What is in this leaflet
This leaflet answers some common questions about ZYKADIA.
The information in this leaflet was last updated on the date listed on the final page. More recent information on the medicine may be available.
You should ensure that you speak to your pharmacist or doctor to obtain the most up to date information on the medicine. You can also download the most up to date leaflet from www.novartis.com.au.
Those updates may contain important information about the medicine and its use of which you should be aware.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking this medicine against the benefits they expect it will provide.
If you have any concerns about this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What ZYKADIA is used for
ZYKADIA capsules contain an active substance called ceritinib. ZYKADIA is used to treat people with non-small cell lung cancer (NSCLC) that is advanced or has spread to other parts of the body (metastatic) and is caused by a defect in a gene called ALK (anaplastic lymphoma kinase).
If you have any questions about how ZYKADIA works or why this medicine has been prescribed for you, ask your doctor or pharmacist.
ZYKADIA is not to be used in children or adolescents under 18 years of age.
This medicine is not addictive.
This medicine is available only with a doctor's prescription.
Before you take ZYKADIA
When you must not take it
Do not take ZYKADIA if you have an allergy to ceritinib, the active ingredient, or to any of the other ingredients listed at the end of this leaflet.
If you develop these symptoms tell your doctor straight away. They are some of the signs of an allergic reaction:
- difficulty in breathing or swallowing
- swelling of the face, lips, tongue or throat
- severe itching of the skin, with a red rash or raised bumps
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. In that case, return it to your pharmacist.
Before you start to take it
Take special care with ZYKADIA.
If any of these apply to you, tell your doctor or pharmacist before taking ZYKADIA:
- If you have problems with your liver.
- If you have problems with your lungs or problems breathing.
- If you have problems with your heart, including a condition called long QT syndrome.
- If you have diabetes (a high level of sugar in the blood).
- If you have/had problems with your pancreas.
- If you are currently taking steroids.
- If you are pregnant, think you may be pregnant, or plan to become pregnant (see section Pregnancy).
- If you are breast-feeding or plan to breast-feed (see section Breast-feeding).
Taking other medicines
Tell your doctor or pharmacist if you are taking or have recently taken any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop such as vitamins or herbal supplements, because they might interact with ZYKADIA.
It is particularly important that you mention any of the following medicines:
- Medicines used to treat AIDS/HIV such as ritonavir or saquinavir
- Medicines used to treat infections. These include medicines which treat fungal infections, such as antifungals like ketoconazole, itraconazole, voriconazole or posaconazole, or medicines which treat certain types of bacterial infections, such as antibiotics like telithromycin
- Medicines used to treat depression such as nefazodone, or psychosis such as pimozide
- St. John's Wort - an herbal product used to treat depression and other conditions, also known as Hypericum perforatum
- Medicines which stop seizures or fits (anti-epileptics such as phenytoin, carbamazepine, or phenobarbitone)
- Medicines used to treat tuberculosis such as rifampin or rifabutin
- Medicines used to treat irregular heartbeat such as quinidine
- Medicines used for increasing motility in the upper gastrointestinal tract such as cisapride
- Midazolam, a medicine used to treat acute seizures, or as a sedative before or during surgery or medical procedures
- Warfarin, an anticoagulant medicine used to prevent blood clots
- Diclofenac, a medicine used to treat joint pain and inflammation
- Cyclosporin, tacrolimus and sirolimus, medicines used in organ transplantation to prevent transplant organ rejection
- Dihydroergotamine and ergotamine, medicines used to treat migraine
- Alfentanil and fentanyl, medicines used to treat severe pain
- Gastric acid-reducing agents (e.g. proton pump inhibitors, H2-receptor antagonists, antacids)
Ask your doctor or pharmacist if you are not sure whether your medicine is one of the medicines listed above.
These medications should be used with care or may need to be avoided during your treatment with ZYKADIA.
If you are taking any of these, your doctor might prescribe an alternative medicine for you.
If you have not told your doctor about any of these things, tell him/her before you start taking this medicine.
How to take ZYKADIA
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
Always take ZYKADIA exactly as your doctor has told you. If you do not understand the instructions on the label, ask your doctor or pharmacist for help.
