1 Name of Medicine
Daunorubicin and cytarabine.
2 Qualitative and Quantitative Composition
Each vial contains 44 mg of daunorubicin (as hydrochloride) and 100 mg of cytarabine.
After reconstitution, the solution contains 2.2 mg/1 mL of daunorubicin and 5.0 mg/1 mL of cytarabine encapsulated in liposomes in a fixed combination in a 1:5 molar ratio.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Vyxeos powder for injection is a purple lyophilised cake.
4.1 Therapeutic Indications
Vyxeos is indicated for the treatment of adults with newly diagnosed, therapy-related acute myeloid leukaemia (t-AML) or AML with myelodysplasia-related changes (AML-MRC).
4.2 Dose and Method of Administration
Vyxeos must be initiated and monitored under the supervision of physicians experienced in the use of chemotherapeutic medicinal products.
Patients may be pre-medicated for nausea and vomiting. An anti-hyperuricaemic therapy should be considered (e.g. allopurinol) prior to initiating Vyxeos.
Vyxeos has a different dosage than daunorubicin injection and cytarabine injection and it must not be interchanged with other daunorubicin and/or cytarabine-containing products (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
A baseline cardiac evaluation with an electrocardiogram (ECG) and a multi-gated radionuclide angiography (MUGA) scan or an echocardiography (ECHO) is recommended, especially in patients with risk factors for increased cardiac toxicity. Cardiac function should be closely monitored.
Dosage.
Vyxeos dosing is based on the patient's body surface area (BSA) according to the following schedule. See Table 1.
 Recommended dosing schedule for induction of remission. The recommended dosing schedule of Vyxeos 44 mg/100 mg/m2, administered intravenously over 90 minutes:
Recommended dosing schedule for induction of remission. The recommended dosing schedule of Vyxeos 44 mg/100 mg/m2, administered intravenously over 90 minutes:
on days 1, 3, and 5 as the first course of induction therapy;
on days 1 and 3 as subsequent course of induction therapy, if needed.
A subsequent course of induction may be administered in patients who do not show disease progression or unacceptable toxicity. The attainment of a normal-appearing bone marrow may require more than one induction course. Evaluation of the bone marrow following recovery from the previous course of induction therapy determines whether a further course of induction is required. Treatment should be continued as long as the patient continues to benefit or until disease progression up to maximum of 2 induction courses.
Recommended dosing schedule for consolidation. The first consolidation cycle should be administered 5 to 8 weeks after the start of the last induction.
The recommended dosing schedule of Vyxeos is 29 mg/65 mg/m2, administered intravenously over 90 minutes:
on days 1 and 3 as subsequent courses of consolidation therapy, if needed.
Consolidation therapy is recommended for patients achieving remission who have recovered to absolute neutrophil count (ANC) > 500/microL and the platelet count has recovered to greater than 50,000/microL in the absence of unacceptable toxicity. A subsequent course of consolidation may be administered in patients who do not show disease progression or unacceptable toxicity within the range of 5 to 8 weeks after the start of the first consolidation. Treatment should be continued as long as the patient continues to benefit or until disease progression, up to maximum of 2 consolidation courses.
Elderly patients.
No dose adjustment is required in elderly patients (≥ 65 years) (see Section 5.2 Pharmacokinetic Properties).
Dose adjustment.
Dose adjustment following undesirable effects/interactions. Patients should be monitored for haematologic response and toxicities.
Dosing should be delayed or permanently discontinued, if necessary, as described below.
Hypersensitivity.
For mild hypersensitivity symptoms (e.g. mild flushing, rash, pruritus), the treatment should be stopped, and the patient should be supervised, including monitoring of vital signs. The treatment should be restarted slowly once the symptoms have resolved, by halving the rate of infusion and intravenous antihistamines and intravenous dexamethasone should be given.
For moderate hypersensitivity symptoms (e.g. moderate rash, flushing, mild dyspnoea, chest discomfort) the treatment should be stopped. Intravenous antihistamines and intravenous dexamethasone should be given. The infusion should not be restarted. When the patient is retreated, Vyxeos should be given at the same dose and rate and with premedication.
For severe/life-threatening hypersensitivity symptoms (e.g. hypotension requiring vasopressor therapy, angioedema, respiratory distress requiring bronchodilation therapy, generalised urticaria), the treatment should be stopped. Intravenous antihistamines and dexamethasone should be given, and an epinephrine (adrenaline) or bronchodilators should be added if indicated. Do not reinitiate infusion, and do not retreat. Treatment with Vyxeos should be permanently discontinued. Patients should be monitored until symptoms resolve.
Missed dose.
If a planned dose of Vyxeos is missed, the dose should be administered as soon as possible and the dosing schedule adjusted accordingly, maintaining the treatment interval.
Cardiotoxicity.
