What is in this leaflet
This leaflet answers some common questions about Zavicefta. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Zavicefta against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Zavicefta is used for
Zavicefta is an antibiotic used to treat serious infections caused by bacteria (germs), such as:
- serious infections of the tissues and organs within the abdomen
- serious infections of the urinary tract including kidney infections.
- infections of the lungs that are caught in hospital including pneumonia caught from the ventilator (machine that helps a patient to breathe).
Zavicefta will not work against fungal or viral infections (such as colds or the flu).
Zavicefta is a combination of two medicines, ceftazidime and avibactam.
Ceftazidime belongs to a group of antibiotics called cephalosporins. It works by killing the bacteria that is causing the infection.
Avibactam belongs to a group of medicines called beta-lactamase inhibitors. It works by inhibiting the enzymes that break down ceftazidime in the body.
Zavicefta is given by injection and is usually given in hospital.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is available only with a doctor's prescription.
There is not enough information to recommend the use of this medicine for children under the age of 18 years.
Before treatment with Zavicefta
When Zavicefta must not be used
Zavicefta must not be given if you have an allergy to:
- any medicine containing ceftazidime or avibactam, the active ingredients in Zavicefta
- any of the ingredients listed at the end of this leaflet.
- any other cephalosporin antibiotics
- severe hypersensitivity to any other type of beta-lactam antibacterial agents such as penicillins, monobactams or carbapenems.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not use Zavicefta if you have had a serious allergy to penicillins or beta-lactam antibiotics (e.g., carbapenems).
You may be more likely to have an allergic reaction to ceftazidime if you have had an allergic reaction to these other antibiotics.
The hospital staff will check that the expiry date printed on the pack has not passed and that the packaging is not torn or showing signs of tampering.
If you are not sure whether you should be given this medicine, talk to your doctor.
Before treatment with Zavicefta
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- kidney problems
- liver problems
- severe diarrhoea after taking any antibiotics
- anaemia
- if you are on a controlled sodium diet.
Tell your doctor if you are pregnant or plan to become pregnant. Your doctor can discuss with you the risks and benefits involved.
Do not breast-feed if you are being given this medicine. The active ingredient, ceftazidime passes into breast milk and there is a possibility that your baby may be affected. The effect of the other active ingredient, avibactam, is unknown.
Tell your doctor if you are planning to have any urine tests conducted. Zavicefta may interfere with laboratory tests for the detection of glycosuria.
If you have not told your doctor about any of the above, tell him/ her before you start treatment with Zavicefta. Your doctor can discuss with you the risks and benefits involved.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Zavicefta may interfere with each other. These include:
- probenecid, a medicine used to prevent gout and gouty arthritis
- a type of antibiotic called aminoglycosides, such as gentamicin and tobramycin
- a type antibiotic called cephalosporins, such as cefazolin and ceftazidime
- chloramphenicol, a medicine used to treat eye infections.
- medicines used to pass urine, such as frusemide.
These medicines may be affected by Zavicefta or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How Zavicefta is given
Zavicefta treatment is mostly given in hospital by your doctor or nurse.
The usual adult dose is 1 vial containing 2000 mg ceftazidime and 500 mg avibactam every 8 hours. It is given as a slow injection (infusion) directly into a vein, which usually takes 120 minutes.
Treatment is usually given every day for 5 days to up to 14 days. Your doctor will decide how many days you will need to treatment with Zavicefta.
Ask the doctor if you want more information about the dose of Zavicefta and how it is given.
Your doctor may lower the dose if you have kidney problems.
If too much has been given (overdose)
As Zavicefta is given under the close supervision of the doctor or nurse, it is unlikely that you will be given too much. If you are concerned that too much has been given, tell the doctor, nurse or pharmacist immediately.
Symptoms of an overdose may include confusion, difficulty remembering or focusing, drowsiness, muscle weakness, involuntary twitching, difficulty speaking or swallowing, seizures or coma.
While being treated with Zavicefta
Things you must do
If you are about to be started on any new medicine remind your doctor and pharmacist that you are taking Zavicefta.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are being treated with this medicine. It may affect other medicines used during surgery.
If you become pregnant while taking this medicine, tell your doctor immediately.
If you are about to have any blood tests tell your doctor that you are being treated with this medicine. It may interfere with the results of some tests.
If you get severe diarrhoea tell your doctor, pharmacist or nurse immediately. Do this even if it occurs several weeks after Zavicefta has been stopped.
Diarrhoea may mean that you have a serious condition affecting your bowel. You may need urgent medical care. Do not take any diarrhoea medicine without first checking with your doctor.
