Notes
Distributed by Stada Pharmaceuticals Australia Pty Ltd
1 Name of Medicine
Zolpidem tartrate.
2 Qualitative and Quantitative Composition
Each metered actuation (one spray) of ZolpiMist delivers 5 mg of zolpidem tartrate in 100 microL. Two actuations deliver 10 mg of zolpidem tartrate in 200 microL.
Excipient with known effect.
Benzoates.3 Pharmaceutical Form
ZolpiMist is an oromucosal spray which is a clear, colourless to yellowish, cherry flavoured solution designed to be sprayed directly into the mouth over the tongue.
4.1 Therapeutic Indications
ZolpiMist is indicated for the short-term treatment of insomnia in adults (see Section 4.2 Dose and Method of Administration).
4.2 Dose and Method of Administration
ZolpiMist acts rapidly and should therefore be taken immediately before retiring, or in bed, with at least 7-8 hours remaining before the planned time of awakening. For faster sleep onset, as with all zolpidem products, ZolpiMist should not be administered with or immediately after a meal (see Section 5.2 Pharmacokinetic Properties).
ZolpiMist should be taken in a single intake and not be re-administered during the same night. As with all hypnotics, long term use of zolpidem is not recommended. Treatment should be as short as possible and should not exceed four weeks.
For oral use only. ZolpiMist is designed to be sprayed directly into the mouth over the tongue.
Discontinuation of treatment.
See Section 4.8 Adverse Effects (Undesirable Effects).
Withdrawal effects.
See Section 4.4 Special Warnings and Precautions for Use.
Recommended dosage.
Adults.
5 mg (one actuation) for women and 5 mg (one actuation) or 10 mg (two actuations) for men to be taken at night. The lowest effective daily dose of ZolpiMist should be used and must not exceed 5 mg in women and 10 mg in men (see Section 5.2 Pharmacokinetic Properties).
Elderly or debilitated patients.
Since elderly or debilitated patients may be especially sensitive to the effects of zolpidem, 5 mg (one actuation) to be taken at night is recommended. This dose should not be exceeded.
Hepatic impairment.
As clearance and metabolism of zolpidem is reduced in hepatic impairment, dosage of 5 mg to be taken at night is recommended, with particular caution being exercised in elderly patients.
In adults, less than 65 years, the dosage may be increased if the clinical response is inadequate, and the drug is well tolerated.
Renal impairment.
No dosage adjustment is necessary in these patients, although they should be closely monitored.
Children.
The use of ZolpiMist in children under 18 years is contraindicated.
ZolpiMist is packaged in a child-resistant container. For detailed instructions on how to use ZolpiMist, refer to the Consumer Medicine Information (CMI). ZolpiMist must be primed before it is used for the first time. To prime, patients should be told to point the black spray opening away from their face and other people and spray 7 times. For administration, the child-resistant container should be held upright with the black spray opening pointed directly into the mouth. The patient should fully press down on the pump to make sure a full dose (5 mg) of ZolpiMist is sprayed directly into the mouth over the tongue. If a 10 mg dose is prescribed, a second spray should be administered. If the patient does not use ZolpiMist for at least 14 days, it must be primed again with 1 spray.4.3 Contraindications
Sleep apnoea.
Known hypersensitivity to zolpidem or other ingredients in the tablet.
Myasthenia gravis.
Severe hepatic insufficiency.
Acute and/or severe pulmonary insufficiency.
Prior or concomitant intake with alcohol.
ZolpiMist should not be prescribed for children.
Patients who have previously experienced complex sleep behaviours after taking zolpidem.
4.4 Special Warnings and Precautions for Use
The cause of insomnia should be identified wherever possible and the underlying factors treated before a hypnotic is prescribed.
The failure of insomnia to remit after a 7-14 day course of treatment may indicate the presence of a primary psychiatric or physical disorder, and the patient should be carefully reevaluated at regular intervals.
Withdrawal, rebound, dependence and tolerance.
Tolerance.
Continuous long-term use of ZolpiMist is not recommended and should not exceed four weeks.
Some loss of efficacy to the hypnotic effects of sedative/hypnotic agents may develop after repeated use for a few weeks.
Dependence.
Use of zolpidem may lead to the development of abuse and/or physical and psychological dependence. The risk of dependence increases with dose and duration of treatment; it is also greater in patients with a history of psychiatric disorders and/or alcohol or drug abuse. ZolpiMist should be used with extreme caution in patients with current or a history of alcohol or drug abuse. These patients should be under careful surveillance when receiving hypnotics.
Once physical dependence has developed, abrupt termination of treatment will be accompanied by withdrawal symptoms. These may consist of headaches or muscle pain, extreme anxiety and tension, restlessness, confusion and irritability. In severe cases the following symptoms may occur: derealisation, depersonalisation, hyperacusis, numbness and tingling of the extremities, hypersensitivity to light, noise and physical contact, hallucinations, delirium, or epileptic seizures.