Vyxeos treatment should be discontinued in patients who develop signs or symptoms of cardiomyopathy unless the benefits outweigh the risks (see Section 4.4 Special Warnings and Precautions for Use).
Special dosage instructions. Patients with impaired hepatic function.
Dose adjustment is not required for patients with a bilirubin level less than or equal to 50 micromol/L. There is no experience with Vyxeos in patients with hepatic impairment resulting in a bilirubin level greater than 50 micromol/L. Vyxeos should only be used in patients with severe hepatic impairment if the benefits outweigh the risks (see Section 4.4 Special Warnings and Precautions for Use).
Patients with impaired renal function.
Dose adjustment is not required for patients with mild (creatinine clearance [CrCL] 60 mL/min to 89 mL/min by Cockcroft Gault equation [C-G]), moderate (CrCL 30 mL/min to 59 mL/min) or severe (CrCL < 30 mL/min) renal impairment. There is no experience with Vyxeos in patients with end-stage renal disease managed with dialysis. (See Section 5.2 Pharmacokinetic Properties.)
Method of administration.
Vyxeos is for intravenous use only. It must not be administered via an intramuscular, intrathecal, or subcutaneous route.
Vyxeos is administered by intravenous infusion over a period of 90 minutes. Care should be taken to ensure there is no extravasation to prevent the risk of tissue necrosis (see Section 4.4 Special Warnings and Precautions for Use).
Instructions for handling.
Vyxeos is a cytotoxic medicinal product. Applicable special handling and disposal procedures should be followed. Product is for single use in one patient only. Discard any residue.
Preparation instructions.
Determine the dose and number of vials of Vyxeos based on the individual patient's BSA as outlined in the Dosage section.
Remove the appropriate number of vials of Vyxeos from the refrigerator and equilibrate to the room temperature (15°C to 30°C) for 30 minutes.
Then, reconstitute each vial with 19 mL of sterile water for injections using a 20 mL syringe, and immediately thereafter start a 5-minute timer.
Carefully swirl the contents of the vial for 5 minutes while gently inverting the vial every 30 seconds.
Do not heat, vortex, or shake vigorously.
After reconstitution, let it rest for 15 minutes.
The reconstituted product should be an opaque, purple, homogeneous dispersion, essentially free from visual particulates.
If the reconstituted product is not diluted into an infusion bag immediately, store in a refrigerator (2°C to 8°C) for up to 4 hours.
Following the storage of reconstituted product in the vial for up to 4 hours at 2°C to 8°C, the reconstituted product must immediately be diluted into an infusion solution and run for the 90-minute infusion time.
Reconstituted product in the vial and reconstituted product which has been diluted into an infusion solution are stable for 4 hours when stored at 2°C to 8°C.
The 4 hour stability period for the reconstituted product in the vial does not allow for an additional 4 hour stability period after the appropriate dose from the reconstituted vial is diluted into the infusion solution.
The 4 hour stability period when reconstituted product diluted in the infusion bag is stored at 2°C to 8°C does not include the time required for reconstitution or the 90-minute infusion time.
The diluted infusion solution must be immediately run for the 90-minute infusion time following the up to 4 hour stability period.
Calculate the volume of reconstituted Vyxeos required using the following formula: [volume required (mL) = dose of daunorubicin (mg/m2) x patient's BSA (m2)/2.2 (mg/mL)].
The concentration of the reconstituted solution is 44 mg/20 mL (2.2 mg/mL) daunorubicin and 100 mg/20 mL (5 mg/mL) cytarabine.
Gently invert each vial 5 times prior to withdrawing the concentrate for dilution.
Aseptically withdraw the calculated volume of reconstituted Vyxeos from the vial(s) with a sterile syringe and transfer it to an infusion bag containing 500 mL of sodium chloride 9 mg/mL (0.9%) solution for injection, or 5% glucose. There may be residual product remaining in the vial. Discard unused portion.
Gently invert the bag to mix the solution. The dilution of the reconstituted product results in a deep purple, translucent, homogeneous dispersion.
If the diluted infusion solution is not used immediately, store in a refrigerator (2°C to 8°C) for up to 4 hours.
Gently invert the bag to mix the solution after refrigeration.
Administration instructions.
Do not mix Vyxeos with, or administer as an infusion with, other medicinal products.
Administer Vyxeos by constant intravenous infusion over 90 minutes via an infusion pump through a central venous catheter or a peripherally inserted central catheter. An in-line membrane filter may be used for the intravenous infusion of Vyxeos, provided the minimum pore diameter of the filter is greater than or equal to 15 micrometre.
Flush the line after administration with sodium chloride 9 mg/mL (0.9%) solution for injection.4.3 Contraindications
Hypersensitivity to the active substances or to any of the excipients.
4.4 Special Warnings and Precautions for Use
Other daunorubicin and/or cytarabine-containing products.
