What is in this leaflet
This leaflet answers some common questions about Wafesil.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.All medicines have risks and benefits. Your doctor has weighed the risks of you taking Wafesil against the benefits they expect it will have for you.
It should be used only under strict medical supervision.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Wafesil is used for
Wafesil is used to treat erectile dysfunction, more commonly known as impotence, in men. This is when a man cannot get, or keep, a hard erect penis suitable for sexual activity.
Wafesil belongs to a group of medicines called phosphodiesterase type 5 inhibitors.
It works by relaxing the blood vessels in your penis when you are sexually excited. This allows blood to flow into your penis, allowing you to get an erection in the natural way.
Wafesil will work only if you are sexually excited.
Wafesil will not increase your sex drive.
Wafesil is not for use in women.
This medicine is available only with a doctor's prescription.
Before you take Wafesil
When you must not take it
YOU MUST NOT TAKE Wafesil IF YOU ARE TAKING NITRATES OR NITRITE MEDICATIONS. IT MAY LEAD TO A SEVERE DROP IN YOUR BLOOD PRESSURE, WHICH MAY BE DIFFICULT TO TREAT.
BECAUSE SEXUAL ACTIVITY MAY PLACE A STRAIN ON YOUR HEART, YOUR DOCTOR WILL NEED TO CHECK WHETHER YOU ARE FIT ENOUGH TO TAKE Wafesil.
Do not take Wafesil if you are being treated for angina (chest pain) or other heart conditions with medicines called nitrates. Nitrate medicines include glyceryl trinitrate (also called nitroglycerin). Common trade names for glyceryl trinitrate tablets include Anginine and Lycinate.
Common trade names for glyceryl trinitrate patches include Nitro-Dur, Transiderm-Nitro, Nitroderm TTS, and Minitran.
Common trade names for glyceryl trinitrate sprays include Nitrolingual and Glytrin.
Trade names for glyceryl trinitrate injections include Glyceryl Trinitrate Concentrate and Glyceryl Trinitrate. Common trade names for other nitrate preparations include Imdur Durules, Monodur Durules, Sorbidin, Isordil, Imtrate, Duride, Isomonit, Ikorel and Sodium Nitroprusside.
There may be other trade names not listed here.
Do not take Wafesil if you are taking guanylate cyclase stimulators (GCS), such as Adempas (riociguat). GCS is a type of medicine used to treat high blood pressure in the blood vessels in the lungs caused by blood clots in the lungs (chronic thromboembolic pulmonary hypertension, CTEPH) or narrowing of the vessels that carry blood from the heart to the lungs (pulmonary arterial hypertension or PAH).
Do not take Wafesil if you:
- have heart or blood vessel problems that make sexual intercourse inadvisable
- have suffered a heart attack or stroke in the last 6 months
- have severe liver problems
- have blood pressure that is unusually high or low or is not effectively treated
- have loss of vision in one or both eyes from an eye disease called non-arteritic anterior ischaemic optic neuropathy (NAION)
- have an eye disease called retinitis pigmentosa.
Do not take Wafesil if you have an allergy to sildenafil or similar medicines or any of the ingredients listed at the end of this leaflet
An allergic reaction can include:
- hives, itching or skin rash
- swelling of the face, lips or tongue which may lead to difficulty swallowing or breathing.
Do not take Wafesil if the packaging is torn or shows signs of tampering, or does not look quite right even if the wafers may look alright.
Do not take Wafesil if the expiry date on the pack has passed. If it has expired or is damaged, return it to your pharmacist for disposal.
Before you start to take it
Tell your doctor if you:
- have any allergies to any other medicines or any other substances such as foods, preservatives or dyes.
- have any other heart or blood vessel problems.
- have previously had sudden loss of eyesight in one or both eyes.
- have any of the following medical conditions:
- diabetes, especially if you also have eye problems
- kidney or liver problems
- leukaemia (cancer of the blood cells)
- multiple myeloma (a cancer of the bone marrow)
- any disease or deformity of your penis
- any bleeding disorder such as haemophilia
- stomach ulcer
- a disease of the blood called sickle cell anaemia
- colour vision problems
- previously experienced sudden decrease or loss of hearing. - are taking or using any other treatment for impotence
- are taking any medicines to treat high blood pressure in the vessels of the lungs (pulmonary arterial hypertension) including Tracleer (bosentan) or Revatio which also contains sildenafil.
