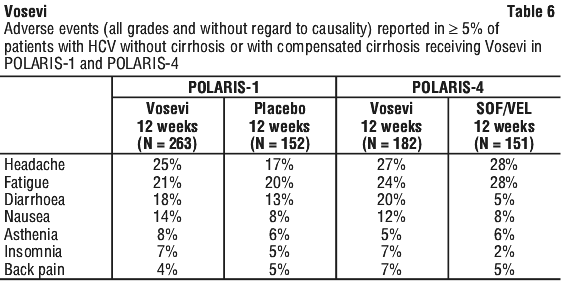 In POLARIS-1, of the patients receiving Vosevi who experienced these adverse events, > 99% of patients had an adverse event of mild or moderate (Grade 1 or 2) severity. Each of these adverse events occurred at a similar frequency or more frequently than in patients treated with placebo.
In POLARIS-1, of the patients receiving Vosevi who experienced these adverse events, > 99% of patients had an adverse event of mild or moderate (Grade 1 or 2) severity. Each of these adverse events occurred at a similar frequency or more frequently than in patients treated with placebo.
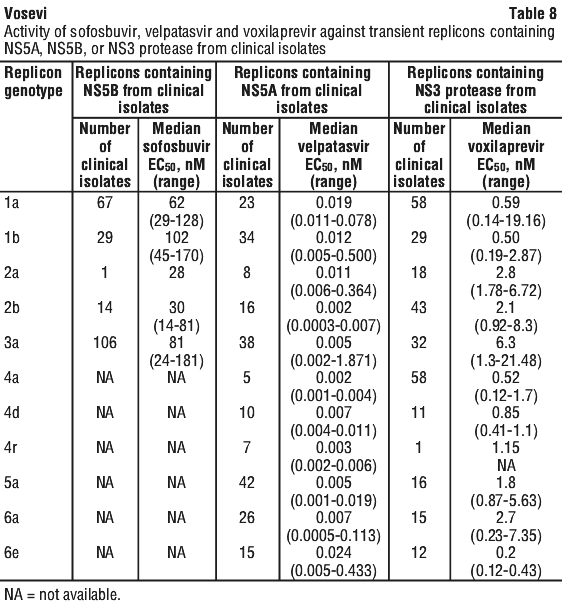 The presence of 40% human serum had no effect on the anti-HCV activity of sofosbuvir, but reduced the anti-HCV activity of velpatasvir and voxilaprevir by 13- and 6.8-fold respectively, against genotype 1a HCV replicons.
The presence of 40% human serum had no effect on the anti-HCV activity of sofosbuvir, but reduced the anti-HCV activity of velpatasvir and voxilaprevir by 13- and 6.8-fold respectively, against genotype 1a HCV replicons. SVR12 was achieved in 18 of 19 (95%) patients who had baseline NS5B NI RAVs in POLARIS-1, including 2 patients that had virus with the S282T NS5B NI RAV in addition to NS5A RAVs at baseline. In POLARIS-4, a total of 14 patients had virus with NS5B NI RAVs at baseline and all achieved SVR12.
SVR12 was achieved in 18 of 19 (95%) patients who had baseline NS5B NI RAVs in POLARIS-1, including 2 patients that had virus with the S282T NS5B NI RAV in addition to NS5A RAVs at baseline. In POLARIS-4, a total of 14 patients had virus with NS5B NI RAVs at baseline and all achieved SVR12. SVR12 was achieved in all 39 patients who had baseline NS5B NI RAVs in POLARIS-2 and 2 of 3 (67%) patients in POLARIS-3. The NS5B NI RAV S282T was not detected in any patient in POLARIS-2 and POLARIS-3 trials.
SVR12 was achieved in all 39 patients who had baseline NS5B NI RAVs in POLARIS-2 and 2 of 3 (67%) patients in POLARIS-3. The NS5B NI RAV S282T was not detected in any patient in POLARIS-2 and POLARIS-3 trials.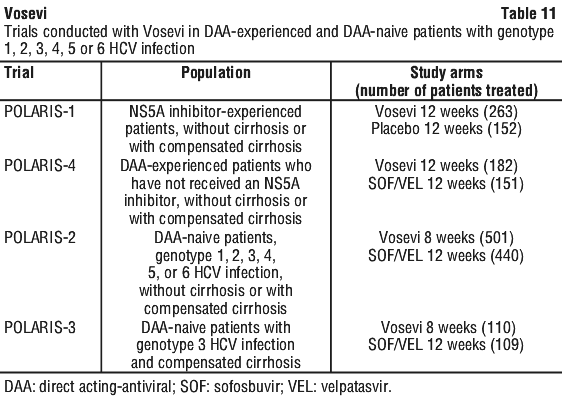 Serum HCV RNA values were measured during the clinical trials using the COBAS AmpliPrep/COBAS Taqman HCV test (version 2.0) with a lower limit of quantification (LLOQ) of 15 IU per mL. Sustained virologic response (SVR12), defined as HCV RNA less than LLOQ at 12 weeks after the cessation of treatment, was the primary endpoint to determine the HCV cure rate.
