1 Name of Medicine
Alanine, arginine, glycine, histidine, isoleucine, leucine, lysine hydrochloride, methionine, phenylalanine, proline, serine, threonine, tryptophan, tyrosine, valine, sodium acetate trihydrate, potassium chloride, calcium chloride dihydrate, magnesium sulfate heptahydrate, sodium glycerophosphate hydrate, zinc sulfate heptahydrate, glucose monohydrate, soya oil, olive oil, medium chain triglycerides and fish oil-rich omega-3 acids.
2 Qualitative and Quantitative Composition
Finomel emulsion for infusion is presented in a 3-compartment plastic bag. Each bag contains a sterile non-pyrogenic combination of a 10% amino acid solution with electrolytes, a 20% lipid emulsion and 42% glucose solution for Finomel.
The general compositions of the reconstituted formulations are summarised in Table 1:
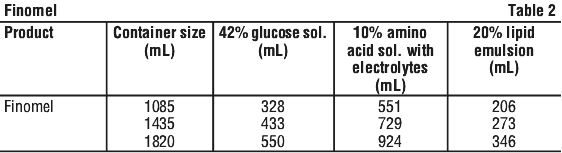
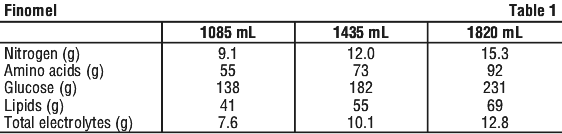 The chamber volume for each bag sizes are detailed in Table 2:
The chamber volume for each bag sizes are detailed in Table 2:
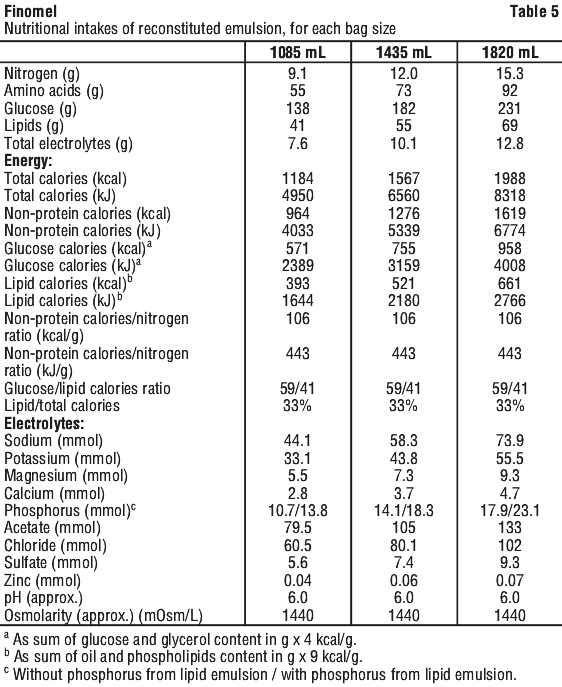
 For the detailed formulations, see Table 4 and Table 5.
For the detailed formulations, see Table 4 and Table 5.
The molecular formula and CAS (Chemical Abstract Service) registry number of the active substances are listed, see Section 6.7 Physicochemical Properties.
Excipients with known effects.
Egg lecithin, contains egg products.
For the full list of excipients, see Section 6.1 List of Excipients.4.1 Therapeutic Indications
Finomel is indicated for parenteral nutrition in adult patients when oral or enteral nutrition is impossible, insufficient or contraindicated.
4.2 Dose and Method of Administration
Use in one patient on one occasion only. Contains no antimicrobial preservative.
It is recommended that after opening the bag, the content should be used immediately, and not stored for subsequent infusion.
The dosage should be individualized depending on energy expenditure, the patient's clinical status, body weight, and ability to metabolize constituents of Finomel, as well as additional energy or proteins given orally/enterally. Therefore, the bag size should be chosen accordingly.
The average daily requirements for adults are:
In patients with normal nutritional state or in conditions with mild catabolic stress: 0.6-0.9 g amino acids/kg bw/day (0.10-0.15 g nitrogen/kg bw/day).
In patients with moderate to high metabolic stress with or without malnutrition: 0.9-1.6 g amino acids/kg bw/day (0.15-0.25 g nitrogen/kg bw/day).
In patients with special conditions (e.g. burns or marked anabolism) the nitrogen need may be even higher.
The maximum daily dose varies with the clinical condition of the patient and may change from day to day.
