1 Name of Medicine
Clascoterone.
2 Qualitative and Quantitative Composition
Each gram of Winlevi cream contains 10 mg of clascoterone.
For the full list of excipients, see Section 6.1.
3 Pharmaceutical Form
White to almost white cream.
4.1 Therapeutic Indications
Winlevi (clascoterone) cream is indicated for the topical treatment of acne vulgaris in patients 12 years of age and older.
4.2 Dose and Method of Administration
Recommended dose and administration.
Cleanse the entire area to be treated and dry gently. After the skin is dry, apply a thin uniform layer of Winlevi twice per day, in the morning and in the evening, over the entire area prone to acne. It is advised to use Winlevi as instructed by the prescribing healthcare professional. For optimal effectiveness, do not spot treat.
Hands should be washed before and after applying Winlevi cream.
Missed dose.
If patients forget to take a dose of Winlevi, they should be instructed to apply the next dose at the usual time. Patients should be instructed to not apply a double dose to make up for forgotten doses.
Dosing considerations.
Winlevi is for external use only. Winlevi is not for ophthalmic, oral or vaginal use.
Avoid accidental transfer of Winlevi into eyes, lips, mouth, corners of the nose, or other mucous membranes. If contact with mucous membranes occurs, rinse immediately and thoroughly with water.
This medication should not be applied to cuts, abrasions, eczematous, or sunburned skin.4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed, see Section 6.1.
4.4 Special Warnings and Precautions for Use
General.
Winlevi is for external use only. Not for ophthalmic, oral or vaginal use.
Winlevi should not be applied to cuts, abrasions, eczematous or sunburned skin.
Avoid accidental transfer of Winlevi into eyes, lips, mouth, corners of the nose, or other mucous membranes. If contact with mucous membranes occurs, rinse immediately and thoroughly with water.
Hypothalamic-pituitary-adrenal (HPA) axis suppression.
Clascoterone is structurally similar to non-androgen corticosteroids and may exert its efficacy by means other than the AR.
The main metabolite of clascoterone, cortexolone, is an intermediate in the synthesis of glucocorticoids and exhibits weak glucocorticoid properties.
Reversible physiologic hypothalamic-pituitary-adrenal (HPA) axis suppression was observed in two maximal use clinical studies with exposure conditions up to 3 times the recommended Winlevi daily dose for up to 2 weeks. Reversible HPA axis suppression may occur during or after treatment with clascoterone. In both studies, all patients who demonstrated an abnormal HPA axis response returned to normal HPA axis response at follow-up visit, 4 weeks after stopping treatment (see Section 4.4 Special Warnings and Precautions for Use, Paediatric use; Section 5.1 Pharmacodynamic Properties). Conditions which augment systemic absorption include use over large surface areas, prolonged use, and the use of occlusive dressings.
If HPA axis suppression develops, an attempt should be made to withdraw the drug.
Local skin reactions.
Winlevi may induce local irritation (edema, erythema/redness, pruritus, scaling/dryness, skin atrophy, stinging/burning, striae rubrea, telangiectasia). Concomitant use with other potentially irritating topical products (medicated or abrasive soaps and cleansers, soaps and cosmetics that have a strong drying effect and products with high concentrations of alcohol, astringents, spices or lime) should be limited.
Use in the elderly.
There are no data available for patients aged older than 65 years as none were included in the clinical programme.
Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Paediatric use.
There are insufficient data to recommend the use of Winlevi in children under the age of 12 years. Under maximal use conditions, HPA-axis suppression was seen more frequently in children less than 12 years old (See Section 4.4, Hypothalamic-pituitary-adrenal (HPA) axis suppression).
The safety and efficacy of Winlevi for the topical treatment of acne vulgaris have been established in 641 paediatric patients, aged 12 to 18 years in two identical multicentre, randomised, double-blind, vehicle-controlled, 12-week trials and 2 open-label pharmacokinetic studies (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
In two, maximum use clinical studies, with Winlevi administered at up to 3 times the recommended daily dose, for up to 2 weeks, in 20 adult patients and 22 adolescent patients, HPA axis suppression (as demonstrated by 30 minute post-stimulation serum cortisol level ≤ 497 nmol/L) was observed in 1/20 (5%) adult patients and 2/22 (9%) adolescent patients. These were generally minimal reductions below normal levels.
All subjects returned to normal HPA axis response at follow-up 4 weeks after stopping the treatment. Children may be more susceptible to systemic toxicity when treated with clascoterone (see Section 5.1 Pharmacodynamic Properties).