Serum HCV RNA values were measured during the clinical trials using the COBAS AmpliPrep/COBAS Taqman HCV test (version 2.0) with a lower limit of quantification (LLOQ) of 15 IU per mL. Sustained virologic response (SVR12), defined as HCV RNA less than LLOQ at 12 weeks after the cessation of treatment, was the primary endpoint to determine the HCV cure rate.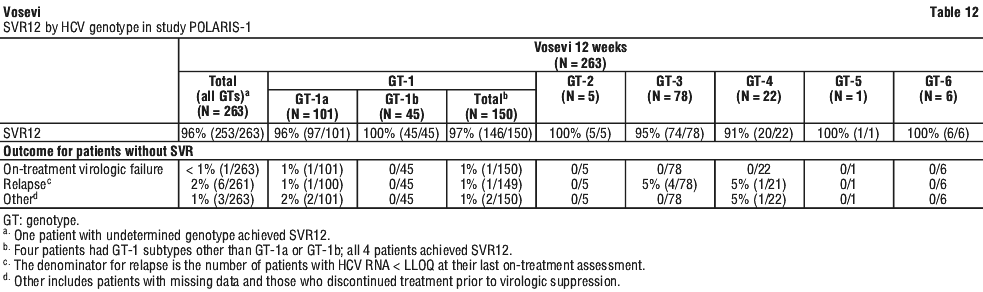 Treatment with Vosevi for 12 weeks in POLARIS-1 was statistically superior relative to the prespecified performance goal of 85% (p < 0.001).
Treatment with Vosevi for 12 weeks in POLARIS-1 was statistically superior relative to the prespecified performance goal of 85% (p < 0.001).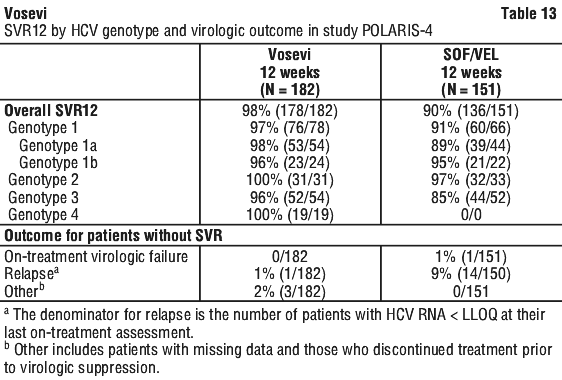 Treatment with Vosevi for 12 weeks resulted in numerically higher SVR12 rates than treatment with sofosbuvir/velpatasvir for 12 weeks in patients with HCV genotype 1a and 3 infection. Comparable SVR12 rates were observed in patients with HCV genotype 1b and 2 infection treated with Vosevi for 12 weeks or with sofosbuvir/velpatasvir for 12 weeks. No comparison data are available for HCV genotypes 4, 5, and 6. Given these data, the additional benefit of Vosevi has not been shown over sofosbuvir/velpatasvir in adults with genotype 1b, 2, 4, 5 or 6 infection previously treated with sofosbuvir without an NS5A inhibitor and Vosevi is only indicated for the treatment of HCV genotypes 1a or 3 infection in adults who previously received sofosbuvir without an NS5A inhibitor.