Rebound insomnia.
A transient syndrome whereby the symptoms that led to treatment with sedative/hypnotic agents recur in an enhanced form may occur on withdrawal of hypnotic treatment. It may be accompanied by other reactions including mood changes, anxiety and restlessness.
It is important that the patient should be aware of the possibility of rebound phenomena, thereby minimising anxiety over such symptoms should they occur when the medicinal product is discontinued.
There are indications that, in the case of sedative/hypnotic agents with a short duration of action, withdrawal phenomena can manifest within the dosage interval, especially when the dosage is high.
When ZolpiMist is used in accordance with the recommendations for dosage, duration of treatment and warnings, the risk of withdrawal symptoms or rebound phenomena occurring is minimal.
Severe injuries.
Due to its pharmacological properties, zolpidem can cause drowsiness and a decreased level of consciousness, which may lead to falls and consequently to severe injuries.
Patients with long QT syndrome.
An in vitro cardiac electrophysiological test showed that under experimental conditions, using very high concentration and pluripotent stem cells, zolpidem may reduce the hERG related potassium currents. As a precaution, the benefit/risk ratio of zolpidem treatment in patients with known congenital long QT syndrome should be carefully considered.
Chemical submission (drug facilitated illicit use for criminal intent).
The rapid onset of sedation, coupled with the amnestic features of Zolpimist, particularly when combined with alcohol, administered without knowledge of the victim, has proven to induce incapacitation and thus facilitate criminal actions (which could be dangerous). Healthcare Providers should prescribe Zolpimist according to their clinical evaluation and only in case of medical need as it may be used illicitly for chemical submission.
CNS effects.
As with all patients taking CNS-depressant medications, patients receiving ZolpiMist should be warned not to operate dangerous machinery or motor vehicles until it is known that they do not become drowsy or dizzy from ZolpiMist therapy. Patients should be advised that their tolerance for other CNS depressants will be diminished and that these medications should either be eliminated or given in reduced dosage in the presence of ZolpiMist. Prior or concomitant intake with alcohol is contraindicated (see Section 4.3 Contraindications).
Respiratory function.
Both animal and human pharmacology studies performed with zolpidem have not observed any effect on the respiratory centre. However, as other sedative/hypnotics have the capacity to depress respiratory drive, caution is advised when ZolpiMist is administered to patients with respiratory insufficiency (see Section 4.3 Contraindications).
Use in hepatic impairment.
As clearance and metabolism of zolpidem is reduced in hepatic impairment, dosage should begin at 5 mg with particular caution being exercised in elderly patients. In adults (under 65 years) dosage may be increased to 10 mg only where the clinical response is inadequate and the drug is well tolerated. (see Section 4.4 Special Warnings and Precautions for Use, Use in the elderly or debilitated patient; Section 4.2 Dose and Method of Administration).
Zolpidem must not be used in patients with severe hepatic impairment as it may contribute to encephalopathy.
Use in renal impairment.
Dosage reduction is not necessary in patients with renal impairment, however, as a general precaution, these patients should be monitored closely (see Section 4.2 Dose and Method of Administration).
Use in the elderly or debilitated patient.
Elderly and debilitated patients may be particularly sensitive to the effects of ZolpiMist, therefore a 5 mg dose is recommended. This dose should not be exceeded in these patients. (See Section 4.2 Dose and Method of Administration).
Such patients may be particularly susceptible to the sedative effects of the medication and associated giddiness, ataxias and confusion, which may increase the possibility of a fall.
Memory impairment.
Sedative/hypnotic agents may induce anterograde amnesia. The condition occurs most often several hours after ingesting the product and therefore to reduce the risk patients should ensure that they will be able to have an uninterrupted sleep of 7-8 hours.
Suicidality, depression, psychosis and schizophrenia.
Several epidemiological studies show an increased incidence of suicide and suicide attempt in patients with or without depression, treated with benzodiazepines and other hypnotics, including zolpidem. ZolpiMist should be administered with caution in patients exhibiting symptoms of depression. ZolpiMist is not recommended as primary therapy in patients with psychotic illness, including depression and psychosis. In such conditions, psychiatric assessment and supervision are necessary as depression may increase in some patients and may contribute to deterioration in severely disturbed schizophrenics with confusion and withdrawal. Pre-existing depression may be unmasked during the use of ZolpiMist. Suicidal tendencies may be present or uncovered and protective measures may be required. Intentional overdosage is more common in this group of patients: therefore, the least amount of drug that is feasible should be prescribed for the patient at any one time.
Other psychiatric and paradoxical reactions.