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
No clinical studies evaluating the drug interaction potential of Winlevi have been conducted.
In vitro studies.
In vitro findings suggest that clascoterone has no clinically meaningful effect on the PK of drugs metabolised by CYP1A2, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1 and 3A4.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
In a fertility and early embryonic development study in rats, clascoterone was administered subcutaneously at doses up to 12.5 mg/kg/day from 2 - 4 weeks before mating and throughout mating. Clascoterone increased pre-implantation loss at 12.5 mg/kg/day (estimated 35 times the AUC at a human dose of 6 g of cream per day). Adverse effects on male reproductive organs and testicular sperm counts were also seen at this dose. No effects were noted at doses up to 2.5 mg/kg/day (7 times the AUC at a human dose of 6 g of cream per day).
(Category D)
There are no available data on the use of Winlevi in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage, or adverse maternal or fetal outcomes.
Teratogenicity and embryofetal death were seen in animal embryofetal development studies. Clascoterone related fetal malformations (omphalocele, severe dilation of the lateral and third cerebral ventricles; thin skin, small size, and protruding tongue) were noted in rats at subcutaneous doses ≥ 1 mg/kg/day (2.8 times the AUC at a human dose of 6 g of cream per day). A no adverse effect level for teratogenicity was not established in this species. Post-implantation loss and resorptions were increased in rabbits at a subcutaneous dose of 1.5 mg/kg/day (13 times the AUC at a human dose of 6 g of cream per day). No embryofetal developmental toxicity was seen in this species at subcutaneous doses up to 0.4 mg/kg/day (~ 4 times the AUC at a human dose of 6 g of cream per day).
Winlevi should not be used during pregnancy.
There are no available data regarding the presence of clascoterone or its metabolite in human milk, the effects on the breastfed infant or the effects on milk production. The lack of clinical data during lactation precludes a clear determination of the risk of clascoterone to an infant during lactation. Therefore, the developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for clascoterone and any potential adverse effects on the breastfed child from clascoterone and any underlying maternal condition.4.7 Effects on Ability to Drive and Use Machines
The effects of Winlevi on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Clinical trial adverse reactions.
Clinical trials are conducted under very specific conditions. The adverse reaction rates observed in the clinical trials, therefore, may not reflect the rates observed in practice and should not be compared to the rates in the clinical trials of another drug. Adverse reaction information from clinical trials may be useful in identifying and approximating rates of adverse drug reactions in real-world use.
Local skin reactions were observed during the 12-week treatment and occurred in a similar percentage of patients treated with vehicle. Local skin reactions reported by ≥ 1% of patients treated with Winlevi are shown in the following table. See Table 1.
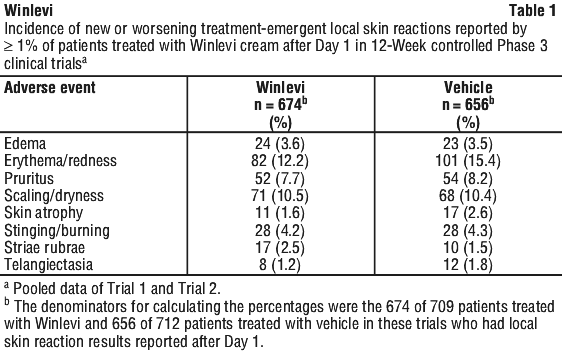 The only other treatment-emergent adverse event (TEAE) reported for ≥ 1% of patients in either treatment group in the pooled randomised, double-blind, vehicle-controlled studies was nasopharyngitis (1.4% Winlevi, 2.8% vehicle).
The only other treatment-emergent adverse event (TEAE) reported for ≥ 1% of patients in either treatment group in the pooled randomised, double-blind, vehicle-controlled studies was nasopharyngitis (1.4% Winlevi, 2.8% vehicle).
Clinical trial adverse reactions - paediatrics.
In patients aged 12 to 18 years of age, TEAEs were reported in 10.8% (34/316) of Winlevi-treated patients and 14.2% (46/325) in vehicle. The only TEAEs reported in ≥ 1% of patients aged 12 to 18 years in the pooled phase 3 studies, are shown in the following table. See Table 2.

Less common clinical trial adverse reactions.
Treatment-emergent adverse events occurring in < 1% of Winlevi treated patients (with a higher incidence compared to vehicle and occurring in at least 2 patients), aged 18 years and older, are presented below. The events are categorized by body system.