The flow rate should be increased gradually during the first hour. The administration flow rate must be adjusted taking into account the dose being administered, the daily volume intake and the duration of the infusion (see Section 4.9 Overdose).
The recommended infusion period is 14-24 hours.
Finomel.
Intravenous use, infusion into a central vein.
The dosage range of 13-31 mL/kg bw/day will provide 0.7-1.6 g amino acids/kg bw/day (corresponds to 0.11-0.26 g nitrogen/kg bw/day) and 14-33 kcal/kg bw/day of total energy (11-27 kcal/kg bw/day of non-protein energy).
The maximum infusion rate for glucose is 0.25 g/kg bw/h, for amino acids 0.1 g/kg bw/h, and for lipids 0.15 g/kg bw/h.
The infusion rate should not exceed 2.0 mL/kg bw/h (corresponding to 0.10 g amino acids, 0.25 g glucose and 0.08 g lipids/kg bw/h).
The recommended maximum daily dose is 35 mL/kg bw/day which will provide 1.8 g amino acids/kg bw/day (corresponding to 0.29 g nitrogen/kg bw/day), 4.5 g glucose/kg bw/day, 1.40 g lipids/kg bw/day and a total energy content of 38 kcal/kg bw/day (corresponding to 30 kcal/kg bw/day of non-protein energy).
Although there is a natural content of trace elements and vitamins in the product, the levels are insufficient to meet body requirements. Trace elements and vitamins should be added in sufficient quantities to meet individual patient requirements and to prevent deficiencies from developing. See instructions for making additions to this product.
Method of preparation. To open.
Remove the protective overpouch.
Discard the oxygen absorber sachet.
Use only if the bag is not damaged, the non-permanent seals are intact (i.e. no content mixture of any of the three chambers), the solution in the amino acids chamber and the solution in the glucose chamber are clear, colourless, or slightly yellow, free of visible particles, and the lipid emulsion is a homogeneous liquid with a milky appearance.
To mix the chambers.
Ensure that the product is at room temperature when breaking the nonpermanent seals.
Manually roll the bag onto itself on a firm, flat surface, starting at the top of the bag (hanger end). The non-permanent seals will disappear from the side near the inlets. Continue to roll the bag until the seals are open along approximately half of their length.
Mix by inverting the bag at least 3 times.
After reconstitution, the mixture is a homogeneous emulsion with a milky appearance.
To perform an addition.
After removing the protective cap from the additive port, one can add compatible additives via the additive port. Additions should be made aseptically.
No additions to the bag should be made without first checking the compatibility, as the formation of precipitates or destabilization of the lipid emulsion could result in vascular occlusion.
The addition of clinically needed electrolytes and trace elements should take into account the amounts already included in the initial bag formulation.
Finomel may only be added to medicinal or nutritional solutions for which compatibility has been documented. Compatibility with different products and the storage conditions of the different admixtures is available from Baxter Healthcare upon request.
Ceftriaxone must not be mixed or administered simultaneously with intravenous calcium containing solutions, including Finomel (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Finomel should not be administered simultaneously with blood through the same infusion tubing.
Preparation of the infusion.
Mix the contents of the bag thoroughly and visually inspect the mixture. There should be no signs of emulsion phase separation. The mixture is a milky white homogeneous emulsion.
When making additions, the final osmolarity of the admixture must be assessed, especially for an administration via a peripheral vein.
Remove the protector cap from the infusion port and attach the infusion set. Hang the bag on an infusion stand and carry out infusion using the standard technique.
After opening the bag, content should be used immediately, and should not be stored for a subsequent infusion.
Do not reconnect any partially used bag. Do not connect in series in order to avoid the possibility of air embolism.
Any unused medicinal product or waste material should be disposed of in accordance with local requirements.4.3 Contraindications
The use of Finomel is contraindicated in the following situations:
Hypersensitivity or known allergy to corn or corn products, fish, egg, soya, or peanut proteins or to any of the active substances or excipients listed (see Section 3 Pharmaceutical Form);
Severe hyperlipidaemia;
Severe hepatic impairment;
Severe blood coagulation disorders;
Congenital abnormalities of amino acid metabolism;
Severe renal impairment without access to haemofiltration or dialysis;
Uncontrolled hyperglycaemia;
Pathologically elevated serum levels of any of the included electrolytes;
General contraindications to infusion therapy: acute pulmonary oedema, hyperhydration, and decompensated cardiac insufficiency;
Unstable conditions (e.g. severe post-traumatic conditions, uncompensated diabetes mellitus, acute myocardial infarction, stroke, embolism, metabolic acidosis, severe sepsis, hypotonic dehydration and hyperosmolar coma).