Things to be careful of
Be careful driving or operating machinery until you know how Zavicefta affects you. This medicine may cause dizziness in some people. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Zavicefta.
This medicine helps most people with certain serious infections but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- diarrhoea, stomach pain
- headache, dizziness
- nausea, vomiting
- rash, itchy skin
- white, furry layer on the tongue
- unusual vaginal discharge
- changes in taste, or a metallic taste in the mouth
- fever
- redness, pain or swelling where Zavicefta was given into a vein.
The above list includes the more common side effects of your medicine.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- abdominal pain
- swelling of the limbs, face, lips, mouth or throat
- shortness of breath or breathing difficulties
- unusual weight gain
- changes to urine volume
- numbness or a tingling sensation
- swelling of the face, lips, mouth or throat which may cause difficulty in swallowing or breathing
- rash, itching, hives or severe skin reactions such as painful red areas, large blisters, peeling of your skin, and bleeding in the lips, eyes, mouth, nose and genitals
- diarrhoea that keeps getting worse or does not go away, or stools that contains blood or mucus
- yellowing of the skin and eyes (jaundice).
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
Occasionally, Zavicefta may be associated with changes in your blood that may require your doctor or nurse to do certain blood tests.
After stopping your treatment
Tell your or your child's doctor or pharmacist if you notice any of the following side effects, particularly if they occur several weeks after stopping treatment with Zavicefta:
- severe stomach cramps
- watery and severe diarrhoea (which may be bloody), fever, in combination with one or both of the above.
Zavicefta can cause some bacteria, which are normally present in the bowel and normally harmless, to multiply and therefore cause the above symptoms. You may need urgent medical attention.
Tell your doctor if you notice any other side effects.
Other side effects not listed above may also occur in some people.
After stopping treatment
Storage
The hospital and/or pharmacy staff will store Zavicefta vials in a safe place at a temperature below 25°C and away from light. They will also prepare and store the infusion bags/ bottles ready for the injection.
Hospital staff will make sure the medicine is not used after the expiry date printed on the bag.
Product description
What it looks like
Zavicefta is a white to yellow powder supplied in a glass vial.
This product is supplied in packs of 10 vials.
Ingredients
Zavicefta contains 2000 mg of ceftazidime (as pentahydrate) and 500 mg of avibactam (as sodium) as the active ingredients.
The inactive ingredient is anhydrous sodium carbonate.
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
Supplier
Zavicefta is supplied in Australia by:
Pfizer Australia Pty Ltd
Sydney, NSW.
Toll Free Number: 1800 675 229
www.pfizer.com.au.
Australian Registration Number
AUST R 301205.
This leaflet was prepared in May 2019.
® Registered trademark.
© Pfizer Australia Pty Ltd 2019.
Published by MIMS December 2019

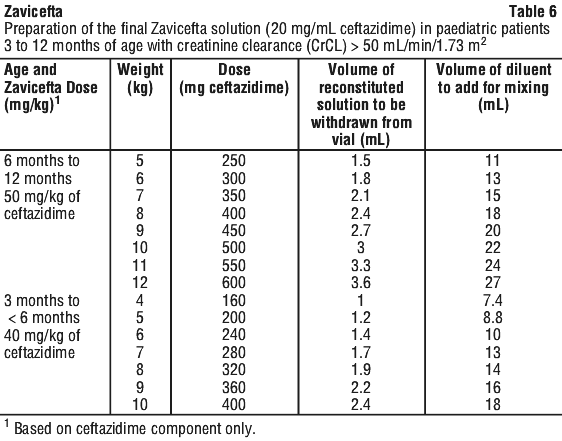
 For cUTI including pyelonephritis, the total duration of treatment could be increased to 14 days for patients with bacteraemia.
For cUTI including pyelonephritis, the total duration of treatment could be increased to 14 days for patients with bacteraemia.

 There is insufficient information to recommend a dosage regimen for paediatric patients less than 2 years of age with a creatinine clearance of < 50 mL/min/1.73 m2.
There is insufficient information to recommend a dosage regimen for paediatric patients less than 2 years of age with a creatinine clearance of < 50 mL/min/1.73 m2.
 The most common adverse reactions occurring in ≥ 5% of patients treated with Zavicefta were Coombs direct test positive, nausea, and diarrhoea. Nausea and diarrhoea were usually mild or moderate in intensity. No clinically significant differences were observed in the safety profile across indications.
