What is in this leaflet
This leaflet answers some common questions about ZOELY. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking ZOELY against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Read this information carefully before you start taking ZOELY tablets.
Keep this leaflet. You may need to read it again.
What ZOELY is used for
ZOELY is an oral contraceptive, commonly known as a "Birth Control Pill" or "The Pill" that has been prescribed to prevent you from getting pregnant.
ZOELY prevents pregnancy in several ways:
- It inhibits the egg release by stopping it maturing.
- Changing the cervical mucus consistency making it difficult for the sperm to reach the egg.
- Changing the lining of the uterus making it less suitable for implantation.
ZOELY consists of 24 white tablets, each containing 2 active ingredients: 2.5 milligrams of nomegestrol acetate (a progestogen) and 1.5 milligrams of estradiol (the natural estrogen) and 4 yellow inactive tablets. ZOELY contains estradiol, the natural estrogen, compared with most Pills which contain the synthetic estrogen ethinylestradiol.
Oral contraceptives are an effective method of birth control. When taken correctly (without missing tablets) the chance of becoming pregnant is very low.
The following non-contraceptive health benefits have been associated with the combined Pill:
- Your periods may be lighter and shorter. As a result, the risk of anaemia may be lower.
- Your period pains may become less severe or may completely disappear.
ZOELY is available only with a doctor's prescription.
When you must not use ZOELY
Do not take ZOELY if you have an allergy to:
- any medicine containing nomegestrol acetate or estradiol
- any ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or troubled breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not use the Combined Pill if you have or have had any of the conditions listed below. If any of these conditions apply to you, tell your doctor before starting to use ZOELY. Your doctor may advise you to use a different type of Pill or an entirely different (non-hormonal) method of birth control.
- a blood clot (venous thrombosis) in a blood vessel of your legs, lungs (embolus) or other organs (see also the section later in this leaflet called "The Pill and Thrombosis")
- a heart attack or stroke
- a condition that may be a first sign of a heart attack (such as angina pectoris which causes severe chest pain) or stroke (such as a transient ischaemic attack)
- a serious risk factor or several risk factors for developing a blood clot
- very high blood pressure
- a very high level of fat in the blood (cholesterol or triglycerides)
- a disorder affecting your blood clotting - for instance Protein C deficiency
- if you have major surgery (e.g., an operation) and your ability to move around is limited for a long period of time (see also the section later in this leaflet called "The Pill and Thrombosis")
- diabetes mellitus with blood vessel damage
- a history of migraine accompanied by e.g. visual symptoms, speech disability, or weakness or numbness in any part of the body
- pancreatitis (an inflammation of the pancreas) associated with high levels of fatty substances in your blood
- jaundice (yellowing of the skin) or severe liver disease and your liver is not yet working normally
- a benign or malignant liver tumour
- a cancer that may grow under the influence of sex hormones (e.g. of the breast or of the genital organs)
- any unexplained vaginal bleeding
- you are pregnant or think you might be pregnant.
If any of these conditions appear for the first time while using the Pill, stop taking it at once and tell your doctor. In the meantime use non-hormonal contraceptive measures. See also "General Notes" in the next section.
Do not take ZOELY if the expiry date printed on the pack has passed.
Do not take ZOELY if the packaging is torn or shows signs of tampering.
What you need to know before using ZOELY
General notes
In this leaflet, several situations are described where you should stop taking the Pill, or where the reliability of the Pill may be decreased. In such situations you should not have sex or you should take extra non-hormonal contraceptive precautions, e.g. use a condom or another barrier method. Do not use rhythm or temperature methods. These methods can be unreliable because the Pill alters the usual changes in temperature and cervical mucus that occur during the menstrual cycle.
If you are concerned about contracting a sexually transmitted infection (STI), ask your partner to wear a condom when having sexual intercourse with you.
ZOELY will not protect you from HIV (AIDS) or any other sexually transmitted infections. To help protect yourself from STIs, you need to use a barrier contraceptive such as a condom, but even barrier contraceptives may not protect you against human papilloma virus (HPV).
Before you start to use ZOELY
You should have a thorough medical check-up, including a Pap smear, breast check, blood pressure check and urine check.
You must tell your doctor if you are allergic to any foods, dyes, preservatives or any other medicines.
You must tell your doctor if you smoke. The risk of having a heart attack or stroke increases as you get older. It also increases the more you smoke. When using the Pill you should stop smoking, especially if you are older than about 35 years of age.