Other psychiatric and paradoxical reactions such as acute rage, restlessness, insomnia exacerbated, agitation, irritability, aggression, delusions, anger, nightmares, hallucinations, stimulation or excitement, abnormal behaviour, delirium, and other adverse behavioural effects are known to occur when using sedative/hypnotic agents like zolpidem. Should such reactions occur, ZolpiMist should be discontinued. These reactions are more likely to occur in the elderly.
Somnambulism and associated behaviours.
Complex sleep behaviours, including sleeping-walking, sleep-driving, and engaging in other activities while not fully awake, may occur following the first or any subsequent use of ZolpiMist. Patients can be seriously injured or injure others during complex sleep behaviours. Such injuries may result in fatal outcome. Other complex sleep behaviours (e.g. preparing and eating food, making phone calls, or having sex) have also been reported. Patients usually do not remember these events. Postmarketing reports have shown that complex sleep behaviours may occur with products containing zolpidem alone at recommended doses, with or without the concomitant use of alcohol or other central nervous system (CNS) depressants (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). Discontinue ZolpiMist immediately if a patient experiences a complex sleep behaviour (see Section 4.3 Contraindications). The use of alcohol and other CNS depressants with zolpidem appears to increase the risk of such behaviours, as does the use of ZolpiMist at doses exceeding the maximum recommended dose (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions; Section 4.8 Adverse Effects (Undesirable Effects)). These events can occur in sedative-hypnotic naive as well as sedative-hypnotic experienced patients.
Psychomotor impairment.
Zolpidem has CNS-depressant effects. The risk of psychomotor impairment, including impaired driving ability, is increased if zolpidem is taken within less than 7-8 hours before performing activities that require mental alertness, a dose higher than the recommended dose is taken, or zolpidem is co-administered with other CNS depressants, alcohol, or with other drugs that increase the blood levels of zolpidem.
Interactions with alcohol.
Prior or concomitant intake with alcohol is contraindicated (see Section 4.3 Contraindications). Patients should be advised that their tolerance for alcohol and other CNS depressants might be reduced and have an additive effect on psychomotor performance (see Section 4.4 Special Warnings and Precautions for Use, Somnambulism and associated behaviours above).
Risks from concomitant use with opioids.
Concomitant use of sedative-hypnotic drugs, including zolpidem, with opioids may result in sedation, respiratory depression, coma and death. Because of these risks, reserve concomitant prescribing of opioids and zolpidem for use in patients for whom alternative treatment options are inadequate.
If a decision is made to prescribe zolpidem concomitantly with opioids, prescribe the lowest effective dosages and minimum durations of concomitant use, and follow patients closely for signs and symptoms of respiratory depression and sedation. (See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Severe anaphylactic and anaphylactoid reactions.
Rare cases of angioedema involving the tongue, glottis or larynx have been reported in patients after taking the first or subsequent doses of sedative-hypnotics, including zolpidem. Some patients have had additional symptoms such as dyspnoea, throat closing, or nausea and vomiting that suggest anaphylaxis. Some patients have required medical therapy in the emergency department. If angioedema involves the tongue, glottis or larynx, airway obstruction may occur and be fatal. Patients who develop angioedema after treatment with zolpidem should not be rechallenged with the drug.
Epilepsy.
Abrupt withdrawal of CNS-depressant drugs in persons with convulsive disorders has been associated with a temporary increase in the frequency and or severity of seizures.
As with other sedative/hypnotics, caution is advised when ZolpiMist is used in these patients.
Abuse.
Caution must be exercised in administering ZolpiMist to individuals known to be addiction prone or those whose history suggests they may increase the dosage on their own initiative. It is desirable to limit repeat prescription without adequate medical supervision.
Paediatric use.
See Section 4.2 Dose and Method of Administration.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
CNS depressants.
Co-administration of ZolpiMist with other CNS depressants should be exercised with caution since the central depressant effect may be additive. CNS depressants include alcohol, benzodiazepines, barbiturates, sedative/hypnotics, anxiolytics, antidepressant agents (including tricyclic antidepressants, MAOIs), antipsychotics (neuroleptics), phenothiazines, skeletal muscle relaxants, antihistamines, neuroleptics, antiepileptic drugs, narcotic analgesics or anaesthetics. Concomitant use of zolpidem with these drugs may increase drowsiness and psychomotor impairment, including impaired driving ability. In the case of narcotic analgesics enhancement of euphoria may also occur.
Opioids.
The concomitant use of sedative-hypnotic drugs, including zolpidem, and opioids increases the risk of sedation, respiratory depression, coma and death because of additive CNS depressant effect. Limit dosage and duration of concomitant use of zolpidem and opioids (see Section 4.4 Special Warnings and Precautions for Use, Risks from concomitant use with opioids).
Alcohol.
