Key points
- Muscle symptoms are the most commonly reported form of statin intolerance.
- The true incidence of statin-associated muscle symptoms is likely to be lower than what is observed in clinical practice.
- There is no standardised definition or diagnostic test for statin-associated muscle symptoms.
- Systematic assessment of muscle complaints can help healthcare professionals establish whether statin therapy is the likely cause, and manage patients accordingly.
Statins for CVD risk – keep calm and carry on
Reduction of LDL cholesterol is a crucial component in the prevention and management of cardiovascular disease (CVD).1,2 Statins, proven and effective medicines for reducing LDL cholesterol, have been shown to significantly reduce the risk of cardiovascular events and mortality, both in primary and secondary prevention.1 However, with increasing use, there is also increasing awareness of and publicity about adverse effects associated with statin use. Some patients on statins report an inability to tolerate therapeutic doses, most commonly because of muscle-related adverse effects.2,3
This perceived high incidence of statin-associated muscle symptoms risks compromising adherence to an otherwise highly efficacious and generally well-tolerated class of medicines. Poor adherence to statin therapy increases risk of cardiovascular events and death.4,5
So what does the evidence say about statin intolerance and statin-associated muscle symptoms? And how should you assess and manage such symptoms in your practice to ensure high-risk patients are not needlessly left without a potentially life-saving medicine?
Statin-associated muscle symptoms – an umbrella term
Statin intolerance is an inability to tolerate the dose of a statin required to sufficiently reduce a person’s LDL cholesterol and/or cardiovascular risk, due to significant adverse effects including abnormally elevated markers of either liver or muscle function.6,7
There is evidence to indicate that statins may increase the risk of muscle symptoms, elevated liver function enzymes and new-onset diabetes.7,8
Muscle symptoms are the most common form of statin intolerance encountered in clinical practice.2,7,8 ‘Statin-associated muscle symptoms’ – abbreviated here as ‘SAMS’ – is the overarching term used to describe any adverse muscle effects linked to the use of a statin. These can range from mild non-life threatening symptoms such as myalgia, to more serious but rare conditions such as rhabdomyolysis.2,9
SAMS are not very common in controlled trials
Myalgia during statin therapy is reported in a variable proportion of patients between 7% and 29% in clinical practice and some observational studies.2,9,10 Although muscle complaints are common among patients receiving statin therapy in practice, evidence from controlled trials suggests the true incidence of SAMS is between 1% and 5%.
STOMP is the only randomised controlled trial (RCT) designed specifically to investigate the incidence of SAMS and the effect of statins on muscle and exercise performance.10
In this study, statin-associated myalgia was defined as new or increased muscle pain, cramps, or aching that :
- was not associated with exercise
- persisted for 2 weeks
- resolved within 2 weeks of stopping the statin and
- recurred within 4 weeks of restarting the medication.
Statin-naïve patients (n = 420) were treated with either atorvastatin 80 mg or placebo for 6 months. At follow up, a myalgia incidence rate of 9.4% was reported among the statin-treated patients.10 Of the placebo-treated group, 4.6% met the same, standardised definition of myalgia.10
Other RCTs have also reported a low incidence of myalgia (1%–5%) and similar rates in statin and placebo groups.2,9 The most recent systematic reviews of this topic have shown that the incidence of muscle symptoms was similar in statin- and placebo-treated patients (see Figure 1).11,12 It is worth noting that strict inclusion and exclusion criteria mean that patients with an increased risk for musculoskeletal symptoms may be underrepresented in RCTs.2,12 Nonetheless, these incidence rates are considerably lower than the upper limit reported in practice.2,9,10