Vyxeos must not be substituted or interchanged with other daunorubicin and/or cytarabine containing products. Due to substantial differences in the pharmacokinetic parameters, the dose and schedule recommendations for Vyxeos are different from those for daunorubicin hydrochloride injection, cytarabine injection, daunorubicin citrate liposome injection, and cytarabine liposome injection. The medicinal product name and dose should be verified prior to administration to avoid dosing errors.
Severe myelosuppression.
Severe myelosuppression (including fatal infections and haemorrhagic events) has been reported in patients after administration of a therapeutic dose of Vyxeos. Serious or fatal haemorrhagic events, including fatal central nervous system (CNS) haemorrhages, associated with severe thrombocytopenia, have occurred in patients treated with Vyxeos. Baseline assessment of blood counts should be obtained, and patients should be carefully monitored during treatment with Vyxeos for possible clinical complications due to myelosuppression. Due to the long plasma half-life of Vyxeos, time to recovery of ANC and platelets may be prolonged and require additional monitoring.
Prophylactic anti-infectives (including anti-bacterial, anti-virals, anti-fungals) may be administered during the period of profound neutropenia until ANC returns to 500/microL or greater. If myelosuppressive complications occur, appropriate supportive measures should be used, e.g. anti-infectives, colony-stimulating factors, transfusions. Blood counts should be regularly monitored until recovery (see Section 4.2 Dose and Method of Administration, Dose adjustments).
Cardiotoxicity.
Cardiotoxicity is a known risk of anthracycline treatment. Prior therapy with anthracyclines (including patients who have previously received the recommended maximum cumulative doses of doxorubicin or daunorubicin hydrochloride), pre-existing cardiac disease (including impaired cardiac function), previous radiotherapy of the mediastinum, or concomitant use of cardiotoxic products may increase the risk of daunorubicin-induced cardiac toxicity.
Total cumulative doses of non-liposomal daunorubicin greater than 550 mg/m2 have been associated with an increased incidence of treatment-induced congestive heart failure. This limit appears lower (400 mg/m2) in patients who received radiation therapy to the mediastinum. The relationship between cumulative Vyxeos dose and the risk of cardiac toxicity has not been determined. Total cumulative exposure of daunorubicin has been described in Table 2.
 A baseline cardiac evaluation with an electrocardiogram (ECG) and a multi-gated radionuclide angiography (MUGA) scan or an echocardiography (ECHO) is recommended, especially in patients with risk factors for increased cardiac toxicity. Cardiac function should be closely monitored.
A baseline cardiac evaluation with an electrocardiogram (ECG) and a multi-gated radionuclide angiography (MUGA) scan or an echocardiography (ECHO) is recommended, especially in patients with risk factors for increased cardiac toxicity. Cardiac function should be closely monitored.
Treatment with Vyxeos should be discontinued in patients with impaired cardiac function unless the benefit of initiating or continuing treatment outweighs the risk (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Pregnancy warning/women of childbearing potential.
Patients should be advised to avoid becoming pregnant while receiving Vyxeos. Male patients and women of childbearing potential must use an effective method of contraception during treatment and for 6 months following the last dose of Vyxeos (see Section 4.6 Fertility, Pregnancy and Lactation).
Hypersensitivity reactions.
Serious hypersensitivity reactions, including anaphylactic reactions, have been reported with daunorubicin and cytarabine.
See Section 4.2 Dose and Method of Administration for details on dosage adjustment in patients experiencing hypersensitivity symptoms.
Tissue necrosis.
Daunorubicin has been associated with local tissue necrosis at the site of medicinal product extravasation. In clinical studies with Vyxeos, one event of extravasation occurred, but no necrosis was observed. Care should be taken to ensure that there is no extravasation of medicinal product when Vyxeos is administered. Vyxeos should be administered intravenously only. Do not administer via an intramuscular, intrathecal, or subcutaneous route (see Section 4.2 Dose and Method of Administration).
Use in renal or hepatic impairment.
Hepatic impairment may increase the risk of toxicity associated with daunorubicin and cytarabine. Evaluation of hepatic function using conventional clinical laboratory tests is recommended prior to administration of Vyxeos and periodically during treatment. There is no experience with Vyxeos in patients with baseline serum bilirubin greater than 50 micromol/L or endstage renal disease managed with dialysis. Vyxeos should only be used in patients with severe hepatic impairment if the benefits outweigh the risks (see Section 4.2 Dose and Method of Administration).
Effects on laboratory tests.
Vyxeos may induce hyperuricemia secondary to rapid lysis of leukaemic cells. Blood uric acid levels should be monitored and appropriate therapy initiated in the event that hyperuricemia develops. An anti-hyperuricaemic therapy should be considered (e.g. allopurinol) prior to initiating Vyxeos.
History of Wilson's disease or other copper-related disorder.