Prior or concomitant intake with alcohol is contraindicated (see Section 4.3 Contraindications). Patients should be advised that their tolerance for alcohol and other CNS depressants might be reduced and have an additive effect on psychomotor performance. The use of alcohol and other CNS depressants with zolpidem appears to increase the risk of somnambulism and associated behaviours (see Section 4.4 Special Warnings and Precautions for Use, Somnambulism and associated behaviours).
Imipramine.
The sedative effects of imipramine 75 mg and zolpidem 20 mg were shown to be additive when the two compounds were given concomitantly in healthy volunteers. No pharmacokinetic interaction was shown between zolpidem and imipramine or its metabolite, desipramine.
Chlorpromazine.
The combination of zolpidem 10 mg and chlorpromazine 50 mg in healthy volunteers produced an addition of effects seen in psychometric tests and decreased alertness and psychomotor performance. No pharmacokinetic interaction was observed.
Haloperidol.
No evidence of pharmacokinetic interaction between zolpidem 20 mg and haloperidol 2 mg was seen when they were given concurrently to healthy volunteers.
Caffeine.
No change in the sleep inducing effect of zolpidem was seen when 300 mg caffeine was given in the evening 45 minutes before administration of zolpidem 20 mg to 8 healthy volunteers.
Warfarin.
Prothrombin times were not prolonged in healthy adults when zolpidem 20 mg was administered for 4 consecutive nights concomitantly with warfarin. Warfarin had been given for at least 10 days previously to produce a 1.5 times prolongation of baseline prothrombin time in the volunteers. Zolpidem does not appear to modify the anticoagulant activity of warfarin.
Digoxin.
The concurrent administration of zolpidem 10 mg once daily and digoxin 0.25 mg in healthy volunteers did not show any alteration of the pharmacokinetic or pharmacodynamic profile of digoxin.
H2 - antagonists.
Simultaneous administration of zolpidem 20 mg and cimetidine 200 mg tds and 400 mg at night or ranitidine 150 mg bd did not cause any significant change in psychometric tests from those produced by zolpidem alone. No change in the pharmacokinetics of zolpidem were caused by concomitant administration of either cimetidine or ranitidine.
Hepatic enzyme inhibitors and inducers.
Zolpidem is metabolized via several hepatic cytochrome P450 enzymes: the main enzyme being CYP3A4 with the contribution of CYP1A2. Compounds which inhibit or enhance certain hepatic enzymes (particularly cytochrome P450) may increase or decrease the activity of some hypnotics like zolpidem. The pharmacodynamic effect of zolpidem is decreased when it is administered with a CYP3A4 inducer such as rifampicin and St John's Wort. Coadministration of St John's Wort may decrease blood levels of zolpidem, therefore concurrent use is not recommended.
Ketoconazole has a significant but only quantitatively modest reduction in zolpidem clearance, with an increase in its pharmacodynamic effects. Patients should be advised that use of zolpidem with ketoconazole may enhance the sedative effects of zolpidem. However, when zolpidem is administered with itraconazole (a CYP3A4 inhibitor) its pharmacokinetics and pharmacodynamics were not significantly modified. The clinical relevance of these results is unknown.
Fluvoxamine is a strong inhibitor of CYP1A2 and a moderate to weak inhibitor of CYP2C9 and CYP3A4. Co-administration of fluvoxamine may increase blood levels of zolpidem, concurrent use is not recommended.
Ciprofloxacin has been shown to be a moderate inhibitor of CYP1A2 and CYP3A4.
Coadministration of ciprofloxacin may increase blood levels of zolpidem, concurrent use is not recommended.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category B3)
This drug has been taken by only a limited number of pregnant women and women of childbearing age, without an increase in the frequency of malformation or other direct or indirect harmful effects on the human foetus having been observed. Studies in animals have shown evidence of an increased occurrence of foetal damage, the significance of which is uncertain in humans. The use of zolpidem is not recommended during pregnancy.
Cases of reduced fetal movement and fetal heart rate variability have been described after administration of benzodiazepines or other sedative-hypnotic drugs such as zolpidem during pregnancy.
Administration of zolpidem during the late phase of pregnancy or during labor has been associated with effects on the neonate, such as hypothermia, hypotonia, feeding difficulties, and respiratory depression.
If zolpidem is prescribed to a woman of childbearing potential, she should be warned to contact her physician about stopping the product if she intends to become or suspects that she is pregnant.
Teratogenic effects.
In reproductive toxicity studies, rats treated with oral zolpidem with estimated exposures (AUC) to zolpidem and its major metabolite of 41 and 15 times, respectively, the anticipated clinical exposure did not exhibit teratogenic effects, but post-implantation survival index and postpartum viability of the offspring were significantly reduced. In rats, delayed ossification of foetal skull bones occurred at zolpidem and metabolite exposure levels of 8 and 3 times, respectively, the anticipated clinical exposure.
Rabbits treated with oral zolpidem with estimated exposure to zolpidem of 0.6-2.6 times the anticipated clinical exposure did not exhibit teratogenic effects, but there was increased postimplantation loss.
