1. Why am I using everolimus?
Everocan contains the active ingredient everolimus. EVEROCAN is used to treat adult patients who have had kidney, heart or liver transplants. It helps to prevent your body from rejecting the transplanted organ. EVEROCAN contains the active ingredient everolimus. It belongs to a group of medicines called immunosuppressants. These medicines help to control your body's immune system.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason. EVEROCAN is available only with a doctor's prescription and is not addictive. There is not enough information to recommend the use of this medicine in children and adolescents.
2. What should I know before I use everolimus?
Warnings
- Do not use everolimus if you are allergic to:
- Everolimus, or any of the ingredients listed at the end of this leaflet.
- A similar medicine called Rapamune which contains the active ingredient sirolimus. - Some of the symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty breathing; sudden difficulty swallowing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
- Always check the ingredients to make sure you can use this medicine.
- Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes. Your doctor will want to know if you are prone to allergies.
Check with your doctor if you:
- have any other medical conditions such as problems with your liver, high cholesterol.
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
- Check with your doctor if you are pregnant or intend to become pregnant.
- Talk to your doctor if you are breastfeeding or intend to breastfeed.
- EVEROCAN is not recommended for use during pregnancy. Your doctor can discuss with you the risks and benefits involved.
- Tell your doctor if you are breastfeeding. Breastfeeding is not recommended while you are taking EVEROCAN. It is not known whether EVEROCAN passes into breast milk and could affect your baby.
- Tell your doctor if you are male and you want to father children. EVEROCAN may reduce sperm production in men, thus reducing the ability to father children. The effect is generally reversible. Tell your doctor if you are male or female and you want children.
- EVEROCAN may cause infertility in male or female patients.
- Medicines that suppress the immune system like EVEROCAN reduce your body's ability to fight against infections. It is advisable to consult your doctor if you have a fever or generally feel unwell, or have local symptoms such as coughing or a burning sensation when urinating that are severe or persistent over several days.
- Consult your doctor right away if you feel confused, have problems speaking, memory loss, a headache, impaired vision or seizures, as these may be symptoms of a rare but very serious condition called progressive multiple leukoencephalopathy (PML).
- Tell your doctor if you have had recent major surgery, or if you still have an unhealed wound following surgery.
- EVEROCAN may increase the risk of wound- healing problems.
- If you have not told your doctor about any of the above, tell him/her before you start taking EVEROCAN.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and EVEROCAN may interfere with each other. These include:
- Immunosuppressive medicines other than cyclosporine for microemulsion, tacrolimus or corticosteroids.
- Antibiotics such as rifampicin, clarithromycin, erythromycin and rifabutin.
- Antifungal medicines such as ketoconazole, fluconazole and itraconazole.
- Medicines for high blood pressure or heart problems such as diltiazem and verapamil, dronedarone, a medicine used to help regulate your heartbeat.
- Medicines used to treat HIV/ AIDS such as nelfinavir, indinavir, amprenavir, efavirenz and nevirapine
- Medicines used to lower blood cholesterol, such as atorvastatin, pravastatin or fibrates
- Midazolam, a medicine used to treat acute seizures, or used as a sedative before or during surgery or other medical procedure.
- Epilepsy medicines such as carbamazepine, phenobarbitone and phenytoin.
- Octreotide, a medicine used to treat acromegaly, a rare hormonal disorder that usually occurs in middle- aged adults.
- Medicines for stomach ulcer or reflux such as cimetidine and cisapride.
- Medicines to prevent nausea and vomiting such as metoclopramide.
- Danazol, which is used to treat endometriosis.
- Bromocriptine, which is used to treat Parkinson's disease.
- Imatinib, a medicine used to inhibit the growth of abnormal cells.
- St John's wort, an herbal product used to treat depression and other conditions.
Such medicines may be affected by EVEROCAN or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Some vaccines may be less effective if given when taking EVEROCAN.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect everolimus.
4. How do I use everolimus?
How much to take / use
Kidney and heart transplantations:
- The usual starting dose is 0.75 mg EVEROCAN twice daily, taken in the morning and in the evening together with cyclosporine.
- The first dose of EVEROCAN will be given as soon as possible after transplantation.
Liver transplantation:
- The general recommended daily dose is 1 mg EVEROCAN twice daily, taken in the morning and in the evening, together with tacrolimus.
- The first dose of EVEROCAN will be given approximately four weeks after transplantation.
- Follow the instructions provided and use everolimus until your doctor tells you to stop.
Your doctor will take regular blood tests to measure the amount of EVEROCAN in your blood. If needed, your doctor will adjust your dose depending on the results from your blood tests.
When to take / use everolimus
- Always take EVEROCAN twice a day. It is best to take the doses 12 hours apart if possible.