Each vial contains 100 mg of copper gluconate, which corresponds to 14 mg of elemental copper. Vyxeos should only be used in patients with a history of Wilson's disease or other copper-related disorder if the benefits outweigh the risks. Discontinue Vyxeos in patients with signs or symptoms of acute copper toxicity.
Immunosuppressant effects/increased susceptibility to infections.
Administration of live or live-attenuated vaccines in patients that are immunocompromised by chemotherapeutic agents may result in serious or fatal infections. Vaccination with a live vaccine should be avoided in patients receiving Vyxeos. Killed or inactivated vaccines may be administered; however, the response to such vaccines may be diminished.
Gastrointestinal mucositis and diarrhoea.
It should be taken into consideration that the absorption of oral accompanying medicinal products may be considerably influenced by gastrointestinal mucositis and/or diarrhoea frequently occurring in association with intensive chemotherapy.
Use in the elderly.
The pharmacokinetics of Vyxeos in elderly patients aged > 85 years has not been evaluated (see Section 5.2 Pharmacokinetic Properties).
Paediatric use.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
No interaction studies have been performed with Vyxeos. The delivery of daunorubicin and cytarabine in the Vyxeos formulation is anticipated to reduce the possibility of interactions, because systemic free-drug concentrations of daunorubicin and cytarabine are much lower than when administered as the non-liposomal formulation.
Cardiotoxic agents.
Concurrent use of cardiotoxic agents may increase the risk of cardiotoxicity. Use of Vyxeos in patients who have previously received doxorubicin increases the risk of cardiotoxicity (see Section 4.4 Special Warnings and Precautions for Use). Do not administer Vyxeos in combination with other cardiotoxic agents unless the patient's cardiac function is closely monitored.
Hepatotoxic agents.
Hepatotoxic medicinal products may impair liver function and increase the toxicity. Since daunorubicin is metabolised by the liver, changes in hepatic function induced by concomitant therapies may affect the metabolism, pharmacokinetics, therapeutic efficacy, and/or the toxicity of Vyxeos (see Section 5.2 Pharmacokinetic Properties). Hepatic function should be monitored more frequently when Vyxeos is co-administered with hepatotoxic agents.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
Based on findings in animals, male fertility may be compromised by treatment with Vyxeos. Although no fertility studies have been conducted with Vyxeos, in previous studies cytarabine increased sperm-head abnormalities and chromosomal aberrations following intraperitoneal administration in mice, and daunorubicin caused testicular atrophy and total aplasia of spermatocytes in the seminiferous tubules in dogs.
(Category D)
There are no data on the use of Vyxeos in pregnant women.
Reproductive studies have not been conducted with Vyxeos. In previous studies, both cytarabine and daunorubicin, tested separately, showed teratogenic and embryotoxic effects in animal studies. Based on results from previous animal studies and its mechanism of action, Vyxeos should not be used during pregnancy, unless the clinical condition of the woman requires treatment and justifies the potential risk to the fetus.
If the medicinal product is used during pregnancy, or if the patient becomes pregnant while receiving Vyxeos, the woman should be informed of the potential hazard to the fetus. In any case, cardiologic examination and a blood count are recommended in fetuses and newborns born to mothers who received treatment during pregnancy.
Women of childbearing potential/contraception in males and females.
Women of childbearing potential should avoid becoming pregnant while receiving Vyxeos. Women of childbearing potential should use effective contraception while they or their male partner undergo treatment. Women of childbearing potential should not receive treatment until pregnancy is excluded.
Women of childbearing potential should undergo pregnancy testing before initiation of Vyxeos. Men with sexual partners of reproductive potential and women should use effective contraception during treatment and for 6 months following the last dose of Vyxeos.
It is not known whether Vyxeos is excreted in human milk. Because of the potential for serious adverse reactions in breast feeding children from Vyxeos, mothers should be advised not to breastfeed during Vyxeos therapy.4.7 Effects on Ability to Drive and Use Machines
Vyxeos has minor influence on the ability to drive and use machines. Fatigue and dizziness have been reported with the use of Vyxeos. Therefore, caution is recommended when driving or operating machines.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile.
A total of 629 subjects were treated in the studies, Study 101, Study 204, Study 205, Study 206 and Study 301; of these, 403 subjects received at least 1 dose of Vyxeos. The Pooled Safety population included all subjects in Study 101, Study 204, Study 205, Study 206 and Study 301 who received at least 1 dose of 44 mg/m2 daunorubicin and 100 mg/m2 cytarabine or 44 mg/m2 daunorubicin and 101 mg/m2 Vyxeos (n = 375). See Table 3.

Tabulated list of adverse reactions.