Infections and infestations.
Bronchitis, peritonsillar abscess.
General disorders and administration site conditions.
Application site dryness.
Gastrointestinal conditions.
Vomiting.
Hair colour changes.
Less common clinical trial adverse reactions - paediatrics.
Treatment-emergent adverse events occurring in < 1% of Winlevi treated patients (with a higher incidence compared to vehicle and occurring in at least 2 patients), aged 12 to 18 years, are presented below. The events are categorized by body system.
Infections and infestations.
Respiratory tract infection viral, sinusitis.
Gastrointestinal disorders.
Abdominal pain upper.
Reproductive system and breast disorders.
Dysmenorrhea.
Abnormal laboratory findings: hematologic, clinical chemistry and other quantitative data.
Clinical trial findings. Hyperkalemia.
Although clinical laboratory evaluations were not performed in the Phase 3 studies, changes in the clinical laboratory studies in phase 1 and phase 2 studies were identified. Shifts from normal to elevated potassium levels were observed in 3.7% (17/461) of clascoterone-treated patients and 3.9 (4/103) of vehicle-treated patients ages ranging from 12 to 64 (see Section 5.1 Pharmacodynamic Properties).
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Adverse effects can also be reported to [email protected] or 1800 726 229.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
Winlevi is for twice-daily topical use only.
In case of accidental ingestion, appropriate symptomatic measures should be taken. Although no data are available, the active ingredient in Winlevi has anti-androgenic effects, HPA-axis suppression and hyperkalaemia may be seen following accidental oral ingestion.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Clascoterone is an androgen receptor inhibitor. The exact mechanism of action of Winlevi for the topical treatment of acne vulgaris is unknown.
Hypothalamic-pituitary-adrenal (HPA) axis suppression.
HPA axis suppression was evaluated in adult (n=20), adolescent (n=22) and paediatric (n=27, < 12 years of age) patients with acne vulgaris, following twice daily application of Winlevi for 2 weeks, in two maximum use pharmacokinetic studies. HPA axis suppression, indicated by 30-minute post-stimulation serum cortisol level of ≤ 497 nmol/L, was observed in 1/20 (5%) of adult subjects, 2/22 (9%) of adolescent and 2/23 (8.7%) of paediatric patients at Day 14. All patients returned to normal HPA axis function at follow-up 4 weeks after the end of treatment.
Potassium.
Shifts from normal to elevated potassium levels were observed in 3.7% (17/461) of clascoterone-treated patients and 3.9 (4/103) of vehicle-treated patients ages ranging from 12 to 64.
Cardiac electrophysiology.
At approximately 2 times the systemic exposure observed with the maximum dose, Winlevi does not prolong the QT interval to any clinically relevant extent.
Clinical trials.
The safety and efficacy of Winlevi, when applied twice daily for 12 weeks for the treatment of acne vulgaris, were assessed in two identically designed, multicentre, randomised, double-blind, vehicle-controlled clinical trials (Trial 1 and Trial 2) enrolling 1440 patients with facial acne vulgaris. The trials enrolled patients 9 years or older with Investigator's Global Assessment (IGA) of moderate or severe facial acne vulgaris (score of 3 or 4), 30 to 75 inflammatory lesions (papules, pustules and nodules), and 30 to 100 non-inflammatory lesions (open and closed comedones). Patients with more than 2 facial nodules or nodulocystic acne were excluded.
A total of 1421 patients 12 years and older with facial acne vulgaris were enrolled. The treatment groups in each study were well-balanced with similar demographic and baseline characteristics in the intent-to-treat (ITT) population, both within and between Trial 1 and Trial 2. Of these subjects, 641 (45%) were 12 to 17 years of age, and 780 (55%) were 18 years of age or older. In addition, 62% of the patients were female, and 91% were Caucasian. At baseline, patients had a mean inflammatory lesion count of 42.4 and a mean non-inflammatory lesion count of 61.4. Additionally, approximately 83% of patients had an IGA score of 3 ("moderate").
Primary and secondary efficacy endpoints were assessed at Week 12. Co-primary efficacy endpoints included proportion of patients in each treatment group with at least a 2-point reduction in IGA compared to baseline and an IGA score of 0 (clear) or 1 (almost clear) and absolute change from baseline in non-inflammatory lesion count (NILC) and inflammatory lesion count (ILC). Secondary efficacy endpoints included absolute and percent change from baseline in total lesion count (TLC); percent change from baseline in non-inflammatory and inflammatory lesion count. The IGA success rate and mean absolute and percent reduction from baseline in acne lesion count after 12 weeks of treatment, for subjects 12 years of age and older, are presented in the following table. See Table 3.

