What is in this leaflet
This leaflet answers some common questions about XERGIC.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have benefits and risks.
If you have any concerns about taking this medicine, talk to your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
What XERGIC is used for
XERGIC is used to relieve the symptoms of hayfever (seasonal allergic rhinitis) such as sneezing, itchy, watery or red eyes, and itchy, blocked or runny nose. The XERGIC 180 mg tablets are also used to relieve the symptoms of urticaria, otherwise known as hives or itchy rash.
XERGIC contains the active ingredient fexofenadine and belongs to a group of medicines called antihistamines. It works by blocking the action of histamine and other substances produced by the body, which are causing your allergies or itchiness.
XERGIC is a non-sedating antihistamine, which means it should not make you drowsy or slow down your reactions.
Use in children
XERGIC 120 and 180 are not recommended for use in children under the age of 12, as their safety and effectiveness have not been established in this age group.
There is no evidence that XERGIC is addictive.
Before you take XERGIC
When you must not take it
Do not take XERGIC if you are allergic to any other medicines containing fexofenadine (such as TELFAST), terfenadine (such as TELDANE) or any of the other ingredients in XERGIC. These are listed at the end of this leaflet.
Do not take XERGIC if the expiry date (EXP.) printed on the pack has passed. If you take this medicine after the expiry date, it may not work as well.
Do not take XERGIC if the packaging shows signs of tampering or the tablets do not look quite right.
If you are not sure whether you should start taking this medicine, talk to your doctor or pharmacist.
Before you start to take it
Tell your doctor or pharmacist if you are allergic to any other medicines, foods, dyes or preservatives.
Tell your doctor or pharmacist if you are pregnant or plan to become pregnant. They will discuss the risks and benefits of taking XERGIC during pregnancy.
Tell your doctor or pharmacist if you are breastfeeding or wish to breastfeed. They will discuss the risks and benefits of taking XERGIC when breastfeeding.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from a pharmacy, supermarket or health food shop.
How well XERGIC works may be affected if you are taking antacids. Your doctor or pharmacist can tell you what to do if you are taking antacids.
How to take XERGIC
Follow all directions given to you by your doctor or pharmacist carefully.
They may differ from the information contained in this leaflet.
If you do not understand the instructions on the pack, ask your doctor or pharmacist.
How to take it
Swallow the tablets whole with a glass of water.
XERGIC may be taken with or without food.
Adults and children 12 years and older:
For the relief of the symptoms of hayfever, the usual dosage is one XERGIC 120 mg tablet daily or one XERGIC 180 mg tablet daily, when required.
For relief of the symptoms of urticaria, the usual dosage is one XERGIC 180 mg tablet daily when required.
If XERGIC does not relieve your symptoms, do not take extra tablets. Tell your doctor or pharmacist.
If you take too much (overdose)
Immediately telephone your doctor, or the Poisons Information Centre (telephone 13 11 26), or go to Accident and Emergency at the nearest hospital, if you think you or anyone else may have taken too much XERGIC. Do this even if there are no signs of discomfort or poisoning.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking XERGIC.
All medicines can have side effects.
Although most people will not experience any side effects, some of the side effects that may occur with XERGIC are:
- headache
- tiredness
- nausea
- dizziness
- drowsiness
These same effects were seen in patients taking placebo capsules during the clinical studies on the innovator product, Telfast.
Other side effects not listed above may also occur in some patients.
After taking XERGIC
Storage
Keep XERGIC where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Keep your tablets in the pack until it is time to take them. If you take the tablets out of the pack they will not keep well.
Keep your tablets in a cool dry place where the temperature stays below 25°C.
Do not store XERGIC or any other medicine in the bathroom or near a sink.
Do not leave XERGIC in the car or on window sills. Heat and dampness can destroy some medicines.
Disposal
If your doctor or pharmacist tells you to stop taking XERGIC, or your tablets have passed their expiry date, ask your pharmacist what to do with any that are left over.
Product description
What it looks like
XERGIC tablets are available in 2 strengths:
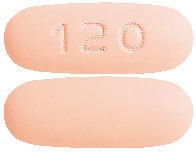
- 120 mg tablets - Peach, oblong, bi-convex film-coated tablets. Plain on both sides.
- 180 mg tablets - Yellow, oblong, bi-convex film-coated tablets. Plain on one side with a central breakline on the other side.
XERGIC 120 and XERGIC 180 blister packs contain either 10 or 30 tablets.
Ingredients
The active ingredient in XERGIC is fexofenadine hydrochloride. Each XERGIC tablet contains either 120 mg or 180 mg of fexofenadine hydrochloride.
The tablets also contain the following inactive ingredients:
- croscarmellose sodium
- maize starch
- microcrystalline cellulose
- magnesium stearate
- hypromellose
- povidone
- titanium dioxide
- macrogol 400
- macrogol 4000
- iron oxide yellow CI77492
- iron oxide red CI77491 (120 mg tablets only)
The tablets do not contain sucrose or lactose.
Supplier
XERGIC is supplied by:
Alphapharm Pty Limited
(ABN 93 002 359 739)
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
Phone: (02) 9298 3999
www.alphapharm.com.au
Medical Information
Phone: 1800 028 365
Australian registration numbers:
XERGIC 120 fexofenadine hydrochloride 120 mg tablets - AUST R 115377
XERGIC 180 fexofenadine hydrochloride 180 mg tablets - AUST R 115378
This leaflet was prepared on
14 July 2011.
Published by MIMS October 2011