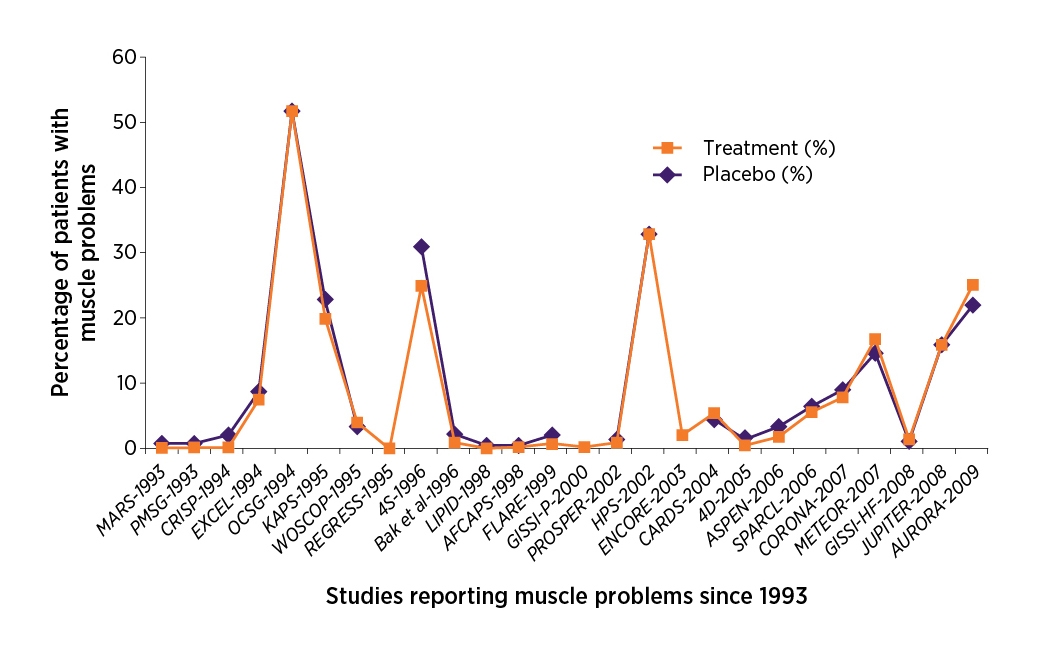
Also notable are results from a 2016 randomised crossover study of patients with a strong history of muscle-related statin intolerance.a,3 At follow up, over a quarter of these patients (26.5%) reported muscle symptoms with placebo but not with a statin. Another 17.3% of patients reported no symptoms with either treatment and another 9.8% experienced symptoms with both treatments.3
Lack of standard definitions for SAMS, and the absence of a standardised diagnostic test or validated questionnaire, probably contribute to the notable difference in reported incidence rates.
Serious statin-associated myopathies are rare. Muscle symptoms with creatine kinase elevations > 10 x the upper limit of normal (ULN) occur in 1/1000 to 1/10,000 patients per year depending on the statin, its dose, and the presence of other risk factors.2 Rhabdomyolysis is also extremely rare, with an incidence risk of approximately 1 per 100,000 patients per year.2,8
a Patients were aged between 18 and 80 years and unable to tolerate atorvastatin at 10 mg and any other statin at any dose or, alternatively, three or more statins (one at lowest average daily starting dose and two others at any dose).
Not all muscle symptoms are SAMS
A patient being treated with a statin presents with muscle complaints and the question arises – is it SAMS? To maximise the benefits of statin therapy and avoid unnecessary medicine changes, use a systemic approach to assess and confirm a diagnosis of SAMS in patients presenting with muscle symptoms.
Guidelines recommend using statin discontinuation and rechallenge to diagnose and gauge severity of statin intolerance.2,13 However, prior to a discontinuation-rechallenge, health professionals can assess the likelihood that muscle symptoms are due to a statin by undertaking a detailed history and clinical examination, and taking into consideration:2,13,14
- nature of the symptoms
- timing of the symptoms
- elevation in creatine kinase (CK) levels and their association with the start of statin therapy.
The SAMS Assessment Guide can help you establish the likelihood that muscle symptoms in your patient may be associated with their statin therapy. For a more comprehensive assessment, you can use the American College of Cardiology’s Statin Intolerance Tool.
Being aware of risk factors for statin intolerance can also help you recognise patients who may be at higher risk of developing SAMS. Clinical studies have identified a range of risk factors including genetics, patient characteristics, comorbid conditions and medicines that increase a patient’s risk for statin intolerance (see Table 1).15
| Patient characteristics | ||
|---|---|---|
|
||
| Comorbid diseases | Medicines | Genetics |
|
|
|
A systematic approach to management of suspected SAMS
In patients with suspected SAMS, some guidelines recommend statin discontinuation and rechallenge to confirm or rule out statin intolerance.2,13 In patients with confirmed SAMS, reconsider the statin treatment regimen. Guidelines recommend trialling reduced or intermittent dosing, or switching to a different statin before considering a non-statin lipid-modifying medicine.2,13
The SAMS Management Algorithmb illustrates how to use the discontinuation-rechallenge technique to determine if presenting muscle symptoms are statin-induced and manage patients accordingly.
b Developed based on the 2012 Therapeutic Guidelines: Cardiovascular and 2016 European Society of Cardiology/European Atherosclerosis Society Guidelines for the management of dyslipidaemias, with input from experts: Assoc Prof David Colquhoun, Prof Ian Hamilton-Craig, Prof Mark Harris, Assoc Prof Karam Kostner, Prof Leonard Kritharides, Prof Mark Nelson, Dr Daniel Scherer, Assoc Prof David Sullivan, Prof Andrew Tonkin, Mr Garth Birdsey, Dr Chris Helms.