You must tell your doctor if you have any of the conditions listed below. You may need to be kept under close observation. Your doctor can explain this to you. Tell your doctor if:
- anyone in your immediate family has had breast cancer
- you suffer from epilepsy (see "Taking other medicines")
- you are overweight
- you have diabetes
- you have high blood pressure
- you have a heart valve disorder or a certain heart rhythm disorder
- anyone in your immediate family has had a thrombosis, a heart attack or a stroke
- you have sickle cell disease you have liver disease (jaundice) or gall bladder disease
- you have Crohn's disease or ulcerative colitis (chronic inflammatory bowel disease)
- you have systemic lupus erythematosus (SLE, a disease affecting the skin all over the body)
- you have haemolytic uraemic syndrome (HUS; a disorder of blood coagulation causing failure of the kidneys)
- if you have elevated fatty acid levels in the blood (hypertriglyderidaemia) or a positive family history for this condition (familial hypertriglyceridaemia). If so, you may be at an increased risk of developing pancreatitis (inflammation of the pancreas) when using combined pills
- you have a condition that occurred for the first time or worsened during pregnancy or previous use of sex hormones (e.g. hearing loss, a metabolic disease called porphyria, a skin disease called herpes gestationis, a neurological disease called Sydenham's chorea)
- you have or have had chloasma (yellow brownish pigmentation patches on the skin, particularly of the face); if so, avoid too much exposure to the sun or ultraviolet radiation
- you have recently given birth you are at an increased risk of blood clots. You should ask your doctor how soon after delivery you can start using ZOELY (see also the section later in this leaflet called "The Pill and Thrombosis")
- You need an operation or if your ability to move around is limited for a long period of time. This includes travelling by plane for greater than 4 hours.
Tell your doctor if any of the above conditions appear for the first time, recur or worsen while using the Pill.
The Pill and Thrombosis
A thrombosis is the formation of a blood clot which may block a blood vessel.
A thrombosis sometimes occurs in the deep veins of the legs (deep venous thrombosis). If this blood clot breaks away from the veins where it is formed, it may reach and block the arteries of the lungs, causing a so-called "pulmonary embolism". Deep venous thrombosis is a rare occurrence. It can develop whether or not you are taking the Pill. The risk is higher in Pill-users than in non-users. The chance of getting a thrombosis is highest during the first year a woman uses the Pill. The risk is also higher if you restart using a combined pill (the same product or a different product) after a break of 4 weeks or more. Thrombosis can also happen if you become pregnant. The risk of thrombosis also increases if you gave birth less than a few weeks ago. ZOELY contains estradiol instead of ethinylestradiol. It is not yet known how ZOELY influences the risk of getting a thrombosis compared with other contraceptive pills.
Blood clots can also occur very rarely in the blood vessels of the heart (causing a heart attack) or the brain (causing a stroke). Extremely rarely blood clots can occur in the liver, gut, kidney or eye.
Very occasionally thrombosis may cause serious permanent disabilities or may even be fatal.
If you develop high blood pressure while using the Pill, you may be told to stop using it.
The risk of having deep venous thrombosis is temporarily increased as a result of an operation or immobilisation (for example when you have your leg or legs in plaster or splints). In women who use the Pill, the risk may be yet higher. Tell your doctor you are using the Pill well in advance of any expected hospitalisation or surgery. Your doctor may tell you to stop taking the Pill several weeks before surgery or at the time of immobilisation. Your doctor will also tell you when you can start taking the Pill again after you are back on your feet.
If you notice possible signs of a thrombosis, stop taking the Pill and consult your doctor immediately (See also "When should you contact your doctor?").
The Pill and Cancer
Regularly examine your breasts.
The information given below was obtained from studies of women who used combined oral hormonal contraceptives, as the combined pill containing ethinylestradiol, and from an additional study that included both oral and non-oral hormonal contraceptive-users.
In studies with the combined Pill, breast cancer has been diagnosed slightly more often in women who use the Pill than in women of the same age who do not use the Pill. This slight increase in the numbers of breast cancer diagnoses gradually disappears during the course of the 10 years after stopping use of the Pill.
In the additional study that included both oral and non-oral hormonal contraceptive-users, the occurrence of breast cancer was reported to increase the longer the women used the contraceptive. The difference in the reported risk of breast cancer between women who have never used the contraceptive and those who had used the contraceptive was small: 13 additional cases of breast cancer per 100,000 women-years.