Treatment with Vosevi for 12 weeks resulted in numerically higher SVR12 rates than treatment with sofosbuvir/velpatasvir for 12 weeks in patients with HCV genotype 1a and 3 infection. Comparable SVR12 rates were observed in patients with HCV genotype 1b and 2 infection treated with Vosevi for 12 weeks or with sofosbuvir/velpatasvir for 12 weeks. No comparison data are available for HCV genotypes 4, 5, and 6. Given these data, the additional benefit of Vosevi has not been shown over sofosbuvir/velpatasvir in adults with genotype 1b, 2, 4, 5 or 6 infection previously treated with sofosbuvir without an NS5A inhibitor and Vosevi is only indicated for the treatment of HCV genotypes 1a or 3 infection in adults who previously received sofosbuvir without an NS5A inhibitor.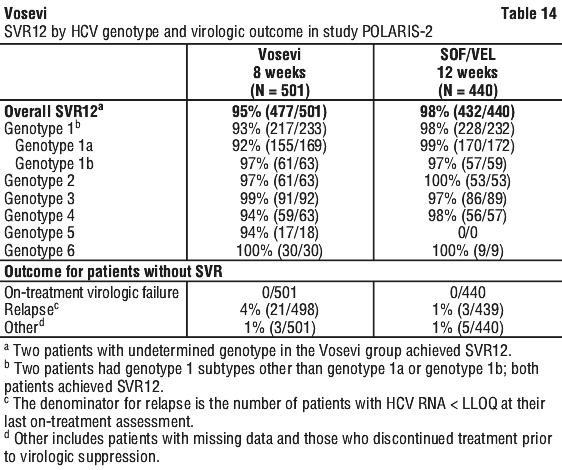 Treatment with Vosevi for 8 weeks in POLARIS-2 did not demonstrate noninferiority to treatment with SOF/VEL for 12 weeks with a prespecified margin of -5%.
Treatment with Vosevi for 8 weeks in POLARIS-2 did not demonstrate noninferiority to treatment with SOF/VEL for 12 weeks with a prespecified margin of -5%. The SVR12 rate for each treatment group was statistically superior relative to the prespecified SVR12 performance goal of 83% (p < 0.001 for both groups).
The SVR12 rate for each treatment group was statistically superior relative to the prespecified SVR12 performance goal of 83% (p < 0.001 for both groups). The chemical name of velpatasvir is Methyl {(1R)-2-[(2S,4S)- 2-(5-{2-[(2S,5S)- 1-{(2S)-2-[(methoxycarbonyl) amino]-3-methylbutanoyl} -5-methylpyrrolidin-2-yl] -1,11-dihydro[2] benzopyrano[4',3':6,7] naphtho[1,2-d]imidazol -9-yl}-1H-imidazol-2-yl)-4- (methoxymethyl)pyrrolidin- 1-yl]-2-oxo-1-phenylethyl} carbamate. It has a molecular formula of C49H54N8O8 and a molecular weight of 883.0. It has the following structural formula:
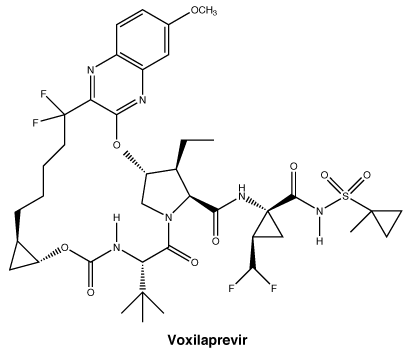
The chemical name of velpatasvir is Methyl {(1R)-2-[(2S,4S)- 2-(5-{2-[(2S,5S)- 1-{(2S)-2-[(methoxycarbonyl) amino]-3-methylbutanoyl} -5-methylpyrrolidin-2-yl] -1,11-dihydro[2] benzopyrano[4',3':6,7] naphtho[1,2-d]imidazol -9-yl}-1H-imidazol-2-yl)-4- (methoxymethyl)pyrrolidin- 1-yl]-2-oxo-1-phenylethyl} carbamate. It has a molecular formula of C49H54N8O8 and a molecular weight of 883.0. It has the following structural formula: The chemical name for voxilaprevir is (1aR,5S,8S,9S,10R,22aR) -5-tert-butyl-N-{(1R,2R)- 2-(difluoromethyl)-1- [(1-methylcyclopropanesulfonyl) carbamoyl] cyclopropyl}-9-ethyl-18,18-difluoro- 14-methoxy-3,6-dioxo- 1,1a,3,4,5,6,9,10, 18,19,20,21,22,22a-tetradecahydro -8H-7,10-methanocyclopropa [18,19][1,10,3,6] dioxadiazacyclononadecino [11,12-b] quinoxaline-8-carboxamide. It has a molecular formula of C40H52F4N6O9S and a molecular weight of 868.9. It has the following structural formula:
The chemical name for voxilaprevir is (1aR,5S,8S,9S,10R,22aR) -5-tert-butyl-N-{(1R,2R)- 2-(difluoromethyl)-1- [(1-methylcyclopropanesulfonyl) carbamoyl] cyclopropyl}-9-ethyl-18,18-difluoro- 14-methoxy-3,6-dioxo- 1,1a,3,4,5,6,9,10, 18,19,20,21,22,22a-tetradecahydro -8H-7,10-methanocyclopropa [18,19][1,10,3,6] dioxadiazacyclononadecino [11,12-b] quinoxaline-8-carboxamide. It has a molecular formula of C40H52F4N6O9S and a molecular weight of 868.9. It has the following structural formula: