What is in this leaflet
This leaflet answers some of the common questions people ask about ZOLADEX 3.6 mg. It does not contain all the information that is known about ZOLADEX 3.6 mg.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor will have weighed the risks of you being administered ZOLADEX 3.6 mg against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What ZOLADEX is for
ZOLADEX 3.6 mg lowers the amount of sex hormones in the body. In women it reduces the level of oestrogen. In men it reduces the level of testosterone.
ZOLADEX 3.6 mg can treat breast cancer in some women before menopause or the 'change of life'. It is not a cure for breast cancer.
ZOLADEX 3.6 mg is also used to treat endometriosis and fibroids of the uterus. These are not cancer.
ZOLADEX 3.6 mg is used to thin the lining of the womb before surgery.
ZOLADEX 3.6 mg is also used in combination with other drugs as part of a treatment for infertility.
ZOLADEX 3.6 mg can treat prostate cancer in some men. It is not a cure for prostate cancer.
Your doctor will have explained why you are being treated with ZOLADEX 3.6 mg and told you what dose you will be given.
Follow all directions given to you by your doctor carefully. They may differ from the information contained in this leaflet.
Your doctor may prescribe this medicine for another use. Ask your doctor if you want more information.
ZOLADEX 3.6 mg is not addictive.
Before you are given ZOLADEX 3.6 mg
When you must not be given it
ZOLADEX 3.6 mg will not be given to children. There is no information on its use in children.
It will not be used after the use by (expiry) date printed on the pack. It may have no effect at all, or worse, an entirely unexpected effect if it is used after the expiry date.
ZOLADEX 3.6 mg will not be used if the packaging is torn or shows signs of tampering.
Before you start to use it
You must tell your doctor if:
- you have any allergies to any ingredients listed at the end of this leaflet or any other substances
If you have an allergic reaction, you may get a rash, hay fever, difficulty breathing or feel faint. - you are pregnant, intend to become pregnant or are breastfeeding.
Unless you are using ZOLADEX 3.6 mg as part of a fertility treatment do not use it while you are pregnant. It may affect the baby. We do not know if ZOLADEX 3.6 mg passes into breast milk. - you have heart or blood vessel conditions, including heart rhythm problems (arrhythmia), or are being treated with medicines for these conditions.
The risk of you having further heart rhythm problems may increase if you are taking ZOLADEX 3.6 mg.
Taking other medicines
Tell your doctor if you are taking any other medicines, including
- medicines that you buy at the chemist, supermarket or health food shop.
Your doctor or pharmacist can tell you what to do if you are taking any other medicines.
If you have not told your doctor about any of these things, tell them before you are administered ZOLADEX 3.6 mg.
Being given ZOLADEX 3.6 mg
How much you will be given
A ZOLADEX 3.6 mg implant will be injected under the skin of your stomach every 28 days.
The implant is a very small pellet that is given by a special needle and syringe known as SafeSystem. The injection will not hurt very much.
The pellet is designed to slowly release the medicine into your body over four weeks.
The ZOLADEX 3.6 mg SafeSystem incorporates a protective needle sleeve that automatically locks in place following administration of the implant to aid in the prevention of needle stick injury.
How long to use it
The length of time you will use ZOLADEX 3.6 mg for depends on what illness is being treated.
Your doctor will tell you how long you will need to be given ZOLADEX 3.6 mg.
If you forget to be given it
Your doctor will make an appointment for your next dose if you need it, so you will not forget your next dose.
Overdose
The doctor or nurse giving you ZOLADEX 3.6 mg will be experienced in the use of it, so it is unlikely that you will be given an overdose.
While you are using it
Things you must do
Make sure to keep your appointment for being given ZOLADEX 3.6 mg every four weeks. If you do not, your hormone level will rise and your illness may get worse.
Use barrier contraceptive methods (condom or diaphragm) while having ZOLADEX 3.6 mg treatment, unless it is being used for infertility. Tell your doctor immediately if you become pregnant.
Things you must not do
Do not take oral contraceptives while being treated with ZOLADEX 3.6 mg. They interfere with the way it works.
Do not stop being given ZOLADEX 3.6 mg unless you have discussed it with your doctor first. Your doctor will have explained why you need to be given ZOLADEX and for how long.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using ZOLADEX 3.6 mg.
ZOLADEX 3.6 mg helps most people with the illnesses it treats, but it may have unwanted side effects in a few people.