- Taking your doses 12 hours apart and at the same time each day will have the best effect. It will also help you remember when to take them.
- EVEROLIMUS can be taken with or without food but should always be taken the same way. (e.g. always with food or always without food).
- If you are taking cyclosporine or tacrolimus, you should take it at the same time as EVEROCAN.
How to take it
- Swallow the tablets whole with a full glass of water.
- Do not crush or chew them.
- Do not take EVEROCAN with grapefruit juice.
How long to take it
- Keep taking this medicine for as long as your doctor tells you.
- You will need to take EVEROCAN as long as you have your transplanted organ, or until your doctor changes your immunosuppressive medication.
If you forget to use everolimus
If it is almost time for your next dose skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed.
This may increase the chance of you getting an unwanted side effect.
If you miss more than one dose, ask your doctor for advice.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you use too much everolimus
If you think that you have used too much everolimus, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre (by calling 13 11 26), or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Published by MIMS July 2021


 Prior to dose reduction of ciclosporin it should be ascertained that steady state everolimus whole blood trough concentrations (C0) are equal to or above 3 nanogram/mL there are limited data regarding the dosing of everolimus with ciclosporin trough concentrations below 50 nanogram/mL, or C2 levels below 350 nanogram/mL, in the maintenance phase. If the patient cannot tolerate reduction of ciclosporin exposure, the continued use of everolimus should be reconsidered.
Prior to dose reduction of ciclosporin it should be ascertained that steady state everolimus whole blood trough concentrations (C0) are equal to or above 3 nanogram/mL there are limited data regarding the dosing of everolimus with ciclosporin trough concentrations below 50 nanogram/mL, or C2 levels below 350 nanogram/mL, in the maintenance phase. If the patient cannot tolerate reduction of ciclosporin exposure, the continued use of everolimus should be reconsidered. Prior to dose reduction of ciclosporin it should be ascertained that steady state everolimus whole blood trough concentrations are equal to or above 3 nanogram/mL. In cardiac transplantation there are limited data regarding dosing everolimus with ciclosporin trough concentrations below 50-100 nanogram/mL after 12 months. If the patient cannot tolerate reduction of ciclosporin exposure, the continued use of everolimus should be reconsidered.
Prior to dose reduction of ciclosporin it should be ascertained that steady state everolimus whole blood trough concentrations are equal to or above 3 nanogram/mL. In cardiac transplantation there are limited data regarding dosing everolimus with ciclosporin trough concentrations below 50-100 nanogram/mL after 12 months. If the patient cannot tolerate reduction of ciclosporin exposure, the continued use of everolimus should be reconsidered.



 The primary efficacy endpoint was a composite failure variable (biopsy-proven acute rejection, graft loss, death or loss to follow-up). The outcome is shown in Table 8.
The primary efficacy endpoint was a composite failure variable (biopsy-proven acute rejection, graft loss, death or loss to follow-up). The outcome is shown in Table 8. Changes in renal function, as shown by calculated glomerular filtration rate (GFR) using the MDRD formula are shown in Table 9.
Changes in renal function, as shown by calculated glomerular filtration rate (GFR) using the MDRD formula are shown in Table 9.

 Renal function in study A2411 did not meet the non-inferiority criteria (-6 mL/min) vs MMF. Mean creatinine clearance (Cockcroft-Gault formula) at 6 months: everolimus: 65.4 v. MMF: 72.2 mL/min (difference: -6.85 mL/min, 95% CI: -14.9, 1.2) and at 12 months: everolimus: 68.7 v. MMF: 71.8 mL/min (Difference: -3.10 mL/min 95% CI (-12.26, 6.06). The change from baseline was: everolimus: -6.0 mL/min v. MMF: -4.2 mL/min; p=0.697. Efficacy, expressed as the rate of biopsy-proven acute rejection episodes (ISHLT grade ≥ 3A), was maintained as comparable in the two groups at 12 months (everolimus: 22.8% v. MMF: 29.8%).
Renal function in study A2411 did not meet the non-inferiority criteria (-6 mL/min) vs MMF. Mean creatinine clearance (Cockcroft-Gault formula) at 6 months: everolimus: 65.4 v. MMF: 72.2 mL/min (difference: -6.85 mL/min, 95% CI: -14.9, 1.2) and at 12 months: everolimus: 68.7 v. MMF: 71.8 mL/min (Difference: -3.10 mL/min 95% CI (-12.26, 6.06). The change from baseline was: everolimus: -6.0 mL/min v. MMF: -4.2 mL/min; p=0.697. Efficacy, expressed as the rate of biopsy-proven acute rejection episodes (ISHLT grade ≥ 3A), was maintained as comparable in the two groups at 12 months (everolimus: 22.8% v. MMF: 29.8%). The primary efficacy endpoint was a composite failure variable, implying occurrence of any of the following: Biopsy Proven Acute Rejection (BPAR) episode of ISHLT grade ≥ 3A, acute rejection (AR) episode associated with hemodynamic compromise (HDC), graft loss/re-transplant, death, or loss to follow-up. Efficacy outcome at 12 and 24 months is shown in Table 13.