Although animal studies have not shown any teratogenic effects with zolpidem, the safety of zolpidem in human pregnancy has not been established.
Non teratogenic effects.
Cases of severe neonatal respiratory depression have been reported when zolpidem was used with other CNS depressants at the end of pregnancy.
Infants born to mothers who took hypnotics chronically during the latter stages of pregnancy may have developed physical dependence and may be at some risk for developing withdrawal symptoms in the postnatal period. Appropriate monitoring of the newborn in the postnatal period is recommended.
The use of ZolpiMist in nursing women is not recommended as small quantities of zolpidem are excreted into breast milk.4.7 Effects on Ability to Drive and Use Machines
This preparation is to aid sleep. Patients should not drive or operate machinery for 8 hours after taking ZolpiMist.
Adverse effects including drowsiness, prolonged reaction time, dizziness, sleepiness, blurred/double vision, reduced alertness and/or impaired driving may continue the following day. In order to minimise this risk a full night of sleep (7-8 hours) is recommended. After ingesting the medicine, patients should be cautioned against engaging in hazardous occupations requiring complete mental alertness or motor co-ordination such as operating machinery or driving a motor vehicle, including potential impairment of the performance of such activities that may occur the day following ingestion of ZolpiMist. Furthermore, the coadministration of zolpidem with alcohol and other CNS depressants increases the risk of such effects. Patients should be warned not to use alcohol or other psychoactive substances when taking zolpidem.
4.8 Adverse Effects (Undesirable Effects)
Clinical trials data.
There is evidence of a dose-relationship for adverse effects associated with zolpidem use, particularly for certain CNS events. These occur most frequently in elderly patients.
Associated with discontinuation of treatment. Approximately 4% of 1,701 patients who received zolpidem at all doses (1.25 to 90 mg) in US premarketing clinical trials discontinued treatment because of an adverse clinical event. Events most commonly associated with discontinuation from US trials were daytime drowsiness (0.5%), dizziness (0.4%), headache (0.5%), nausea (0.6%), and vomiting (0.5%).
Approximately 6% of 1,320 patients who received zolpidem at all doses (5 to 50 mg) in similar European trials discontinued treatment because of an adverse event. Events most commonly associated with discontinuation from these trials were daytime drowsiness (1.6%), amnesia (0.6%), dizziness (0.6%), headache (0.6%) and nausea (0.6%).
Incidence in controlled clinical trials. Most commonly observed adverse events in controlled trials.
During short-term treatment (up to 10 nights) with zolpidem at doses up to 10 mg, the most commonly observed adverse events associated with the use of zolpidem and seen at statistically significant differences from placebo-treated patients were drowsiness (reported by 2% of zolpidem patients), dizziness (1%), and diarrhoea (1%). During longer-term treatment (28 to 35 nights) with zolpidem at doses up to 10 mg, the most commonly observed adverse events associated with the use of zolpidem and seen at statistically significant differences from placebo-treated patients were dizziness (5%) and drugged feelings (3%).
Adverse events observed at an incidence of ≥ 1% in controlled trials.
Tables 1 and 2 enumerate treatment-emergent adverse event frequencies that were observed at an incidence equal to 1% or greater among patients with insomnia who received zolpidem in US placebo controlled trials. Events reported by investigators were classified utilising a modified World Health Organisation (WHO) dictionary of preferred terms for the purpose of establishing event frequencies.
Table 1 was derived from a pool of 11 placebo-controlled short-term US efficacy trials involving zolpidem in doses ranging from 1.25 to 20 mg. The table is limited to data from doses up to and including 10 mg, the highest dose recommended for use.
Oral tissue-related adverse reactions in ZolpiMist pharmacokinetics studies.
The effect of chronic daily administrations of ZolpiMist on oral tissue has not been evaluated. In pharmacokinetic studies conducted with ZolpiMist in healthy subjects, an oral soft tissue exam was performed and no signs of oral irritation were noted following administration of single doses of ZolpiMist.
 Table 2 was derived from a pool of three placebo-controlled long-term efficacy trials involving zolpidem. These trials involved patients with chronic insomnia who were treated for 28 to 35 nights with zolpidem at doses of 5, 10 or 15 mg. The table is limited to data from doses up to and including 10 mg, the highest dose recommended for use. The table includes only adverse events occurring at an incidence of at least 1% for zolpidem patients.
Table 2 was derived from a pool of three placebo-controlled long-term efficacy trials involving zolpidem. These trials involved patients with chronic insomnia who were treated for 28 to 35 nights with zolpidem at doses of 5, 10 or 15 mg. The table is limited to data from doses up to and including 10 mg, the highest dose recommended for use. The table includes only adverse events occurring at an incidence of at least 1% for zolpidem patients.

Postmarketing data.