The most frequently occurring adverse reactions (ADRs), which were observed during the treatment with Vyxeos, were hypersensitivity including rash (66.9%), febrile neutropenia (63.5%), oedema (52.3%), diarrhoea/colitis (49.9%), mucositis (49.9%), fatigue (46.4%), musculoskeletal pain (44.5%), abdominal pain (36.3%), decreased appetite (33.9%), cough (33.9%), headache (32.3%), chills (31.2%), arrhythmia (30.4%), pyrexia (29.6%), sleep disorders (25.1%), and hypotension (23.7%).
The most serious and frequently occurring ADRs were infection (58.7%), cardiotoxicity (18.7%) and haemorrhage (13.1%).
Adverse drug reactions have been included under the appropriate category in Table 4 according to the highest frequency observed in any of the main clinical studies. Frequencies are defined as: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1000 to < 1/100); rare (≥ 1/10,000 to < 1/1000); not known (cannot be estimated from the available data).
Within each frequency grouping, adverse reactions are presented in order of decreasing seriousness. For classification of adverse reactions which occur at Grades 3-5 (G3-5), a comprehensive listing is available from the National Cancer Institute (NCI) at NCI Common Terminology Criteria for Adverse Events (CTCAE). Toxicity is graded as mild (Grade 1), moderate (Grade 2), severe (Grade 3), or life-threatening (Grade 4), with specific parameters according to the organ system involved. Death (Grade 5) is used for some of the criteria to denote a fatality.

Description of selected undesirable effects.
Infections.
Due to the neutropenia experienced with Vyxeos, infections of various types were very common ADRs. Pneumonia, sepsis and bacteraemia were the most frequently seen serious infection ADRs in the clinical studies population. The incidence of infection events was 78.1%; the incidence of non-serious events of infections was 73.1%, the incidence of serious events of infections was 28.5%; the incidence of infections which led to discontinuation is 0.5%. The incidence of fatal infections was 6.9%. The fatal infections experienced were sepsis and pneumonia (see Section 4.4 Special Warnings and Precautions for Use). The incidence of bacteraemia in patients treated with Vyxeos was 10.7% in comparison to 4.2% of all controls.
Haemorrhage.
Due to the thrombocytopenia experienced with Vyxeos a variety of haemorrhagic events were seen in clinical studies. The most common haemorrhagic event was epistaxis, and the majority of these were considered not serious (29.1%). The incidence of haemorrhage events is 69.1%; the incidence of non-serious events of haemorrhage was 67.2%; the incidence of serious events of haemorrhage is 5.6%; the incidence of haemorrhage which led to discontinuation is 0. The incidence of fatal haemorrhage was 2.1%. Serious or fatal haemorrhagic events, including fatal central nervous system (CNS) haemorrhages, associated with severe thrombocytopenia were seen in patients treated with Vyxeos (see Section 4.4 Special Warnings and Precautions for Use).
Hypersensitivity.
Hypersensitivity reactions were very common ADRs in Vyxeos liposomal clinical studies. The most frequently reported hypersensitivity ADRs were rash and the majority of these were not serious (38.9%). The incidence of all hypersensitivity events was 66.9%; the incidence of non-serious events of hypersensitivity was 66.4%, of which 38.9% were rash; the incidence of serious events of hypersensitivity is 1.1%; the frequency of hypersensitivity which led to discontinuation is 0. The frequency of fatal hypersensitivity events was 0 (see Section 4.4 Special Warnings and Precautions for Use).
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
There is no specific experience in the management of overdose in patients. If overdose occurs, exacerbation of adverse reactions associated with Vyxeos are expected and supportive treatment (including anti-infectives, blood and platelet transfusions, colony-stimulating factors, and intensive care as needed) should be provided until the patient recovers. Observe the patient carefully over time for signs of cardiotoxicity and provide appropriate supportive therapy as clinically indicated.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: combinations of antineoplastic agents, ATC Code - L01XY01.
Mechanism of action.
Vyxeos is a liposomal formulation of a fixed combination of daunorubicin and cytarabine in a 1:5 molar ratio. The 1:5 molar ratio has been shown in vitro and in vivo to maximise synergistic antitumour activity in AML.
Daunorubicin has antimitotic and cytotoxic activity, which is achieved by forming complexes with DNA, inhibiting topoisomerase II activity, inhibiting DNA polymerase activity, affecting regulation of gene expression, and producing DNA-damaging free radicals.
Cytarabine is a cell cycle phase-specific antineoplastic agent, affecting cells only during the S-phase of cell division. Intracellularly, cytarabine is converted into cytarabine-5-triphosphate (ara-CTP), which is the active metabolite. The mechanism of action is not completely understood, but it appears that ara-CTP acts primarily through inhibition of DNA synthesis. Incorporation into DNA and RNA may also contribute to cytarabine cytotoxicity. Cytarabine is cytotoxic to proliferating mammalian cells in culture.