It can be hard to work out whether side effects are caused by ZOLADEX 3.6 mg or the disease that is being treated.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- skin rashes
- painful joints
- hot flushes, sweating or feeling faint
- chills
- tingling in fingers or toes
- swelling, soreness or itchiness of the breasts
- vaginal dryness
- headache
- mood changes
- trouble passing urine or experience lower back pain
- your testicles getting smaller
These are all mild side effects of ZOLADEX 3.6 mg.
ZOLADEX 3.6 mg lowers the amount of sex hormones in your body so your sex drive will probably be reduced.
It is unlikely that you can father a child or fall pregnant while using ZOLADEX 3.6 mg, unless it is for infertility treatment.
Use a barrier contraceptive method to make sure.
Most women stop having periods while being treated with ZOLADEX 3.6 mg. Some may go through menopause while being given ZOLADEX 3.6 mg and not have periods again when the treatment is finished.
In men and in women who have not gone through the 'change of life', ZOLADEX 3.6 mg is likely to reduce the amount of calcium in their bones leading to a loss in bone mineral density. It is known that bone mineral density loss partially recovers in the next several months following cessation of therapy. However, doctors do not know if bone mineral density loss will recover completely in every person. Discuss this with your doctor or pharmacist if you are concerned.
If you have a tumour in your pituitary gland, ZOLADEX 3.6 mg may make the tumour bleed or collapse. This is very rare but causes severe headaches, sickness, loss of eyesight and unconsciousness.
Your doctor will be happy to discuss these things with you if you want more information.
Tell your doctor if you notice anything else that is making you feel unwell. Some people may get other side effects while being treated with ZOLADEX 3.6 mg.
After using it
Storage
Keep your ZOLADEX 3.6 mg in the package until you take it to the doctor or nurse to give it to you. If you take ZOLADEX 3.6 mg out of the pack you may dislodge the pellet in the syringe.
It should be kept in a cool, dry place where the temperature stays below 25°C.
Do not store it or any other medicine in the bathroom or near a sink. Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Do not leave it in the car on hot days.
Disposal
Any ZOLADEX 3.6 mg, which is not used, will be disposed of in a safe manner by your doctor or pharmacist.
Product description
What ZOLADEX 3.6 mg looks like
ZOLADEX 3.6 mg comes in a special syringe, called the SAFESYSTEM and a siliconised needle.
The small pellet containing ZOLADEX 3.6 mg is about the size of a grain of rice. It can be seen in the "window" half way up the syringe and is white or cream coloured
Ingredients
ZOLADEX 3.6 mg implant contains the active ingredient goserelin 3.6 mg (as the acetate) plus polyglactin.
Distributor
AstraZeneca Pty Ltd
ABN 54 009 682 311
66 Talavera Road
MACQUARIE PARK NSW 2113
Telephone: 1800 805342
This leaflet was prepared on 16th May 2017.
Australian Registration Number AUST R 24368
Doc ID-002754320 v4.0
Published by MIMS September 2017




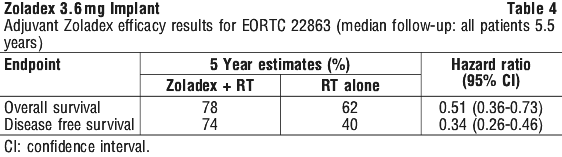





 Overall, the efficacy data demonstrates that Zoladex 3.6 mg is effective as an alternative to combination chemotherapy, in patients with hormone receptor-positive early breast cancer (as demonstrated by the ZEBRA trial). Zoladex may also provide additional therapeutic benefit when used after combination chemotherapy (as demonstrated by study INT0101). The clinical evaluation of Zoladex is mainly in studies of two years duration when it has been shown to be equivalent to chemotherapy. Studies up to five years have shown the efficacy and safety of Zoladex. Treatment for greater than five years has not been studied. Both efficacy and tolerability should be considered when deciding on the treatment duration for an individual patient.
Overall, the efficacy data demonstrates that Zoladex 3.6 mg is effective as an alternative to combination chemotherapy, in patients with hormone receptor-positive early breast cancer (as demonstrated by the ZEBRA trial). Zoladex may also provide additional therapeutic benefit when used after combination chemotherapy (as demonstrated by study INT0101). The clinical evaluation of Zoladex is mainly in studies of two years duration when it has been shown to be equivalent to chemotherapy. Studies up to five years have shown the efficacy and safety of Zoladex. Treatment for greater than five years has not been studied. Both efficacy and tolerability should be considered when deciding on the treatment duration for an individual patient. Molecular formula: C59H84N18O14 (base).
Molecular formula: C59H84N18O14 (base).