Tell your doctor if you have any other medical conditions.
If you have not told your doctor or pharmacist about any of the above, tell him/her before you start taking Wafesil.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including medicines you buy without a prescription from a pharmacy, supermarket or health food shop.
Do not take Wafesil if you are taking or using nitrate medicines for angina.
Do not take Wafesil if you are taking guanylate cyclase stimulators (GCS), such as Adempas (riociguat).
Some medicines and Wafesil may interfere with each other. These include:
- cimetidine, a medicine used to treat ulcers
- some medicines used to treat fungal infections including ketoconazole and itraconazole
- some antibiotics including erythromycin and rifampicin
- some protease inhibitors such as ritonavir and saquinavir for the treatment of HIV infection
- medicines called alpha-blockers. These are used to treat high blood pressure or prostate problems
- Tracleer (bosentan), a medicine used to treat high blood pressure in the vessels of the lungs.
You may need different amounts of your medicine or you may need totake different medicines. Your doctor or pharmacist can tell you what to do if you are taking any of these medicines. They also have a more complete list of medicines to be careful with or avoid while taking Wafesil.
Ask your doctor or pharmacist, if you are not sure if you are taking any of these medicines.
How to take Wafesil
Take Wafesil exactly as your doctor has prescribed.
Follow all directions given to you by your doctor and pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
Your doctor will decide the correct dose for you depending on your condition and response.
This can be one 25 mg wafer a day, one 50 mg wafer a day or up to a maximum of two 50 mg wafers a day.
Do not take more than one dose of Wafesil a day.
How to take it
Wafesil is designed to be taken by placing under the tongue until it is dissolved. It should not be broken, sucked, chewed or swallowed.
The wafer should not be removed from the blister until you are ready to take it.
When you are ready to take the wafer, your mouth should be moist; rinse your mouth with a little water prior to taking it.
Separate one blister pocket from the strip. Carefully peel off the backing and gently tap or push the wafer out. Do not push the wafer through the blister foil as this may crush it.
Use dry hands when handling the wafer.
If you are taking one wafer, place the wafer under the tongue as far back as possible and on one side. Allow it to dissolve. The wafer should dissolve within 2-3 minutes.
If you are taking 2 wafers, place one wafer on one side under your tongue first. Wait 1 to 2 minutes and place the second wafer on the opposite side under the tongue. Keep the wafers under the tongue and allow to dissolve completely.
Do not consume any food or drink until the wafer has dissolved.
When to take it
Take your dose of Wafesil about one hour before you intend to have sex.
The amount of time Wafesil takes to start working varies from person to person, but it normally takes between half an hour and one hour.
You may find Wafesil takes longer to work if you take it with a heavy meal.
Wafesil will work only if you are sexually excited.
If you take too much (Overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone 13 11 26) for advice or go to Accident and Emergency at the nearest hospital if you think that you or anyone else may have taken too much Wafesil.
Do this even if there are no signs of discomfort or poisoning.
While you are taking it
Things you must do
Stop taking Wafesil if you have a loss of eyesight in one or both eyes, experience loss of hearing or have an erection that persists more than 4 hours. Seek medical attention urgently.
If Wafesil does not help you get an erection or if your erection does not last long enough to complete sexual intercourse, tell your doctor. In these cases, your doctor may decide that you need a higher dose.
If you are about to start taking any new medicines, especially nitrates, or Adempas (riociguat), tell your doctor and pharmacist that you are taking Wafesil.
See "Before you take Wafesil" for a list of common nitrate medications.
Tell any other doctors, dentists and pharmacists who are treating you that you are taking Wafesil.
Things you must not do
Do not use drugs containing amyl nitrite (sometimes called "poppers") while you are taking Wafesil.
If you get an angina attack do not take nitrate medicines to relieve the pain but tell your doctor immediately. Make sure your doctor knows you are taking Wafesil.
Do not give Wafesil to anyone else, even if they have the same condition as you.
Things to be careful of
Be careful drinking alcohol while taking Wafesil. Drinking alcohol can temporarily impair the ability to get an erection.
Do not drink large amounts of alcohol before sexual activity.
If you experience changes in vision, or dizziness, when taking Wafesil, you should not drive or operate machinery.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Wafesil.