It is not known whether this is caused by the Pill. It may be that the women were examined more often, so that the breast cancer was noticed earlier.
Tell your doctor immediately if you have severe pain in your stomach.
In rare cases benign liver tumours and even more rarely, malignant liver tumours have been reported in users of the Pill. These tumours may lead to internal bleeding.
Chronic infection with Human Papilloma Virus (HPV) is the single most important risk factor for cervical cancer. HPV is a sexually transmitted infection. In women who use combined oral contraceptives for a long time the chance of getting cervical cancer may be slightly higher. This finding may not be caused by the Pill itself but may be related to sexual behaviour and other factors.
The Pill and Pregnancy
Tell your doctor immediately if you are pregnant or think you are pregnant while you are using ZOELY. ZOELY must not be used during pregnancy.
The Pill and Breastfeeding
ZOELY is not recommended for use during breastfeeding. If you wish to take the Pill while breastfeeding, please seek the advice of your doctor.
The Pill and Ability to Drive
There are no observed effects.
Taking Other Medicines
Tell your doctor if you are taking or have recently taken any other medicines or herbal products you buy without a prescription from a pharmacy, supermarket or health food shop.
Some medicines may stop the Pill from working properly. These include:
- medicines for epilepsy such as
- phenytoin,
- primidone,
- barbiturates,
- carbamazepine,
- oxycarbamazepine,
- topiramate,
- felbamate - medicines for tuberculosis
(e.g. rifampicin and rifabutin) - medicines for HIV infections (ritonavir, nevirapine, nelfinavir, efavirenz)
- medicines for Hepatitis C virus infection (e.g. boceprevir, telaprevir)
- antibiotics
(e.g. penicillin, ampicillin, tetracyclines) - antifungals
(e.g. griseofulvin) - medicines for high blood pressure in the blood vessels of the lungs (bosentan)
- herbal medicines containing St John's Wort primarily for the treatment of depressive moods
If you are taking medicines or herbal products that might make ZOELY less effective, a barrier contraceptive method should also be used. Since the effect of another medicine on ZOELY may last up to 28 days after stopping the medicine, it is necessary to use the additional barrier contraceptive method for that long.
ZOELY may also interfere with the working of other medicines - such as ciclosporin and the anti-epileptic lamotrigine.
The HCV combination drug regimen ombitasvir/paritaprevir/ritonavir with or without dasabuvir may cause increases in liver function blood test results (increase in ALT liver enzyme) in women using CHCs containing ethinylestradiol. ZOELY contains estradiol instead of ethinylestradiol. It is not known whether an increase in ALT liver enzyme can occur when using ZOELY with this HCV combination drug regimen. Your doctor will advise you.
When should you contact your doctor?
Regular check-ups
When you are using the Pill, your doctor will tell you to return for regular check-ups. You should have a check-up at least once a year.
Contact your doctor as soon as possible if:
- you notice any changes in your own health, especially involving any of the items mentioned in this leaflet (see also "When you must not use ZOELY?" and "Before you start to use ZOELY"); do not forget about the items related to your immediate family
- you feel a lump in your breast
- you are going to use other medicines (see also "Taking Other Medicines")
- your ability to move around is limited for a long period of time or you are to have surgery (consult your doctor at least 4 weeks in advance)
- you have unusual, heavy vaginal bleeding
- you forgot tablets at the beginning or end of the pack and had intercourse in the seven days before
- you have severe diarrhoea
- you miss your period twice in a row or suspect you are pregnant. Do not start the next pack until told to by your doctor.
Stop taking tablets and see your doctor immediately if you notice possible signs of thrombosis, myocardial infarction or a stroke such as:
- an unusual cough
- severe pain in the chest which may reach the left arm - this discomfort may include the back, jaw, throat, arm, stomach
- feeling of being full, having indigestion or choking
- sweating, nausea, vomiting, anxiety
- breathlessness or rapid breathing
- any unusual, sudden, severe or prolonged headache or migraine attack
- partial or complete loss of vision, or double vision
- confusion, slurring or speech disability
- sudden changes to your hearing, sense of smell or taste
- dizziness or fainting
- fast or irregular heartbeat
- weakness or numbness in any part of your body
- severe pain in your stomach
- severe pain or swelling in either of your legs
- pain or tenderness in the leg which may be felt only when standing or walking
- warmth, red or discoloured skin on the leg
- sudden pain, swelling and slight blue discoloration of an extremity
- sudden trouble walking, loss of balance or coordination
The situations and symptoms mentioned above are described and explained in more detail in the following section "What you need to know before using ZOELY".