Do not change the dose without talking to your doctor.
How much to take
The recommended dose of ZYKADIA is 450 mg taken once daily with food (for example a snack or full meal) at the same time every day. Your doctor will tell you exactly how many capsules of ZYKADIA you need to take. Do not change the dose without talking to your doctor.
How to take it
Take ZYKADIA with food.
Swallow ZYKADIA capsules whole with water. Do not chew or crush capsules.
You should not eat grapefruit or drink grapefruit juice during your treatment with ZYKADIA. It may make the amount of ZYKADIA in your blood increase to a harmful level.
When to take it
Take ZYKADIA once a day at about the same time each day.
If vomiting occurs after you swallow ZYKADIA capsules, do not take any more capsules until your next scheduled dose.
Do not stop taking ZYKADIA unless your doctor tells you to.
This is a long-term treatment, possibly lasting for months. Your doctor will monitor your condition to see that the treatment is having the desired effect.
If you have questions about how long to take ZYKADIA, talk to your doctor or pharmacist.
If you forget to take it
If you forget to take ZYKADIA, do not take the missed dose; just take your next dose at the regular time. Do not take a double dose of ZYKADIA to make up for the dose you missed.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital (take the pack with you) if you think that you or anyone else may have taken too much ZYKADIA. Do this even if there are no signs of discomfort or poisoning. Keep the telephone number handy.
While you are taking ZYKADIA
Things you must do
Tell your doctor or pharmacist immediately if you get any of these symptoms during treatment with ZYKADIA:
- If you experience tiredness, itchy skin, yellow skin or the whites of your eyes turn yellow, nausea or vomiting, decreased appetite, pain on the right side of your stomach, dark or brown urine, or you bleed or bruise more easily than normal because these may be signs of liver problems.
- If you experience new or worsening symptoms such as cough with or without mucous, fever, chest pain, trouble breathing or shortness of breath because these may be signs of lung problems.
- If you experience chest pain or discomfort, changes in your heartbeat (fast or slow), light-headedness, fainting, dizziness, blue discoloration of your lips, shortness of breath, or swelling of your lower limbs (oedema) or skin because these may be signs of heart problems.
- If you experience diarrhoea, nausea or vomiting because they may be signs of gastrointestinal problems.
- If you experience excessive thirst or increased frequency of urination because they may be signs of a high level of sugar in the blood.
Your doctor may need to adjust, temporarily stop or completely discontinue your treatment with ZYKADIA.
Keep all of your doctor's APPOINTMENTS so that your progress can be checked.
Your doctor should do blood tests before you start treatment with ZYKADIA to check your liver and pancreas and to check the level of sugar in your blood. Your doctor should do blood tests every month thereafter to check your liver while you are taking ZYKADIA. Your doctor should also do blood tests to check your pancreas and the level of sugar in your blood regularly while you are taking ZYKADIA.
WOMEN OF CHILD BEARING AGE
- You must use a highly effective method of birth control during treatment with ZYKADIA and for 3 months after stopping ZYKADIA. ZYKADIA may decrease the effectiveness of the oral contraceptive.
Your doctor will discuss with you the optimal birth control (contraceptive) method for you to use during treatment with ZYKADIA.
PREGNANCY AND BREAST-FEEDING
- ZYKADIA should not be used during pregnancy, unless the potential benefit to the patient outweigh the potential risk to the foetus.
- If you are pregnant or think you might be pregnant or plan to become pregnant, ask your doctor for advice.
Your doctor will discuss with you the potential risks of taking ZYKADIA during pregnancy. - ZYKADIA should not be used during breast-feeding.
You and your doctor will decide together whether you should breast-feed or take ZYKADIA. YOU SHOULD NOT DO BOTH.
Tell your doctor straight away if you become pregnant while taking this medicine.
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking this medicine.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking this medicine.
Things you must not do
This medicine has been prescribed for you only. Do not use it for any other complaints unless your doctor tells you to.
Do not give this medicine to anyone else, even if their condition seems similar to yours. It may harm them.