While Wafesil helps most people, it can cause some unwanted side effects in a few people. All medicines have side effects. If unwanted effects occur, most are likely to be minor and temporary. However, some may be serious and need medical attention.
Do not be alarmed by this list of possible side effects.
You may not get any of them.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- headache
- dizziness
- flushing
- hot flushes
- indigestion
- heart burn
- nasal congestion
- sinus congestion
- swelling of your nose
- diarrhoea
- rash
- dry mouth or dry throat
- dry nose
- dry eye
- tightness in your throat
- feeling hot or irritable
- redness in your mouth or tongue.
Tell your doctor as soon as possible if you notice any of the following:
- unusual heart beat
- urinary tract infection (stinging or burning urine, more frequent need to pass urine)
- blood in the urine
- persistent headache or fainting
- bleeding from the nose
- pain or tingling in your hands, toes or feet
- decreased sensitivity or numbness in your mouth
- irritation or feeling of having something in the eye
- swollen or puffy eye(s)
- fatigue, pain in or around the eyes
- "red eye" due to swollen blood vessels in the white part of the eye and in the eyelids.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- signs of allergy such as shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts
- chest pain
- increased heart rate
- sudden decrease or loss of hearing
- seizures, fits or convulsions
- your erection is increased, painful or persists for longer than usual. If your erection continues for four hours, or sooner if there is pain, you should seek medical attention urgently
- rarely men have lost eyesight sometime after taking drugs to treat erectile dysfunction (known as impotence). If you lose eyesight in one or both eyes or experience changes in vision such as blurring, a blue colour to your vision or a greater awareness of light, seek medical attention urgently
- changes to your normal vision such as:
- red or yellow colour tinges to your vision or colourless objects appear coloured
- you see a halo around lights, sparks or lights when your eyes are closed.
This is not a complete list of all possible side effects. Others may occur in some people and there may be side effects not yet known. If you notice any other symptoms that worry you, check with your doctor.
Ask your doctor or pharmacist if you don't understand anything in this list.
After taking Wafesil
Storage
Keep this medicine where children cannot reach it. A locked cupboard at least one-and- a-half metres above the ground is a good place to store medicines.
Keep Wafesil in a cool; dry place where the temperature stays below 25°C. Do not store it or any other medicine in the bathroom or near a sink.
Do not leave it in the car or on windowsills. Heat and dampness can destroy some medicines; Wafesil wafers should be protected from light and moisture
Keep the wafers in their blisters until it is time to take them. If you take your wafers out of the pack they will not keep as well and may be damaged or crushed.
Disposal
If your doctor tells you to stop taking Wafesil, or the wafers have passed their expiry date, ask yourpharmacist what to do with any left over.
Product description
What it looks like
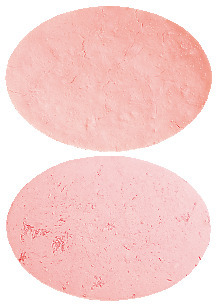
- 25 mg – A light pink to pink oval disc
- 50 mg – A light yellow to yellow oval disc
All strengths are available in cartons of 4, 8 or 12 wafers.
Ingredients
Wafesil wafers contain sildenafil (as citrate) equivalent to 25 mg or 50 mg sildenafil.
Other ingredients
- microcrystalline cellulose
- Carmellose sodium
- Mannitol
- Lactose monohydrate
- Potato starch
- Glycine
- Macrogol 1500
- Citric acid
- Peppermint flavour
- Allura Red AC (25 mg only)
- Quinoline Yellow (50 mg only)
Supplier
Wafesil is supplied by:
iX Biopharma Pty Ltd
ABN 55 136 680 601
110 Merrindale Dve
Croydon VIC 3136 Australia.
Phone: +61 3 9737 4333
Email: [email protected]
Australian Registration Numbers
25 mg - AUST R 288496
50 mg - AUST R 288497.
This leaflet was prepared in June 2018
Published by MIMS October 2018


 Other adverse reactions occurred at a rate of > 2%, but equally commonly on placebo: respiratory tract infection, back pain, flu syndrome and arthralgia.
Other adverse reactions occurred at a rate of > 2%, but equally commonly on placebo: respiratory tract infection, back pain, flu syndrome and arthralgia.