See Table 3.

Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Signs and symptoms.
In reports of overdose with zolpidem alone or with other CNS depressant agents (including alcohol), impairment of consciousness has ranged from somnolence to coma and more severe symptomology, including fatal outcomes have been reported. Fatalities have occurred when overdoses of multi CNS depressants were taken.
Recommended treatment.
General symptomatic and supportive measures should be used, along with immediate gastric lavage where appropriate. Intravenous fluids should be administered as needed. Sedative drugs should be withheld, even if excitation occurs.
Zolpidem has been shown in trials to be non-dialysable.
Use of flumazenil may be considered when serious symptoms are observed. However, flumazenil administration may contribute to the appearance of neurological symptoms, such as convulsions, since zolpidem does not exhibit the anticonvulsant effects of benzodiazepines.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Zolpidem belongs to the imidazopyridine group of compounds and is structurally unrelated to other hypnotic agents. Zolpidem selectively binds the omega-1 receptor subtype (also known as the benzodiazepine-1 subtype) which is the alpha unit of the GABA-A receptor complex. Whereas benzodiazepines non-selectively bind all three omega receptor subtypes, zolpidem preferentially binds the omega-1 subtype. The modulation of the chloride anion channel via this receptor leads to the specific sedative effects demonstrated by zolpidem i.e. the preservation of deep sleep (stage 3 and 4 slow wave sleep).
These effects are reversed by the benzodiazepine antagonist flumazenil.
In animals.
The selective binding of zolpidem to omega-1 receptors may explain the virtual absence at hypnotic doses of myorelaxant and anti-convulsant effects in animals which are normally exhibited by benzodiazepines which are not selective for omega-1 sites.
In humans.
The preservation of deep sleep (stages 3 and 4 - slow-wave sleep) may be explained by the selective omega-1 binding by zolpidem. All identified effects of zolpidem are reversed by the benzodiazepine antagonist flumazenil.
Clinical trials.
Insomnia in non-elderly adults. Short term (1 to 2 nights) placebo controlled studies in 620 volunteers showed that zolpidem 2.5 to 10 mg decreased the latency of persistent sleep in a dose dependent manner. No further increase in efficacy was seen in doses up to zolpidem 40 mg.
The efficacy of zolpidem 2.5 - 20 mg was investigated in 11 placebo-controlled studies in 1606 (513 received zolpidem 10 mg) non-elderly insomniacs over a period of 2-35 nights. Zolpidem 10 mg was superior to placebo using both objective (polysomnography) and subjective methods of assessment. Zolpidem 20 mg showed little increase in efficacy.
Insomnia in the elderly. Four studies in 145 elderly (> 65 years) patients showed, using objective (2 studies) and subjective (4 studies) methods of assessment that zolpidem 5 mg was the dose giving the optimum efficacy/safety ratio.
Next day residual effects.
There was no evidence of residual next-day effects seen with zolpidem in several studies utilising the Multiple Sleep Latency Test (MSLT), the Digit Symbol Substitution Test (DSST), and patient ratings of alertness. In one study involving elderly patients, there was a small but statistically significant decrease in one measure of performance, the DSST, but no impairment was seen in the MSLT in this study.
Rebound effects.
Although there were no studies to exclude this effect, there was no objective (polysomnographic) evidence of rebound insomnia at recommended doses seen in studies evaluating sleep on the nights following discontinuation of zolpidem. There was subjective evidence of impaired sleep in the elderly on the first post-treatment night at doses above the recommended elderly dose of 5 mg.
Memory impairment.
Two small studies (n=6 and n=9) using objective measures of memory yielded little evidence for memory impairment following the administration of zolpidem. There was subjective evidence from adverse event data for anterograde amnesia occurring in association with the administration of zolpidem, predominantly at doses above 10 mg.
Effects on sleep stages.
In studies that measured the percentage of sleep time spent in each sleep stage, zolpidem has generally been shown to preserve sleep stages. Sleep time spent in stages 3 and 4 (deep sleep) was found comparable to placebo with only inconsistent, minor changes in REM (paradoxical) sleep at the recommended dose.
The hypnotic efficacy and safety of zolpidem has not been assessed in children and pregnant women.
5.2 Pharmacokinetic Properties
ZolpiMist (zolpidem tartrate) Oromucosal Spray is bioequivalent to Stilnox tablets (SanofiAventis). The pharmacokinetic profile of ZolpiMist is characterised by rapid absorption from the oral mucosa and gastrointestinal tract, and a short t1/2 in healthy subjects.
In a single-dose crossover study in 10 healthy young (18-40 years of age) male subjects administered 2.5, 5, and 10 mg doses of an early formulation of ZolpiMist*, the results demonstrated a linear relationship to dose for mean Cmax and AUC0-∞ over the range of doses administered in the study.