How to use ZOELY
A ZOELY pack contains strips of 28 tablets: 24 white tablets with active substances and 4 yellow tablets that do not contain active substances. Each time you start a new strip of ZOELY, take the first white active tablet in the left-hand top corner (see 'Start').
Choose from the 7 stickers with day indicators the one that begins with your starting day, e.g. if you start on a Wednesday, use the day label sticker that starts 'WED'. Stick it on the strip, just above the row of white active tablets where it reads 'Place day label here'. This allows you to check whether you took your daily tablet.
Take one tablet each day at about the same time, with some liquid if necessary.
Follow the direction of the arrows on the pack taking the white active tablets first followed by the yellow inactive tablets.
A period should begin during the 4 days that you use the yellow inactive tablets (the withdrawal bleed). Usually it will start on day 2-3 after the last white tablet and may not have finished before the next strip is started.
Start taking your next pack immediately after the last yellow inactive tablet, even if your period hasn't finished. This means you will always start new packs on the same day of the week, and also means that you have your period on about the same days, each month.
When no hormonal contraception has been used in the past month
Start taking ZOELY on the first day of your cycle, i.e. the first day of menstrual bleeding. ZOELY will work immediately, it is not necessary to use an additional contraceptive method.
You may also start on days 2-5 of your cycle, but in that case make sure you also use an additional contraceptive method (barrier method) for the first 7 days of tablet-taking in the first cycle.
When changing from another combined hormonal contraceptive (combined oral contraceptive (COC), no problem vaginal ring, or transdermal patch)
You can start taking ZOELY the day after you take the last tablet from your present Pill pack. If your present Pill pack contains inactive tablets (placebo) tablets you can start ZOELY on the day after taking the last active tablet (if you are not sure which this is, ask your doctor or pharmacist). You can also start later, but never later than the day following the tablet-free break of the present Pill (or the day after the last inactive tablet of your present Pill).
In case you use a vaginal ring or transdermal patch, you should start using ZOELY preferably on the day of removal, but at latest when the next ring or patch would have been applied.
If you have used the Pill, patch or ring consistently and correctly and if you are sure that you are not pregnant, you can stop taking the Pill or remove the ring or patch on any day and start using ZOELY immediately.
If you follow these instructions, it is not necessary to use an additional contraceptive method.
When changing from a progestogen-only method (minipill)
You can stop taking the minipill any day and start taking ZOELY the next day, at the same time. But make sure you also use an additional contraceptive method (a barrier method) for the first 7 days of tablet-taking.
When changing from an injectable, an implant or a progestogen-releasing intrauterine device (IUD)
Start using ZOELY when your next injection is due or on the day that your implant or your IUD is removed. Make sure you also use an additional contraceptive method (a barrier method) for the first 7 days of tablet-taking.
After having a baby
If you have just had a baby, your doctor may tell you to wait until after your first normal period before you start taking ZOELY. Sometimes it is possible to start sooner. Your doctor will advise you. If you are breast-feeding and want to take ZOELY, you should discuss this first with your doctor.
After a miscarriage or abortion
Your doctor will advise you.
Additional contraceptive precautions
When additional contraceptive precautions are required you should either abstain from vaginal sex, or use a barrier method of contraception, a condom or a cap (diaphragm) plus spermicide. Rhythm methods are not advised as the Pill disrupts the cyclical changes associated with the natural menstrual cycle e.g. changes in temperature and cervical mucus.
What to do if...