4.4 Special Warnings and Precautions for Use
Administration.
Finomel must only be administered through a central vein.
Hypersensitivity or anaphylactic reaction.
The infusion must be stopped immediately if any signs or symptoms of an allergic reaction (such as fever, shivering, rash or dyspnea) develop.
Finomel contains soya oil, fish oil and egg phospholipids, which may rarely cause allergic reactions. Cross allergic reaction has been observed between soybean and peanut.
Solutions containing dextrose should be used with caution in patients with known allergy to corn or corn products.
Pulmonary vascular precipitates.
Pulmonary vascular precipitates causing pulmonary vascular emboli and pulmonary distress have been reported in patients receiving parenteral nutrition. In some cases, fatal outcomes have occurred. Excessive addition of calcium and phosphate increases the risk of the formation of calcium phosphate precipitates. Precipitates have been reported even in the absence of phosphate salt in the solution. Suspected in vivo precipitate formation has also been reported.
In addition to inspection of the solution, the infusion set and catheter should also periodically be checked for precipitates.
If signs of pulmonary distress occur, the infusion should be stopped and medical evaluation initiated.
Infection and sepsis.
Since an increased risk of infection is associated with the use of any vein, strict aseptic precautions should be taken to avoid any contamination during catheter insertion and manipulation.
Immunosuppression and other factors such as hyperglycaemia, malnutrition and/or their underlying disease state may predispose patients to infectious complications.
Fat overload syndrome.
Fat overload syndrome has been reported with similar products. This may be caused by inappropriate administration (e.g. overdose and/or infusion rate higher than recommended); however, the signs and symptoms of this syndrome may also occur when the product is administered according to instructions. The reduced or limited ability to metabolize the lipids contained in Finomel accompanied by prolonged plasma clearance may result in a fat overload syndrome. This syndrome is associated with a sudden deterioration in the patient's clinical condition and is characterized by findings such as fever, anaemia, leucopoenia, thrombocytopenia, coagulation disorders, hyperlipidaemia, liver fatty infiltration (hepatomegaly), deteriorating liver function, and central nervous system manifestations (e.g. coma). The syndrome is usually reversible when the infusion of the lipid emulsion is stopped.
Use in patients with impaired lipid metabolism.
Monitor the patient's capacity to eliminate lipids by checking the triglyceride levels. The concentration of triglycerides in serum should not exceed 4.6 mmol/L during infusion.
Use with caution in conditions of impaired lipid metabolism, which may occur in patients with renal failure, diabetes mellitus, pancreatitis, impaired liver function, hypothyroidism and sepsis.
See Monitoring section for tests that should be performed.
Refeeding syndrome.
Refeeding severely undernourished patients may result in the refeeding syndrome that is characterized by the shift of potassium, phosphorus, and magnesium intracellularly as the patient becomes anabolic. Thiamine deficiency and fluid retention may also develop. Careful monitoring and slowly increasing nutrient intakes while avoiding overfeeding can prevent these complications. This syndrome has been reported with similar products.
In malnourished patients, initiation of parenteral nutrition can precipitate fluid shifts resulting in pulmonary oedema and congestive heart failure as well as a decrease in the serum concentration of potassium, phosphorus, magnesium and water soluble vitamins. These changes can occur within 24 to 48 hours, therefore careful and slow initiation of parenteral nutrition is recommended in this patient group, together with close monitoring and appropriate adjustments of fluid, electrolytes, minerals and vitamins.
Parenteral nutrition associated liver disease.
Patients on parenteral nutrition may experience hepatic complications (including cholestasis, hepatic steatosis, fibrosis and cirrhosis, possibly leading to hepatic failure, as well as cholecystitis and cholelithiasis), and should be monitored accordingly. The aetiology of these disorders is thought to be multifactorial and may differ between patients. Patients developing abnormal laboratory parameters or other signs of hepatobiliary disorders should be assessed by a clinician knowledgeable in liver diseases in order to identify possible causative and contributory factors, and possible therapeutic and prophylactic interventions.
Use with caution in patients with hepatic impairment, including cholestasis and/or elevated liver enzymes. Liver function parameters should be closely monitored.