*ZolpiMist administered in this study used an older formulation containing less propylene glycol (amongst other minor differences) compared to the commercial formulation.
In a single-dose crossover study in 43 healthy young (18-45 years of age) subjects administered 5 and 10 mg ZolpiMist, the means for Cmax were 114 (range: 19 to 197) and 210 nanogram/mL (range: 77 to 401), respectively, occurring at a mean Tmax of approximately 0.9 hours for both. The mean zolpidem t1/2 was 2.7 (range: 1.7 to 5.0) and 3.0 hours (range: 1.7 to 8.4), for 5 and 10 mg ZolpiMist, respectively. In the same study, the means for Cmax were 123 (range: 53 to 221) and 219 nanogram/mL (range: 101 to 446) for 5 and 10 mg Stilnox tablets, respectively, occurring at a mean Tmax of 0.9 and 1.0 hours, respectively. The mean zolpidem t1/2 was 2.8 (range: 1.5 to 6.0) and 3.1 hours (range: 1.1 to 8.6) for the 5 and 10 mg Stilnox tablets, respectively.
A food-effect crossover study in 14 healthy young (18-45 years of age) male subjects compared the pharmacokinetics of ZolpiMist 10 mg when administered while fasting at least 8 hours or 5 minutes after eating a standard high-fat meal. Results demonstrated that with food, mean AUC0-∞ and Cmax were decreased by 27% and 58%, respectively, while mean Tmax was prolonged by 225% (from 0.8 to 2.6 hours). These results suggest that, for faster sleep onset, as with all zolpidem products, ZolpiMist should not be administered with or immediately after a meal.
Absorption.
Zolpidem has both a rapid absorption and onset of hypnotic action. Peak plasma concentration is reached at between 0.5 and 3 hours. Following oral administration, bioavailability is 70% due to a moderate first-pass metabolism.
The elimination half-life is short, with a mean value of 2.4 hours (± 0.2 h) and a duration of action of up to 6 hours. Zolpidem pharmacokinetic profile is linear in the therapeutic dose range, and is not modified upon repeated administration.
Distribution.
Protein binding amounts to approximately 90%. The distribution volume in adults is 0.54 ± 0.02 L/kg and decreases to 0.34 ± 0.05 L/kg in the very elderly.
Metabolism.
The main cytochrome P450 enzyme involved in the hepatic biotransformation of zolpidem is CYP3A4. CYP1A2 and CYP2D6 contribute minimally to the metabolism of zolpidem (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Excretion.
All metabolites are pharmacologically inactive and are eliminated in the urine (56%) and in the faeces (37%). Furthermore, they do not interfere with zolpidem plasma binding.
Effect of food.
A food-effect study in 30 healthy male volunteers compared the pharmacokinetics of oral zolpidem 10 mg when administered while fasting or 20 minutes after a meal. Results demonstrated that with food, mean AUC and Cmax were decreased by 15% and 25%, respectively, while mean Tmax was prolonged by 60% (from 1.4 to 2.2 hr). The half-life remained unchanged. These results suggest that, for faster sleep onset, ZolpiMist should not be administered with or immediately after a meal.
Special populations.
Zolpidem did not accumulate in young adults following nightly dosing with 20 mg zolpidem tartrate tablets for 2 weeks. In the elderly, the recommended dose for ZolpiMist is 5 mg (see Section 4.2 Dose and Method of Administration; Section 4.4 Special Warnings and Precautions for Use). This recommendation is based on several studies in which the mean Cmax, t1/2 and AUC were significantly increased when compared to results in young adults. In a pharmacokinetic study of 24 elderly (≥ 65 years of age) subjects administered 5 mg ZolpiMist, the means for Cmax and AUC were 134 nanogram/mL and 493 nanogram.hr/mL respectively, following administration of a single 5 mg oral dose of ZolpiMist.
Zolpidem did not accumulate in elderly subjects following nightly oral dosing of 10 mg for 1 week.
The pharmacokinetics of zolpidem in eight patients with chronic hepatic insufficiency were compared to results in healthy subjects. Following a single 20 mg oral zolpidem dose, mean Cmax and AUC were found to be two times (250 vs 499 nanogram/mL) and five times (788 vs 4,203 nanogram.hr/mL) higher, respectively, in hepatically compromised patients. Tmax did not change. The mean half-life in cirrhotic patients of 9.9 hr (range: 4.1 to 25.8 hr) was greater than that observed in normals of 2.2 hr (range: 1.6 to 2.4 hr). Dosing should be modified accordingly in patients with hepatic insufficiency (see Section 4.2 Dose and Method of Administration; Section 4.4 Special Warnings and Precautions for Use).
In patients with renal insufficiency, whether dialysed or not, there is a moderate reduction in clearance. The other pharmacokinetic parameters are unaffected. Zolpidem has been shown to be non-dialysable.