Things to be careful of
Be careful driving, operating machinery or doing jobs that require you to be alert until you know how ZYKADIA affects you.
Side effects
Tell your doctor or pharmacist as soon as possible if you notice any side effects not listed in this leaflet.
As with all medicines, patients treated with ZYKADIA may experience side effects, although not everybody gets them.
STOP taking ZYKADIA and seek medical help immediately if you experience any of the following, which may be signs of an allergic reaction:
- difficulty in breathing or swallowing
- swelling of the face, lips, tongue or throat
- severe itching of the skin, with a red rash or raised bumps
Tell your doctor or pharmacist IMMEDIATELY if you experience any of the following SERIOUS SIDE EFFECTS. You may need medical treatment.
- Irregular or slow heartbeat
- Pain in the chest
- Cough, difficult or painful breathing, wheezing, pain in chest when breathing in, fever
- Yellow skin and eyes, nausea, loss of appetite, dark urine
- Severe upper stomach pain resulting from inflammation of the pancreas
The following list includes OTHER SIDE EFFECTS of your medicine. If they BECOME SEVERE, please tell your doctor, pharmacist or healthcare provider.
VERY COMMON SIDE EFFECTS
- Diarrhoea
- Nausea
- Vomiting
- Abdominal pain
- Tiredness (fatigue)
- Decreased appetite
- Weight decreased
- Constipation
- Heartburn
- Rash
COMMON SIDE EFFECTS
- Vision problems
- Excessive thirst, high urine flow, increased appetite with weight loss
- Significantly decreased urine flow
Some people may have other side effects not yet known or mentioned in this leaflet. If you notice any side effects not listed in this leaflet, please inform your doctor or pharmacist.
Some side effects, for example, changes in kidney and liver function or abnormal blood test results (high levels of pancreatic enzymes called lipase and amylase in the blood), can only be found when your doctor does tests from time to time to check your progress.
After using ZYKADIA
Storage
- Keep your medicine in the original package until it is time to take it.
- Store it in a cool, dry place, below 30°C
- Do not store this medicine or any other medicine in the bathroom or near a sink
- Do not leave it in the car or on window sills.
Keep the medicine where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine you have left over.
Product description
What it looks like
ZYKADIA hard capsules are opaque white and blue. The opaque blue cap is marked with black ink "LDK 150MG" and the opaque white body is marked with black ink "NVR". The capsule contains a white to almost white powder. One blister strip contains 10 hard capsules. Multipacks contain 150 (3 packs of 50) hard capsules.
Ingredients
ZYKADIA hard capsules contain 150 mg of the active ingredient ceritinib.
Excipients with known effect:
contains sulfites.
The inactive capsule contents are: microcrystalline cellulose, hyprolose, sodium starch glycolate type A, magnesium stearate, colloidal anhydrous silica.
The hard capsule colouring agents are titanium dioxide, indigo carmine.
The printing ink used to mark the capsule is OPACODE monogramming ink S-1-277002 BLACK.
Sponsor
ZYKADIA® is supplied in Australia by:
NOVARTIS Pharmaceuticals Australia Pty Limited
ABN 18 004 244 160
54 Waterloo Road
Macquarie Park NSW 2113
Telephone 1 800 671 203
Web site: www.novartis.com.au
®= Registered Trademark
This leaflet was prepared on
16 June 2020
Australian Registration Number.
AUST R 235737
Internal Document Code:
(zyk070521c.doc) based on PI (zyk070521i.doc)
Published by MIMS July 2021


 Avoid concurrent use of strong CYP3A inhibitors during treatment with Zykadia (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). If concomitant use of a strong CYP3A inhibitor is unavoidable, reduce the Zykadia dose by approximately one-third, rounded to the nearest multiple of the 150 mg dosage strength. Patients should be carefully monitored for safety. If long-term concomitant treatment with a strong CYP3A inhibitor is necessary and the patient tolerates the reduced dose well, the dose may be increased again with careful monitoring for safety, to avoid potential under-treatment. After discontinuation of a strong CYP3A inhibitor, resume the Zykadia dose that was taken prior to initiating the strong CYP3A inhibitor.