Based on data in animals, Vyxeos liposomes accumulate and persist at a higher concentration in the bone marrow, where they are preferentially taken up intact by leukaemia cells. In leukaemia-bearing mice, the liposomes are taken up by leukaemia cells to a greater extent than by normal bone marrow cells. After internalisation, Vyxeos liposomes undergo degradation, releasing daunorubicin and cytarabine within the intracellular environment.
Cardiac electrophysiology.
The effect of Vyxeos on cardiac repolarization following the first induction cycle as determined by the Fridericia's corrected QT-interval (QTcF) was evaluated in an open-label, single arm study in 26 patients who received Vyxeos on days 1, 3 and 5. No patients had QTcF changes from baseline were more than 60 msecs, and no QTcF values were greater than 500 msecs, indicating absence of risk for prolongation of the QT interval.
Clinical trials.
The efficacy of Vyxeos in adults for the treatment of newly diagnosed AML was evaluated in one controlled study (Study 301).
Study 301 in patients with untreated high risk AML. Study 301 was a Phase 3 randomised, multicentre, open-label, parallel-arm, superiority study which evaluated Vyxeos vs. a standard combination of cytarabine and daunorubicin (7 + 3) in 309 patients between 60 to 75 years of age with untreated high risk AML. Patients with the following AML sub types were included in the study: therapy-related AML (t-AML), myelodysplastic syndrome AML (MDS AML) and chronic myelomonocytic leukaemia AML (CMMoL AML) with documented history of MDS or CMMoL prior to transformation to AML, and de novo AML with karyotype changes characteristic of myelodysplasia, (per 2008 WHO criteria).
The study included 2 phases, 1) Treatment Phase during which patients received up to 2 induction and 2 consolidation courses, and 2) a Follow-up Phase, which began 30 days after the last induction or consolidation course and continued for up to 5 years from randomisation. The number of inductions and consolidations a patient received depended upon Complete Response (CR) or Complete Response with incomplete recovery (CRi), which was confirmed by bone marrow assessment. In clinical studies only, Vyxeos 100 units/m2/day (equivalent to 44 mg/100 mg/m2) was administered intravenously over 90 minutes on days 1, 3, and 5 for the first induction and on days 1 and 3 for patients requiring a second induction. A second induction was highly recommended for patients who did not achieve a CR or CRi in the first induction course and was mandatory for patients achieving greater than 50% reduction in percent blasts. Post-remission therapy with haematopoietic stem cell transplantation (HSCT) was permitted either in place of or after consolidation chemotherapy. For consolidation courses, in clinical studies only, the Vyxeos dose was reduced to 65 units/m2/day (equivalent to 29 mg/65 mg/m2) on days 1 and 3. In the 7 + 3 arm, first induction consisted of cytarabine 100 mg/m2/day on days 1 to 7 by continuous infusion, and daunorubicin 60 mg/m2/day on days 1, 2, and 3 whereas second induction and consolidation cytarabine was dosed on days 1 to 5 and daunorubicin on days 1 and 2.
There were 153 patients randomised to Vyxeos and 156 patients randomised to the 7 + 3 control arm. The randomised patients had a median age of 68 (range 60 - 75 years), 61% were male, and 88% had an ECOG performance status of 0 - 1. At baseline, 20% had t-AML, 54% had AML with an antecedent haematological disorder, and 25% had de novo AML with myelodysplasia-related cytogenetic abnormalities; 34% had been treated previously with a hypomethylating agent for MDS; 54% had an adverse karyotype.
The demographic and baseline disease characteristics were generally balanced between the study arms. FLT3 mutation was identified in 15% (43/279) of patients tested and NPM1 mutation was identified in 9% (25/283) patients tested.
The primary endpoint was overall survival measured from the date of randomisation to death from any cause. Vyxeos demonstrated superiority in overall survival in the ITT population compared with the comparator 7 + 3 treatment regimen (Figure 1). The median survival for the Vyxeos treatment group was 9.56 months compared with 5.95 months for the 7 + 3 treatment group (Hazard Ratio = 0.69, 95% CI = 0.52, 0.90, two-sided log-rank test p = 0.005).
The overall rate of HSCT was 34% (52/153) in the Vyxeos arm and 25% (39/156) on the control arm. See Table 5.


60 month follow-up.
The 60 month overall survival rate was higher for the Vyxeos treatment arm (18%) versus the 7+3 treatment arm (8%); the hazard ratio was 0.70 (95% CI: [0.55, 0.91]).
5.2 Pharmacokinetic Properties
Vyxeos liposomes exhibit a prolonged plasma half-life following intravenous infusion in preclinical studies, with greater than 99% of the daunorubicin and cytarabine in the plasma remaining encapsulated within the liposomes. Vyxeos delivered a synergistic combination of daunorubicin and cytarabine to leukaemia cells for a prolonged period of time based on in vitro and in vivo studies.