Administration of amino acid solutions to a patient with hepatic insufficiency may result in serum amino acid imbalances, hyperammonaemia, stupor and coma. Hyperammonaemia appears to be related to a deficiency of the urea cycle amino acids of genetic or product origin.
The absence of taurine may contribute to the risk of parenteral nutrition associated cholestatic hepatoxicity.
Should symptoms of hyperammonaemia develop, administration should be discontinued and the patient's clinical status re-evaluated.
Hyperglycaemia and hypoglycaemia.
If hyperglycaemia occurs, it should be treated according to the clinical situation either by appropriate insulin administration and/or adjustment of the infusion rate (see Section 4.9 Overdose).
Sudden cessation in administration of a concentrated glucose solution may result in insulin reaction due to continued endogenous insulin production. Parenteral nutrition mixtures should be withdrawn slowly.
Renal impairment.
Use with caution in patients with renal impairment. The phosphate, magnesium, and potassium intake should be carefully controlled to prevent hyperphosphatemia, hypermagnesemia and/or hyperkalaemia.
Disturbances of the electrolyte and fluid balance (e.g. abnormally high or low serum levels of the electrolytes) should be corrected before starting the infusion.
Azotaemia may occur in particular in the presence of renal impairment, and has been reported with parenteral administration of solutions containing amino acids.
Monitoring.
Disturbances of the electrolyte and fluid balance (e.g. abnormally high or low serum levels of the electrolytes) should be corrected before starting the infusion.
Throughout treatment, monitor water and electrolyte balance (including potassium, phosphorus, and magnesium), serum osmolarity, serum triglycerides, acid-base balance, blood glucose, liver (including enzymes) and kidney function (including creatinine, urea), and blood count, including platelets and coagulation parameters.
Especially with long term parenteral nutrition vitamins, zinc and copper intake should be watched.
Lactic acidosis.
Use with caution in patients with lactic acidosis, insufficient cellular oxygen supply and/or increased serum osmolarity.
Thiamine diphosphate, cocarboxylase, is an essential co-enzyme in the carbohydrate metabolism; therefore, patients having thiamine deficiency (e.g. in patients with chronic alcoholism [risk of severe lactic acidosis due to impaired oxidative metabolisation of pyruvate) should be treated cautiously.
Long-term use.
Intravenous infusion of amino acids is accompanied by increased urinary excretion of the trace elements, in particular copper and zinc. This should be considered in the dosing of trace elements, especially during long-term intravenous nutrition. The quantity of zinc administered with Finomel should be taken into account.
Cardiovascular.
Use with caution in patients with pulmonary oedema or heart failure. Fluid status should be closely monitored in all patients receiving parenteral nutrition.
Excess of amino acid infusion.
As with other amino acid solutions, the amino acid content in Finomel may cause undesirable effects when the recommended infusion rate is exceeded. These effects are nausea, vomiting, shivering and sweating. Amino acid infusion may also cause a rise in body temperature. With an impaired renal function, increased levels of nitrogen containing metabolites (e.g. creatinine, urea) may occur.
Electrolyte retention.
Finomel should be given with caution to patients with a tendency towards electrolyte retention e.g. patients receiving corticosteroids or corticotrophin as it may lead to hypernatremia. Special clinical monitoring is required at the beginning of any intravenous infusion. Should any abnormal sign occur, the infusion must be stopped.
Bleeding disorders.
There is a theoretical possibility of the risk of bleeding disorders associated with the use of fish oil.
Use in the elderly.
In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Paediatric use.
There have been no studies performed with Finomel in the paediatric population.
Effects on laboratory tests.
The lipids contained in this emulsion may interfere with the results of certain laboratory tests (for example, bilirubin, lactate dehydrogenase, oxygen saturation, blood haemoglobin) if the blood sample is taken before the lipids are eliminated (these are generally eliminated after a period of 5 to 6 hours without receiving lipids).4.5 Interactions with Other Medicines and Other Forms of Interactions
No interaction studies have been performed with Finomel.
Finomel should not be administered simultaneously with blood through the same infusion tubing due to the risk of pseudoagglutination.
Ceftriaxone must not be administered simultaneously with intravenous calcium-containing solutions, including Finomel, through the same infusion line (e.g. via Y-connector) because of the risk of precipitation of ceftriaxone-calcium salt.