Gender difference in pharmacokinetics.
Women clear zolpidem tartrate from the body at a lower rate than men. Cmax and AUC parameters of zolpidem were approximately 45% higher at the same dose in female subjects compared with male subjects. Given the higher blood levels of zolpidem tartrate in women compared to men at a given dose, the recommended dose of ZolpiMist for adult women is 5 mg, and the recommended dose for adult men is 5 or 10 mg (see Section 4.2 Dose and Method of Administration).
In geriatric patients, clearance of zolpidem is similar in men and women. The recommended dose of ZolpiMist in geriatric patients is 5 mg regardless of gender (see Section 4.2 Dose and Method of Administration).
5.3 Preclinical Safety Data
Genotoxicity.
Zolpidem was not genotoxic in assays for gene mutations (Salmonella typhimurium histidine reversion assay, L5178Y mouse lymphoma assay), for chromosomal aberrations (human lymphocytes, mouse micronucleus assay) and for DNA repair assays (in human fibroblasts and rat hepatocytes). The mutagenic activity of zolpidem and/or its metabolites was equivocal in a Chinese hamster V79/HRPT gene mutation assay in the presence of metabolic activation.
Carcinogenicity.
Two year dietary carcinogenicity studies on zolpidem were conducted in rats and mice. No evidence of carcinogenic potential was observed in mice at plasma concentrations (AUC) of zolpidem and its major human metabolite of about 2 and 7-12 times, respectively, the anticipated clinical exposure at the maximum recommended clinical dose. An increased incidence of renal liposarcomas was observed in male rats (6% cf. 0 in controls) at plasma concentrations (AUC) of zolpidem and its major metabolite of at least 22 and 9 times, respectively, the anticipated human exposure.6 Pharmaceutical Particulars
6.1 List of Excipients
ZolpiMist includes the following inactive ingredients: benzoic acid, Cherry Flavor Art 825.382 (ARTG PI No: 111494), citric acid monohydrate, dilute hydrochloric acid, neotame, propylene glycol, and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Do not refrigerate or freeze. Store in original container.
Store below 30°C.
6.5 Nature and Contents of Container
ZolpiMist is presented in a two-part child-resistant polypropylene (PP) packaging system. An outer child-resistant PP base houses a type 1 amber glass bottle filled with ZolpiMist spray solution fitted with a snap-on metered-dose pump assembly and clear over cap. The base and fitted components are then sealed using an outer child-resistant PP cap. One and two actuations of ZolpiMist are equal to 5 and 10 mg of zolpidem tartrate, respectively.
ZolpiMist is supplied in two different volumes.
The 7.7 mL oromucosal spray contains 8.2 g of product formulation. There are 60 metered actuations per container after 7 initial priming actuations.
The 4.5 mL oromucosal spray contains 4.8 g of product formulation. There are 28 metered actuations per container after 7 initial priming actuations.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Zolpidem tartrate is a white to off white colourless, crystalline powder, sparingly soluble in water.
Chemical structure.
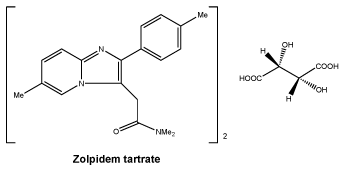 Its chemical name is 2-(4-methylphenyl)-N,N,6-trimethylimidazo [1,2,a] pyridine-3acetamide hemitartrate.
Its chemical name is 2-(4-methylphenyl)-N,N,6-trimethylimidazo [1,2,a] pyridine-3acetamide hemitartrate.
Its molecular formula is (C19H21N3O)2,C4H6O6. MW is 764.9.
CAS number.
99294-93-6 (zolpidem tartrate) and 82626-48-0 (zolpidem).7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (Schedule 4).
Summary Table of Changes


 Table 2 was derived from a pool of three placebo-controlled long-term efficacy trials involving zolpidem. These trials involved patients with chronic insomnia who were treated for 28 to 35 nights with zolpidem at doses of 5, 10 or 15 mg. The table is limited to data from doses up to and including 10 mg, the highest dose recommended for use. The table includes only adverse events occurring at an incidence of at least 1% for zolpidem patients.
Table 2 was derived from a pool of three placebo-controlled long-term efficacy trials involving zolpidem. These trials involved patients with chronic insomnia who were treated for 28 to 35 nights with zolpidem at doses of 5, 10 or 15 mg. The table is limited to data from doses up to and including 10 mg, the highest dose recommended for use. The table includes only adverse events occurring at an incidence of at least 1% for zolpidem patients.

 Its chemical name is 2-(4-methylphenyl)-N,N,6-trimethylimidazo [1,2,a] pyridine-3acetamide hemitartrate.
Its chemical name is 2-(4-methylphenyl)-N,N,6-trimethylimidazo [1,2,a] pyridine-3acetamide hemitartrate.