The pharmacokinetics of daunorubicin and cytarabine administered as Vyxeos were investigated in patients who received a dose of daunorubicin 44 mg/m2 and cytarabine 100 mg/m2 administered as a 90-minute intravenous infusion on days 1, 3, and 5. The pharmacokinetics of each medicinal product was based on total plasma concentrations (i.e. encapsulated plus unencapsulated medicinal product). Following the dose administered on day 5, the mean (% coefficient of variation [CV]) maximum plasma concentrations (Cmax) for daunorubicin was 26.0 microgram/mL (32.7%) and cytarabine was 62.2 microgram/mL (33.7%). The mean (% CV) area under the curve (AUC) during one dosing interval for daunorubicin was 637 microgram.h/mL (38.4%) and cytarabine was 1900 microgram.h/mL (44.3%).
When daunorubicin and cytarabine are administered as components of Vyxeos, the liposomes appear to govern their tissue distribution and rates of elimination; therefore, while the non-liposomal medicinal products have markedly different clearance (CL), volume of distribution (V), and terminal half-life (t1/2) Vyxeos causes these pharmacokinetic parameters to converge.
The accumulation ratio was 1.3 for daunorubicin and 1.4 for cytarabine. There was no evidence of time-dependent kinetics or major departures from dose proportionality over the range of 1.3 mg/3 mg per m2 to 59 mg/134 mg per m2 (0.03 to 1.3 times the approved recommended dosage).
Distribution.
The volume of distribution (% CV) for daunorubicin is 6.6 L (36.8%) and cytarabine is 7.1 L (49.2%). Plasma protein binding was not evaluated.
Metabolism.
Similar to non-liposomal daunorubicin and cytarabine, subsequent to release from Vyxeos liposomes, both daunorubicin and cytarabine are extensively metabolised in the body. Daunorubicin is mostly catalysed by hepatic and non-hepatic aldo-keto reductase and carbonyl reductase to the active metabolite daunorubicinol. Cytarabine is metabolised by cytidine deaminase to the inactive metabolite 1-β (beta)-D-arabinofuranosyluracil (AraU). Unlike non-liposomal daunorubicin and cytarabine, which are quickly metabolised to the respective metabolites, daunorubicin and cytarabine after Vyxeos administration are free bases encapsulated in liposomes. Plasma concentration-time profiles obtained from 13 to 26 patients who received Vyxeos 100 units/m2 (equivalent to 44 mg/m2 of daunorubicin and 100 mg/m2 of cytarabine) on days 1, 3, and 5 show the mean AUClast metabolite:parent ratio for daunorubicinol and AraU were 1.79% and 3.22% to that for daunorubicin and cytarabine, respectively, which are lower than those typically reported for non liposomal products, ~40-60% for daunorubicinol:daunorubicin and ~80% for AraU:cytarabine. The lower percentages of metabolite:parent ratios after Vyxeos administration indicate that most of the total daunorubicin and cytarabine in the circulation is trapped inside the Vyxeos liposomes, where they are inaccessible to medicinal product-metabolising enzymes.
Excretion.
Vyxeos exhibits a prolonged half-life (% CV) of 31.5 h (28.5%) for daunorubicin and 40.4 h (24.2%) for cytarabine with greater than 99% of the daunorubicin and cytarabine in the plasma remaining encapsulated within the liposomes. The clearance (% CV) is 0.16 L/h (53.3%) for daunorubicin and 0.13 L/h (60.2%) for cytarabine.
Urinary excretion of daunorubicin and daunorubicinol accounts for 9% of the administered dose of daunorubicin, and urinary excretion of cytarabine and AraU accounts for 71% of the administered dose of cytarabine.
Special populations.
No clinically meaningful effects on the pharmacokinetics of daunorubicin and cytarabine were observed based on age (1 to 81 years), sex, race, body weight, body mass index, and white blood cell count after adjusting dose by body surface area.
Elderly patients.
The pharmacokinetics of Vyxeos in patients aged > 85 years has not yet been evaluated. No data are available.
Renal impairment.
Based on a dedicated study to evaluate the impact of moderate to severe renal impairment on the pharmacokinetics of Vyxeos and a population pharmacokinetic analysis using data from clinical studies in patients with mild to moderate renal impairment, no significant difference in clearance of daunorubicin or cytarabine was observed in patients with pre-existing mild, moderate or severe renal impairment compared to patients with baseline normal renal function. The potential effects of end-stage renal disease managed with dialysis on the pharmacokinetics of daunorubicin and cytarabine administered as Vyxeos are unknown (see Section 4.2 Dose and Method of Administration).
Hepatic impairment.
The pharmacokinetics of total daunorubicin and cytarabine were not altered in patients with bilirubin ≤ 50 micromol/L. The pharmacokinetics in patients with bilirubin greater than 50 micromol/L is unknown.