Avoid concurrent use of strong CYP3A inhibitors during treatment with Zykadia (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). If concomitant use of a strong CYP3A inhibitor is unavoidable, reduce the Zykadia dose by approximately one-third, rounded to the nearest multiple of the 150 mg dosage strength. Patients should be carefully monitored for safety. If long-term concomitant treatment with a strong CYP3A inhibitor is necessary and the patient tolerates the reduced dose well, the dose may be increased again with careful monitoring for safety, to avoid potential under-treatment. After discontinuation of a strong CYP3A inhibitor, resume the Zykadia dose that was taken prior to initiating the strong CYP3A inhibitor. The most frequently (> 10%) reported AEs for study A2303 regardless of study drug relationship in either treatment group are reported in Table 3.
The most frequently (> 10%) reported AEs for study A2303 regardless of study drug relationship in either treatment group are reported in Table 3.
 In the dose optimisation study A2112 (ASCEND-8) in both previously treated and untreated patients with ALK-positive advanced NSCLC, the overall safety profile of Zykadia at the recommended dose of 450 mg with food (N = 108) was consistent with Zykadia 750 mg fasted (N = 110), except for a reduction in gastrointestinal adverse drug reactions, while achieving comparable steady-state exposure (see Section 5.2 Pharmacokinetic Properties). The incidence and severity of gastrointestinal adverse drug reactions (diarrhoea 59.3%, nausea 42.6%, vomiting 38.0%; 1.9% reported a grade 3/4 event) were reduced for patients treated with Zykadia 450 mg with food compared to 750 mg fasted (diarrhoea 80%, nausea 60%, vomiting 65.5%; 17.3% reported a grade 3/4 event). In patients treated with Zykadia 450 mg with food, 24.1% of patients had at least one adverse event that required dose reduction and 55.6% of patients had at least one adverse event that required study drug interruption.
In the dose optimisation study A2112 (ASCEND-8) in both previously treated and untreated patients with ALK-positive advanced NSCLC, the overall safety profile of Zykadia at the recommended dose of 450 mg with food (N = 108) was consistent with Zykadia 750 mg fasted (N = 110), except for a reduction in gastrointestinal adverse drug reactions, while achieving comparable steady-state exposure (see Section 5.2 Pharmacokinetic Properties). The incidence and severity of gastrointestinal adverse drug reactions (diarrhoea 59.3%, nausea 42.6%, vomiting 38.0%; 1.9% reported a grade 3/4 event) were reduced for patients treated with Zykadia 450 mg with food compared to 750 mg fasted (diarrhoea 80%, nausea 60%, vomiting 65.5%; 17.3% reported a grade 3/4 event). In patients treated with Zykadia 450 mg with food, 24.1% of patients had at least one adverse event that required dose reduction and 55.6% of patients had at least one adverse event that required study drug interruption.
 There was no significant difference in OS in a pre-specified interim analysis conducted at 42% of the events required for the final analysis.
There was no significant difference in OS in a pre-specified interim analysis conducted at 42% of the events required for the final analysis.
 There was no significant difference in OS in a pre-specified interim analysis conducted at approximately 50% of the events required for the final analysis.
There was no significant difference in OS in a pre-specified interim analysis conducted at approximately 50% of the events required for the final analysis. Analyses of patient-reported outcome measures suggested ceritinib prolonged time to deterioration for the lung cancer specific symptoms cough, pain and dyspnoea compared to chemotherapy. The patient-reported delay to deterioration may be an overestimation, because patients were not blinded to treatment assignment.
Analyses of patient-reported outcome measures suggested ceritinib prolonged time to deterioration for the lung cancer specific symptoms cough, pain and dyspnoea compared to chemotherapy. The patient-reported delay to deterioration may be an overestimation, because patients were not blinded to treatment assignment.

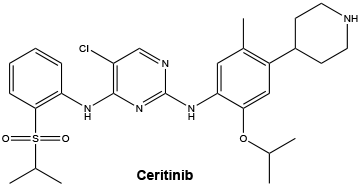 INN: ceritinib (free base).
INN: ceritinib (free base).