The primary efficacy endpoint was a composite failure variable, implying occurrence of any of the following: Biopsy Proven Acute Rejection (BPAR) episode of ISHLT grade ≥ 3A, acute rejection (AR) episode associated with hemodynamic compromise (HDC), graft loss/re-transplant, death, or loss to follow-up. Efficacy outcome at 12 and 24 months is shown in Table 13. The higher fatality rate in the everolimus arm relative to the MMF arm was mainly the result of an increased rate of fatalities from infection in the first three months among everolimus patients in the study sub-group of patients receiving thymoglobulin induction therapy. A notably higher 3-month incidence in severe infections in everolimus than MMF patients in the thymoglobulin subgroup appears to reflect greater immunosuppressive potency. The imbalance in fatalities within the thymoglobulin subgroup being particularly evident among patients hospitalised prior to transplantation and with L-ventricular assistance devices, suggests greater vulnerability in such patients to the consequences of infectious complications.
The higher fatality rate in the everolimus arm relative to the MMF arm was mainly the result of an increased rate of fatalities from infection in the first three months among everolimus patients in the study sub-group of patients receiving thymoglobulin induction therapy. A notably higher 3-month incidence in severe infections in everolimus than MMF patients in the thymoglobulin subgroup appears to reflect greater immunosuppressive potency. The imbalance in fatalities within the thymoglobulin subgroup being particularly evident among patients hospitalised prior to transplantation and with L-ventricular assistance devices, suggests greater vulnerability in such patients to the consequences of infectious complications. The reduced increase in intimal coronary thickness in everolimus relative to MMF patients was apparent regardless of age, gender, presence or absence of diabetes and maximum level of serum cholesterol observed by Month 12.
The reduced increase in intimal coronary thickness in everolimus relative to MMF patients was apparent regardless of age, gender, presence or absence of diabetes and maximum level of serum cholesterol observed by Month 12.

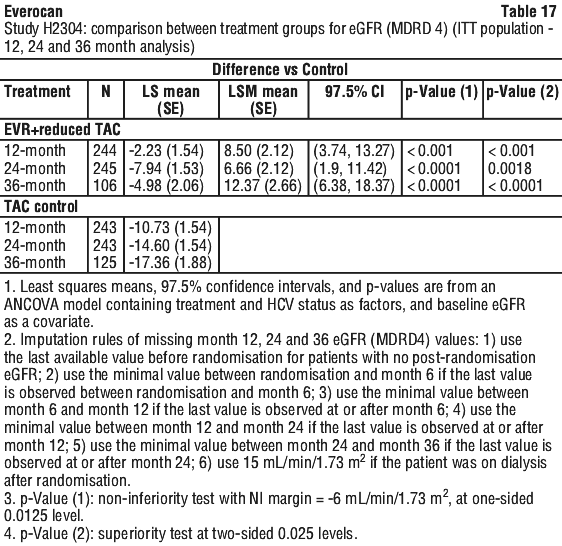 Statistically significant between-treatment group difference (everolimus + Reduced tacrolimus vs. tacrolimus control arm) was observed in favor of everolimus + Reduced tacrolimus arm for the mean eGFR from Week 6 up to Month 36 (including at study endpoint and treatment endpoint). At randomization, mean eGFR was 85.0 and 78.0 mL/min/1.73 m2 for the everolimus + Reduced tacrolimus and tacrolimus control arms respectively. At the Month 36 time point, the difference in mean eGFR between everolimus + Reduced tacrolimus and tacrolimus control was 15.2 mL/min/1.73 m2. The mean eGFR at Month 36 was 78.7 and 63.5 mL/min/1.73 m2 for the everolimus + Reduced tacrolimus and tacrolimus control arms respectively.
Statistically significant between-treatment group difference (everolimus + Reduced tacrolimus vs. tacrolimus control arm) was observed in favor of everolimus + Reduced tacrolimus arm for the mean eGFR from Week 6 up to Month 36 (including at study endpoint and treatment endpoint). At randomization, mean eGFR was 85.0 and 78.0 mL/min/1.73 m2 for the everolimus + Reduced tacrolimus and tacrolimus control arms respectively. At the Month 36 time point, the difference in mean eGFR between everolimus + Reduced tacrolimus and tacrolimus control was 15.2 mL/min/1.73 m2. The mean eGFR at Month 36 was 78.7 and 63.5 mL/min/1.73 m2 for the everolimus + Reduced tacrolimus and tacrolimus control arms respectively.