You forget to take your tablets
The following advice only refers to missed white active tablets
- If you are less than 24 hours late in taking a tablet,
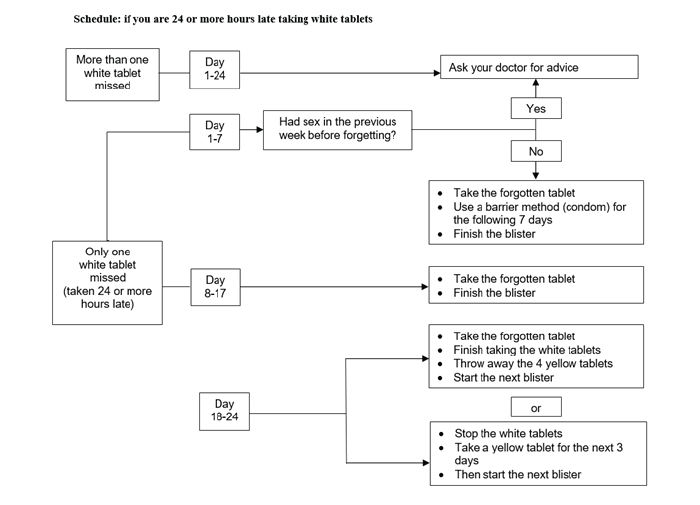
the reliability of the Pill is maintained. Take the tablet as soon as you remember and take further tablets at the usual time. - If you are 24 or more hours late in taking any tablet,
the reliability of the Pill may be reduced. The more consecutive tablets you have missed, the higher the risk that the contraceptive efficacy is decreased. There is a particularly high risk of becoming pregnant if you miss white active tablets at the beginning or at the end of the strip. Therefore you should follow the rules given below.
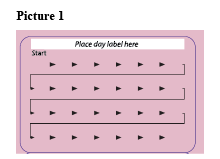
Day 1-7 (the first 7 days of white active tablet intake, see picture 1 and the schedule at the end of this leaflet)
Take the last white active missed tablet as soon as you remember (even if this means taking two tablets at the same time) and take the next tablets at the usual time. Use a barrier method such as a condom as an extra precaution until you have taken your tablets correctly for 7 days in a row. If you had sexual intercourse in the week before missing the tablets, there is a possibility of becoming pregnant. So tell your doctor immediately.
Days 8 to 17 from the first white active tablet intake (see picture 1 and the schedule at the end of this leaflet)
Take the last missed tablet as soon as you remember (even if this means taking two tablets at the same time) and take the next tablets at the usual time. If you have taken your tablets correctly in the 7 days prior to the missed tablet, the protection against pregnancy is not reduced, and you do not need to use extra precautions. However, if you have missed more than 1 tablet, use a barrier method such as a condom as an extra precaution until you have taken your tablets correctly for 7 days in a row.
Day 18-24 (the last 7 days of white active tablet intake, see picture 1 and the schedule at the end of this leaflet)
There is a particularly high risk of becoming pregnant if you miss white active tablets close to the yellow placebo tablet interval. By adjusting your intake schedule this higher risk can be prevented.
The following two options can be followed. You do not need to use extra precautions if you have taken your tablets correctly in the 7 days prior to the missed tablet. If this is not the case, you should follow the first of these two options and use a barrier method such as a condom as an extra precaution until you have taken your tablets correctly for 7 days in a row.
Option 1)
Take the last missed white active tablet as soon as you remember (even if this means taking 2 tablets at the same time) and take the next tablets at the usual time. Start the next strip as soon as the white active tablets in the current strip are finished, so skip the yellow placebo tablets. You may not have your period until you take the yellow placebo tablets at the end of the second strip, but you may have spotting (drops or flecks of blood) or breakthrough bleeding while taking the white active tablets.
Option 2)
Stop taking the active white tablets and start taking the placebo yellow tablets so that the total number of placebo plus missed active white tablets is not more than 4. At the end of this interval, start the next blister. For example, if you missed one active white tablet, you should take 3 days of the placebo yellow tablets; if you missed 2 active white tablets, you should take 2 days of the placebo yellow tablets; and if you missed 3 active white tablets, you should take only 1 day of the placebo yellow tablets.
If you cannot remember how many white active tablets you have missed, follow the first option, use a barrier method such as a condom as an extra precaution until you have taken your tablets correctly for 7 days in a row and contact your doctor (as you may not have been protected from becoming pregnant).
If you have forgotten to take white active tablets in a strip and you do not have the expected monthly period while taking the yellow placebo tablets from the same strip, you may be pregnant. Consult your doctor before you start with the next strip.
Yellow tablets missed
The last 4 yellow tablets of the fourth row are placebo tablets which do not contain active substances. If you forgot to take one of these tablets the reliability of ZOELY is maintained. Throw away the yellow tablet that you missed and continue taking the next tablets at the usual time.