Involving the patient in SAMS
Large clinical trials and extensive post-marketing experience have shown that long-term statin therapy is generally well tolerated.17 Nonetheless, adverse effects remain the main reported reason for non-adherence and discontinuation of statin therapy.2 Increased media coverage of statins and their perceived adverse effects further contributes towards patient non-adherence to statin therapy.2,8 For example, more than 28,000 Australians ceased statin treatment after the ABC’s Catalyst program aired ‘Heart of the Matter’, a report critical of statins, in October 2013.18
While changes to therapy may be necessary and beneficial in some patients, sudden discontinuation of a medicine without consultation with a health professional, or intentional poor adherence, are generally linked to poorer health outcomes. Inadequate adherence to statins in at-risk patients is associated with increased risk of cardiovascular events and higher mortality.2
Ongoing patient education and regular review can help address concerns around medicine safety and underline the importance of adherence. In addition to explaining the purpose of the medicine and how to take it, ensure patients understand that lipid-lowering medicines must be taken continuously and long term in order to be effective. It may also be helpful to actively raise any concerns the patient has about statins prior to starting treatment and to reassure them of the benefit-risk ratio of the medicine.
When discussing safety of statin therapy, it may help to:
- describe potential adverse effects, including SAMS
- explain that SAMS may not be as common as they are sometimes said to be, and are generally mild and manageable in patients who experience them3,7,10,12
- reassure patients that serious muscle damage associated with statin treatment is rare2,7,8
- advise patients to contact you if they experience muscle symptoms
- remind patients to keep taking their statin as recommended as CVD outcomes are worse in those with poor adherence.2,4,9
For patients presenting with muscle complaints, systematic assessment including statin discontinuation and rechallenge, can help confirm or rule out SAMS, and guide ongoing lipid-lowering therapy.2,13 Consider the NPS MedicineWise Statins Patient Action Plan for assessing and managing muscle symptoms, available for use with patients who report muscle symptoms while taking a statin. It includes information that supports a systematic and collaborative approach to the assessment and management of muscle symptoms in patients taking statins.
Long-term concerns with statins
Publicised reports of adverse effects caused by statin therapy have been linked to reluctance among doctors to discuss and prescribe statins, and reduced adherence among patients.8 Keeping in mind the negative impact of non-adherence in high-risk patients, it is important for health professionals to be aware of the evidence on these concerns and educate patients appropriately.
Adverse events shown in studies to be associated with long-term use of statins include diabetes and possibly haemorrhagic stroke.8 However, the overall benefits of decreased cardiovascular events in at-risk patients outweigh the risk of these adverse events.8 Lowering LDL cholesterol by 2 mmol/L with statin therapy over 5 years in 10 000 patients would be expected to prevent major vascular events in about 1000 secondary prevention patients and 500 high-risk primary prevention patients.
Cancer

No apparent link between statin therapy and cancer has been found.6,8,11
- Based on large meta-analyses and systematic reviews, statin therapy does not increase risk of cancer – either overall or at any particular site.
- Based on large meta-analyses and systematic reviews, statin therapy does not increase risk of cancer – either overall or at any particular site.
Dementia

No apparent link between statin therapy and cognitive impairment can be reported.8,11
- Large RCTs and systematic reviews report no evidence of any adverse cognitive effects of statin therapy, including increase in risk of dementia or Alzheimer disease.
- Large RCTs and systematic reviews report no evidence of any adverse cognitive effects of statin therapy, including increase in risk of dementia or Alzheimer disease.
Diabetes

A small absolute risk of developing diabetes with statin therapy has been documented.8
- Treating 10,000 people with statins over 5 years would cause 50‒100 new cases of diabetes. This may be of greatest clinical relevance to those on intensive statin regimens or with other risk factors for diabetes.