5.3 Preclinical Safety Data
Genotoxicity.
Mutagenicity studies have not been conducted with Vyxeos.
Previous studies have demonstrated that cytarabine was mutagenic and clastogenic in vitro (chromosome aberrations and sister-chromatid exchanges (SCE) in human leukocytes), and in vivo (chromosome aberrations and SCE in rodent). Cytarabine also caused the transformation of hamster embryo cells and rat H43 cells in vitro and was clastogenic to meiotic cells. Previous studies have also demonstrated daunorubicin was mutagenic (bacterial assay, V79 hamster cell assay) and clastogenic (CCRF-CEM human lymphoblasts) in vitro, and clastogenic in vivo (SCE assay in mouse bone marrow).
Carcinogenicity.
Carcinogenicity studies have not been conducted with Vyxeos.
Published data with daunorubicin suggest possible tumorigenicity in rats after a single intravenous dose of 5 mg/kg (approximately 0.7 times the MRHD on an mg/m2 basis). A high incidence of mammary tumours was observed about 120 days after a single intravenous dose of 12.5 mg/kg daunorubicin in rats (about 1.7 times the recommended human dose on an mg/m2 basis). A carcinogenic evaluation of daunorubicin by the IARC Working Group classified daunorubicin in Group 2B - Drugs which are possibly carcinogenic to humans based on sufficient evidence in animals and inadequate data in humans. Studies with cytarabine were not identified. Published data with Ara-C, the active metabolite of cytarabine, did not provide evidence of carcinogenicity.6 Pharmaceutical Particulars
6.1 List of Excipients
Distearoylphosphatidylcholine, distearoylphosphatidylglycerol, cholesterol, copper gluconate, trolamine (for pH adjustment), sucrose.
6.2 Incompatibilities
This medicinal product must not be mixed with other medicinal products except those mentioned in Section 4.2 Dose and Method of Administration, Method of administration.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
Stability of the reconstituted suspension in the vial and diluted infusion solution.
The reconstituted suspension in the vial does not contain a preservative.
The maximum combined storage time for reconstituted product in the vial and reconstituted product diluted into an infusion bag is up to a total of 4 hours at 2°C to 8°C.
6.4 Special Precautions for Storage
Store in the refrigerator (2 - 8°C).
Keep the container in the outer carton to protect the contents from light. Store in an upright position.
For specific storage conditions after reconstitution/dilution of the drug product, see Section 6.3 Shelf Life.
Keep out of the reach of children.
6.5 Nature and Contents of Container
50 mL vial (type 1 glass) with a stopper (chlorobutyl rubber), and an aluminium overseal.
Each pack contains either 1 vial or 2 vials. Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
All materials used for dilution and administration should be disposed of according to local procedures applicable to the discarding of antineoplastic agents. Any unused medicinal product or waste material should be disposed of in accordance with local requirements for cytotoxic agents.
6.7 Physicochemical Properties
Cytarabine.
The chemical name is 4-amino-1-β-D-arabinofuranosyl-1H-pyrimidine-2-one.
Cytarabine is white or almost white, crystalline powder. It is freely soluble in water, very slightly soluble in alcohol and methylene chloride.
Chemical structure.
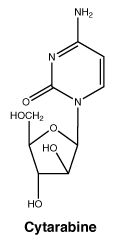 Molecular formula: C9H13N3O5.
Molecular formula: C9H13N3O5.
Molecular weight: 243.2.
CAS number.
147-94-4.
Daunorubicin.
The chemical name is (8S,10S)-8-Acetyl-10-[(3-amino-2,3,6-trideoxy-α-L-lyxo-hexopyranosyl)oxy]-6,8,11-trihydroxy-1-methoxy-7,8,9,10-tetrahydrotetracene-5,12-dione hydrochloride.
Daunorubicin hydrochloride is an orange-red powder. It is freely soluble in water and in methanol, slightly soluble in alcohol, practically insoluble in acetone.
Chemical structure.
 Molecular formula: C27H29NO10.HCl.
Molecular formula: C27H29NO10.HCl.
Molecular weight: 564.
CAS number.
23541-50-6.7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes



 A baseline cardiac evaluation with an electrocardiogram (ECG) and a multi-gated radionuclide angiography (MUGA) scan or an echocardiography (ECHO) is recommended, especially in patients with risk factors for increased cardiac toxicity. Cardiac function should be closely monitored.
A baseline cardiac evaluation with an electrocardiogram (ECG) and a multi-gated radionuclide angiography (MUGA) scan or an echocardiography (ECHO) is recommended, especially in patients with risk factors for increased cardiac toxicity. Cardiac function should be closely monitored.



 Molecular formula: C9H13N3O5.
Molecular formula: C9H13N3O5. Molecular formula: C27H29NO10.HCl.
Molecular formula: C27H29NO10.HCl.