You want to delay your period
You can delay your period if you continue with the white active tablets in your next pack of ZOELY immediately after finishing the white tablets in your current pack. You can continue with this pack for as long as you wish, until this pack is empty. When you wish your period to begin, just stop tablet-taking. While using the second pack you may have some breakthrough bleeding or spotting on active tablet-taking days. Start your next pack after the usual 4 day inactive tablet interval.
You want to change the starting day of your period
If you take your tablets as directed, you will have your period on about the same day every 4 weeks. If you want to change this, just shorten (never lengthen) the next placebo tablet interval. For example, if your period usually starts on a Friday and in future you want it to start on Tuesday (3 days earlier) you should now start your next pack 3 days sooner than you usually do. If you make your placebo tablet interval very short (e.g. 3 days or less), you may not have a bleeding during the interval. You may have some breakthrough bleeding or spotting during the use of the white tablets in the next pack.
If you vomit or have diarrhoea
If you vomit within 3-4 hours of taking an active white tablet, or you have severe diarrhoea, the active ingredients of your ZOELY tablet may not have been completely absorbed into your body. The situation is similar to if you forget an active white tablet. After vomiting or diarrhoea, you must take another active white tablet from a reserve blister as soon as possible. If possible take it within 24 hours of when you normally take your pill. Take the next tablet at the usual time. If this is not possible or 24 or more hours have passed, you should follow the advice given under "What to do if… you forget to take your tablets". If you have severe diarrhoea, please contact your doctor.
The yellow tablets are placebo tablets which do not contain active substances. If you vomit or have severe diarrhoea within 3-4 hours of taking a yellow tablet, the reliability of ZOELY is maintained.
If you have unexpected bleeding
With all Pills, for the first few months, you can have breakthrough bleeding or spotting between your periods. You may need to use sanitary protection, but continue to take your tablets as normal. Breakthrough bleeding or spotting usually stops once your body clock has adjusted to the Pill (usually after about 3 tablet-taking cycles). Tell your doctor if it continues, becomes heavy or starts again.
If you have missed a period
If you have taken all of your tablets at the right time, and you have not vomited, or had severe diarrhoea or used other medicines then you are very unlikely to be pregnant. Continue to take ZOELY as usual.
If you miss your period twice in a row, you may be pregnant. Do not start the next pack of ZOELY until your doctor has checked you are not pregnant.
If you take too much (overdose)
There have been no reports of serious harmful effects from taking too many ZOELY tablets at one time. If you take too much ZOELY you may feel sick, vomit or have vaginal bleeding.
If you discover a child has taken ZOELY, ask your doctor for advice.
If you are not sure what to do, telephone your doctor or the Poisons Information Centre on 13 11 26 for advice.
Side Effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while taking ZOELY.
Like all medicines, ZOELY can have side effects. Sometimes they are serious, most of the time they are not.
Serious side effects
Serious reactions associated with the use of the Pill, as well as the related symptoms, are described in the following sections "The Pill and Thrombosis/The Pill and Cancer". Please read these sections for additional information and consult your doctor at once where appropriate.
Other possible side effects
The following side effects have been linked with the use of ZOELY.
Very common side effects (occurring in more than one per 10 users):
- acne
- changes in the menstrual cycle (e.g. absence of menstruation or breakthrough bleeding/spotting)
Common side effects (occurring in more than one per 100 users, but less than one in 10 users):
- decreased interest in sex;
- depression/depressed mood;
- mood changes;
- headache or migraine;
- feeling sick (nausea);
- breast pain;
- pelvic pain;
- weight gain.
Uncommon side effects (occurring in more than one per 1000 users, but less than one in 100 users):
- increased appetite;
- fluid retention (oedema);
- hot flush;
- swollen abdomen;
- increased sweating;
- hair loss;
- itching;
- dry skin;
- oily skin;
- sensation of heaviness;
- regular but scanty periods;
- larger breasts;
- breast lump;
- milk production while not pregnant;
- premenstrual syndrome;
- pain during intercourse;
- dryness in the vagina or vulva;
- spasm of the uterus;
- irritability;
- increase in the level of liver enzymes.
Rare side effects (occurring in more than one per 10,000 users but less than one per 1000 users):
- decreased appetite;
- increased interest in sex;
- disturbance in attention;
- dry eye; contact lens intolerance;
- dry mouth;
- golden brown pigment patches, mostly in the face;
- excessive hair growth;
- vaginal smell;
- discomfort in the vagina or vulva;
- hunger.