Stroke
A small probable increase in risk of haemorrhagic strokes.8,20
- Statin therapy reduces overall risk of stroke.8,11
- Data from RCTs and meta-analyses on the link between statins and haemorrhagic stroke are conflicting.8,20
- Based on studies that showed an increased risk, the absolute risk of haemorrhagic stroke is small. Treating 10,000 people with statins over 5 years would cause 5‒10 cases of haemorrhagic strokes.8
- Statin therapy reduces overall risk of stroke.8,11
- Data from RCTs and meta-analyses on the link between statins and haemorrhagic stroke are conflicting.8,20
- Based on studies that showed an increased risk, the absolute risk of haemorrhagic stroke is small. Treating 10,000 people with statins over 5 years would cause 5‒10 cases of haemorrhagic strokes.8
Download and print
Please note updates made to article references on the website have not been reflected in this pdf.
Expert reviewers
Prof Ian Hamilton-Craig MB, BS (Adel), PhD (McMaster), FRACP, FCSANZ, FLS (Lond)
Professor of Preventive Cardiology
Flinders University School of Medicine
A/Prof Karam Kostner
Director Cardiology, Mater Hospital Brisbane
Associate Professor Medicine University of Queensland
References
- National Vascular Disease Prevention Alliance. Guidelines for the management of absolute cardiovascular disease risk. 2012.
- Stroes ES, Thompson PD, Corsini A, et al. Statin-associated muscle symptoms: impact on statin therapy-European Atherosclerosis Society Consensus Panel Statement on Assessment, Aetiology and Management. Eur Heart J 2015;36:1012-22.
- Nissen SE, Stroes E, Dent-Acosta RE, et al. efficacy and tolerability of evolocumab vs ezetimibe in patients with muscle-related statin intolerance: The GAUSS-3 Randomized Clinical Trial. JAMA 2016;315:1580-90.
- Chowdhury R, Khan H, Heydon E, et al. Adherence to cardiovascular therapy: a meta-analysis of prevalence and clinical consequences. Eur Heart J 2013;34:2940-8.
- De Vera MA, Bhole V, Burns LC, et al. Impact of statin adherence on cardiovascular disease and mortality outcomes: a systematic review. Br J Clin Pharmacol 2014;78:684-98.
- Mancini GB, Tashakkor AY, Baker S, et al. Diagnosis, prevention, and management of statin adverse effects and intolerance: Canadian Working Group Consensus update. Can J Cardiol 2013;29:1553-68.
- Fitchett DH, Hegele RA, Verma S. Cardiology patient page. Statin intolerance. Circulation 2015;131:e389-91.
- Collins R, Reith C, Emberson J, et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet 2016;388:2532-61.
- Rosenson RS, Baker SK, Jacobson TA, et al. An assessment by the Statin Muscle Safety Task Force: 2014 update. J Clin Lipidol 2014;8:S58-71.
- Parker BA, Capizzi JA, Grimaldi AS, et al. Effect of statins on skeletal muscle function. Circulation 2013;127:96-103.
- Chou R, Dana T, Blazina I, et al. Statins for Prevention of Cardiovascular Disease in Adults: Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA 2016;316:2008-24.
- Ganga HV, Slim HB, Thompson PD. A systematic review of statin-induced muscle problems in clinical trials. Am Heart J 2014;168:6-15.
- Therapeutic Guidelines Ltd (eTG March 2021 edition). Cardiovascular. West Melbourne: Therapeutic Guidelines Limited, 2018 (accessed 20 September 2021).
- Australian Medicines Handbook. Statins. Adelaide: Australian Medicines Handbook Pty Ltd, 2021. (accessed 23 September 2021)..
- Ahmad Z. Statin intolerance. Am J Cardiol 2014;113:1765-71.
- Zhang H, Plutzky J, Skentzos S, et al. Discontinuation of statins in routine care settings: a cohort study. Ann Intern Med 2013;158:526-34.
- Bruckert E, Hayem G, Dejager S, et al. Mild to moderate muscular symptoms with high-dosage statin therapy in hyperlipidemic patients--the PRIMO study. Cardiovasc Drugs Ther 2005;19:403-14.
- Schaffer AL, Buckley NA, Dobbins TA, et al. The crux of the matter: Did the ABC's Catalyst program change statin use in Australia? Med J Aust 2015;202:591-5.
- Thompson PD, Panza G, Zaleski A, et al. Statin-associated side effects. J Am Coll Cardiol 2016;67:2395-410.
- McKinney JS, Kostis WJ. Statin therapy and the risk of intracerebral hemorrhage: a meta-analysis of 31 randomized controlled trials. Stroke 2012;43:2149-56.