5.2 Pharmacokinetic Properties
Absorption.
Following topical treatment of Winlevi for 2 weeks with a mean dose of approximately 6 grams applied twice daily to adult patients with moderate to severe acne vulgaris (n=20), systemic concentrations of clascoterone were at a steady state by Day 5. On Day 14, the mean ± SD maximum plasma concentration (Cmax) was 4.5 ± 2.9 nanogram/mL, the mean ± SD area under the plasma concentration-time over the dosing interval (AUCτ) was 37.1 ± 22.3 h*nanogram/mL and the mean ± SD average plasma concentration (Cavg) was 3.1 ± 1.9 nanogram/mL.
Distribution.
Plasma protein binding of clascoterone is 84% to 89% and is independent of concentrations, in vitro.
Metabolism.
Following topical treatment with Winlevi, the plasma concentrations of cortexolone, a possible primary metabolite of clascoterone, were detectable and generally below or near the lower limit of quantitation (0.5 nanogram/mL) in patients ≥ 12 years of age with acne vulgaris.
The in vitro study indicated that incubation of clascoterone with human cryopreserved hepatocytes generated cortexolone as the possible primary metabolite and other metabolites, including conjugated metabolites.
Excretion.
The excretion of clascoterone has not been fully characterized in humans.
Paediatrics.
In adolescent patients 12 to < 18 years of age (n=22), after 2 weeks of twice daily treatment, with a mean dose of approximately 6 grams of Winlevi per application (or mean dose of approximately 4 grams per application in younger, smaller subjects), steady-state concentrations of clascoterone were achieved by Day 5. Clascoterone systemic exposure in adolescents was similar to those observed in adults (see Section 5.2 Pharmacokinetic Properties, Absorption).
5.3 Preclinical Safety Data
Genotoxicity.
Clascoterone was not mutagenic in the Ames reverse mutation assay and was not clastogenic in the in vitro human lymphocyte chromosomal aberration assay. In rats, clascoterone administered via subcutaneous injection did not induce micronuclei in the bone marrow at 500 or 1000 mg/kg but a slight increase in micronuclei occurred in 2 of 5 rats, at 2000 mg/kg at 24 hours. At 48 hours, no increase was noted in any group. Overall, the weight of evidence indicates that clascoterone does not represent a genotoxic risk.
Carcinogenicity.
Clascoterone cream (0.1%, 1%, or 5%) was not carcinogenic after daily topical administration in a 2-year carcinogenicity study in rats. An increased incidence of the non-neoplastic finding of atrophy of the skin and subcutis, at the application site, was reported in males and females treated with the 1% and 5% clascoterone cream.6 Pharmaceutical Particulars
6.1 List of Excipients
Cetyl alcohol, citric acid monohydrate, disodium edetate, dl-alpha-tocopherol, mono- and di-glycerides, liquid paraffin, polysorbate 80, propylene glycol and purified water.
6.2 Incompatibilities
No data available.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Prior to dispensing.
Store the product in a refrigerator between 2°C and 8°C. Do not freeze.
Dispensing instructions for the pharmacist.
Direct the patient to store the product while in use at room temperature (below 25°C). Do not freeze. Discard the unused product 6 months after the date of dispensing.
6.5 Nature and Contents of Container
Winlevi is supplied in an epoxy-lined aluminum blind-end tube with a polypropylene cap closure.
Winlevi is supplied in tubes of 2 g, 10 g, 30 g and 60 g.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed by talking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
 Molecular formula: C24H34O5.
Molecular formula: C24H34O5.
Molecular weight: 402.5.
CAS number.
19608-29-8.7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Summary Table of Changes


 The only other treatment-emergent adverse event (TEAE) reported for ≥ 1% of patients in either treatment group in the pooled randomised, double-blind, vehicle-controlled studies was nasopharyngitis (1.4% Winlevi, 2.8% vehicle).
The only other treatment-emergent adverse event (TEAE) reported for ≥ 1% of patients in either treatment group in the pooled randomised, double-blind, vehicle-controlled studies was nasopharyngitis (1.4% Winlevi, 2.8% vehicle).

 Molecular formula: C24H34O5.
Molecular formula: C24H34O5.