The following side effect has been reported during market use of Zoely: allergic (hypersensitivity) reactions.
You should stop taking ZOELY if you experience any signs of thrombosis (headache or pain elsewhere in your body, dizziness, fainting, disturbances in vision, swollen ankles), or jaundice (yellowing of the eyes or skin).
Tell your doctor or pharmacist if you notice any side effects not mentioned in this leaflet.
After taking ZOELY
If you want to stop taking ZOELY
You can stop taking ZOELY any time you want to.
If you do not want to get pregnant, use another reliable birth control method after stopping ZOELY. Ask your doctor for advice.
If you stop because you want to get pregnant, it is generally recommended that you wait until you have had a natural period before trying to conceive. This helps you to work out when the baby will be due.
Ask your doctor or pharmacist for advice about taking folate if you plan to become pregnant.
Storage
Store your tablets below 30°C in a dry place.
Do not store ZOELY or any other medicine in the bathroom or near a sink. Do not leave it in the car or on a window sill. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least 1.5 metres above the ground is a good place to store medicines.
Do not use after the expiry date stated on the blister and outer box.
Do not use the product if you notice, for example, colour change in the tablet, crumbling of the tablet or any other visible signs of deterioration.
Disposal
If your doctor tells you to stop using ZOELY or the tablets have passed their expiry date, ask your pharmacist what to do with any left over.
Product description
What it looks like
ZOELY is presented in 1 or 3 PVC/Aluminium backed blister strips containing 28 film-coated tablets (24 white active tablets and 4 yellow inactive tablets). The blister is packed in a carton together with the package leaflet. Each carton also contains a single paper sleeve, designed to carry one strip of tablets when removed from the outer carton.
The white, round active tablets are coded with 'ne' on both sides. The yellow, round inactive tablets are coded with 'p' on both sides.
Active Substance
In the white tablets:
- nomegestrol acetate 2.5 milligrams
- estradiol 1.5 milligrams
In the yellow tablets:
- no active ingredients
Other ingredients
In the white tablets:
- lactose monohydrate, microcrystalline cellulose, crospovidone, purified talc, magnesium stearate, colloidal anhydrous silica, polyvinyl alcohol, titanium dioxide, macrogol 3350.
In the yellow tablets:
- lactose monohydrate, microcrystalline cellulose, crospovidone, purified talc, magnesium stearate, colloidal anhydrous silica, poly vinyl alcohol, titanium dioxide, iron oxide yellow, iron oxide black, macrogol 3350.
Supplier
Theramex Australia Pty Ltd
Level 22, 60 Margaret Street,
Sydney NSW 2000
1800 THERAMEX or 1800 843 726
This leaflet was prepared in March 2021.
AUST R 168332
Published by MIMS May 2021

 The risk for venous thromboembolic complications in CHC users may increase substantially in a woman with additional risk factors, particularly if there are multiple risk factors (see Section 4.4 Special Warnings and Precautions for Use, Risk factors for VTE).
The risk for venous thromboembolic complications in CHC users may increase substantially in a woman with additional risk factors, particularly if there are multiple risk factors (see Section 4.4 Special Warnings and Precautions for Use, Risk factors for VTE). The adverse reaction rates for Zoely (N = 3490) in comparison to the reference COC containing drospirenone 3 mg and ethinylestradiol 30 microgram (21/7 regimen) (N = 1105) in the integrated safety data set were acne (15.4% versus 7.9%), weight increased (8.5% versus 5.9%) and abnormal withdrawal bleeding (predominantly absence of withdrawal bleeding) (10.2% versus 0.6%).
The adverse reaction rates for Zoely (N = 3490) in comparison to the reference COC containing drospirenone 3 mg and ethinylestradiol 30 microgram (21/7 regimen) (N = 1105) in the integrated safety data set were acne (15.4% versus 7.9%), weight increased (8.5% versus 5.9%) and abnormal withdrawal bleeding (predominantly absence of withdrawal bleeding) (10.2% versus 0.6%). Chemical name: 17α-acetoxy-6-methyl-19-norpregna-4,6-dien-3,20-dione.
Chemical name: 17α-acetoxy-6-methyl-19-norpregna-4,6-dien-3,20-dione. Chemical name: estra-1,3,5(10)-triene-3,17β-diol.
Chemical name: estra-1,3,5(10)-triene-3,17β-diol.