The most common adverse reactions occurring in ≥ 5% of patients treated with Zavicefta were Coombs direct test positive, nausea, and diarrhoea. Nausea and diarrhoea were usually mild or moderate in intensity. No clinically significant differences were observed in the safety profile across indications.


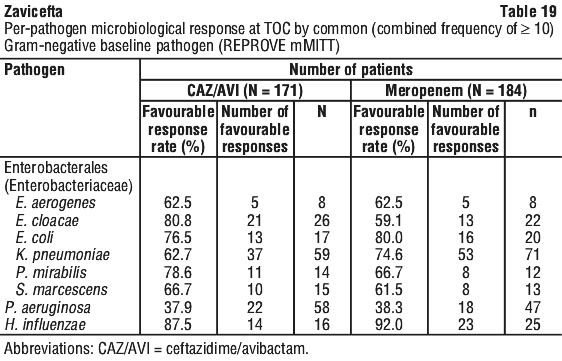
 Clinical cure rates at TOC by pathogen in the microbiologically modified intent-to-treat (mMITT) population for Gram-negative aerobes are shown in Table 12.
Clinical cure rates at TOC by pathogen in the microbiologically modified intent-to-treat (mMITT) population for Gram-negative aerobes are shown in Table 12. A further 432 adults with complicated intra-abdominal infections were randomised and received treatment in a multi-centre, double-blind study (RECLAIM 3) conducted in 3 Asian countries (China, Republic of Korea and Vietnam). The patient population and key aspects of the study design were identical to RECLAIM apart from the primary efficacy endpoint of clinical response at the TOC visit being solely in the CE population (see Table 13).
A further 432 adults with complicated intra-abdominal infections were randomised and received treatment in a multi-centre, double-blind study (RECLAIM 3) conducted in 3 Asian countries (China, Republic of Korea and Vietnam). The patient population and key aspects of the study design were identical to RECLAIM apart from the primary efficacy endpoint of clinical response at the TOC visit being solely in the CE population (see Table 13). Clinical cure rates at TOC by pathogen in the microbiologically modified intent-to-treat (mMITT) population for Gram-negative aerobes are shown in Table 14.
Clinical cure rates at TOC by pathogen in the microbiologically modified intent-to-treat (mMITT) population for Gram-negative aerobes are shown in Table 14. In Phase 3 cIAI clinical trials, death occurred in 2.1% (18/857) of patients who received Zavicefta and metronidazole and in 1.4% (12/863) of patients who received meropenem. Among a subgroup with baseline CrCl 30 to 50 mL/min, death occurred in 16.7% (9/54) of patients who received Zavicefta and metronidazole and 6.8% (4/59) of patients who received meropenem. Patients with CrCl 30 to 50 mL/min received a lower dose of Zavicefta than is currently recommended for patients in this sub-group.
In Phase 3 cIAI clinical trials, death occurred in 2.1% (18/857) of patients who received Zavicefta and metronidazole and in 1.4% (12/863) of patients who received meropenem. Among a subgroup with baseline CrCl 30 to 50 mL/min, death occurred in 16.7% (9/54) of patients who received Zavicefta and metronidazole and 6.8% (4/59) of patients who received meropenem. Patients with CrCl 30 to 50 mL/min received a lower dose of Zavicefta than is currently recommended for patients in this sub-group. Favourable microbiological response rates at TOC by pathogen in the mMITT population are shown in Table 16.
Favourable microbiological response rates at TOC by pathogen in the mMITT population are shown in Table 16.
 All-cause mortality rates at Day 28 (cMITT) was 8.4% (30/356) and 7.3% (27/370) ceftazidime/avibactam and meropenem treated patients, respectively.
All-cause mortality rates at Day 28 (cMITT) was 8.4% (30/356) and 7.3% (27/370) ceftazidime/avibactam and meropenem treated patients, respectively.
 The predominant pathogens isolated at baseline were E. coli (55/69, 79.7%) and P. aeruginosa 23/69 (33.3%). Favourable clinical response rates at TOC by baseline pathogen in the micro-ITT population are presented in Table 21.
The predominant pathogens isolated at baseline were E. coli (55/69, 79.7%) and P. aeruginosa 23/69 (33.3%). Favourable clinical response rates at TOC by baseline pathogen in the micro-ITT population are presented in Table 21.
 The predominant baseline pathogen was E. coli (71/77, 92.2%). Favourable microbiological response rates by baseline pathogen at TOC in the micro-ITT population are presented in Table 23.
The predominant baseline pathogen was E. coli (71/77, 92.2%). Favourable microbiological response rates by baseline pathogen at TOC in the micro-ITT population are presented in Table 23.