If the same infusion line is used for sequential administration, the line must be thoroughly flushed between infusions with a compatible fluid.
Soya oil has a natural content of vitamin K1. However, the concentration in Finomel is so low that it is not expected to significantly influence the coagulation process in patients treated with coumarin derivatives.
Heparin given in clinical doses causes a transient increase in lipoprotein lipase release into the circulation. This may initially result in increased plasma lipolysis, followed by a transient decrease in triglyceride clearance.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No studies have been conducted to assess the effects of Finomel on fertility.
There are no adequate data from the use of Finomel in pregnant women. Physicians should carefully consider the potential risks and benefits for each specific patient before administering Finomel.
No animal studies have been conducted with the combined lipid components of Finomel to evaluate effects on reproduction. Embryotoxicity and increased incidences of fetal skeletal variations have been observed in rabbits that had received medium chain fatty acid-containing lipids similar to those in Finomel during the period of organogenesis.
Finomel should not be used during pregnancy unless the expected therapeutic benefit clearly outweighs the potential risk to the fetus.
There are no adequate data from the use of Finomel in lactating women. It is not known whether Finomel can enter maternal milk. As zinc is excreted in milk, there is a theoretical risk of zinc-induced copper deficiency in the infant at high doses of Finomel. Physicians should carefully consider the potential risks and benefits for each specific patient before administering Finomel.4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
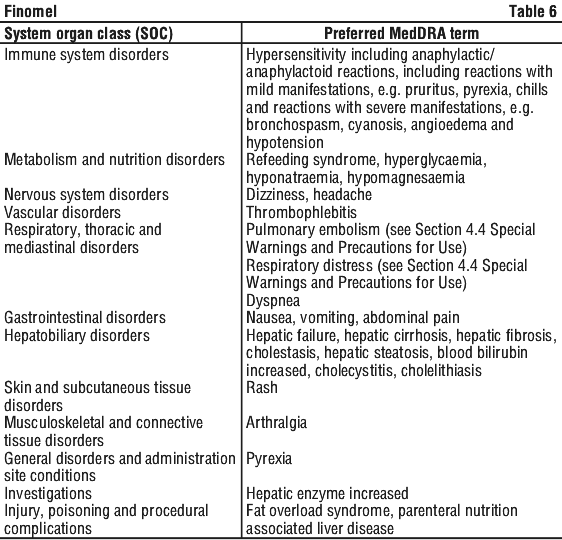
The following adverse reactions have been reported with other similar products and components. The frequency of these events cannot be estimated from available data (see Table 6):
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
In the event of an overdose, nausea, vomiting, chills, hyperglycaemia, and electrolyte disturbances and signs of hypervolaemia or acidosis may occur. In such situations the infusion must be stopped immediately.
If hyperglycaemia occurs, it should be treated according to the clinical situation either by appropriate insulin administration and/or adjustment of the infusion rate. Additionally, overdose might cause fluid overload, electrolyte imbalances and hyperosmolality.
If symptoms persist after discontinuing infusion, haemodialysis, haemofiltration or haemodiafiltration may be considered.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Pharmacotherapeutic group: Solutions for parenteral nutrition/combination, ATC code: B05 BA10.
Lipid emulsion.
The lipid component of Finomel is a lipid mixture composed of a combination of four different oil sources: soya oil (30%), medium chain triglyceride oil (25%), olive oil (25%) and fish oil (20%).
Soya oil has a high content of essential fatty acids. The omega-6 fatty acid linoleic acid is the most abundant (approx. 55-60%). Alpha-linolenic acid, an omega-3 fatty acid, constitutes about 8%.
Medium-chain fatty acids are rapidly oxidized and provide the body with a form of immediately available energy.
Olive oil mainly provides energy in the form of mono-unsaturated fatty acids, which are much less prone to peroxidation than the corresponding amount of polyunsaturated fatty acids.
Fish oil is characterized by a high content of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA). DHA is an important structural component of cell membranes, whereas EPA is a precursor of eicosanoids such as prostaglandins, thromboxanes and leucotrienes.
Amino acids and electrolytes.
The amino acids, constituents of protein in ordinary food, are utilized for tissue protein synthesis and any surplus is channelled to a number of metabolic pathways.
Glucose.
Glucose should serve as a source of energy and contributes to the maintenance of the normal nutritional status.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
Distribution and metabolism.
Amino acids and electrolytes.
The principal pharmacokinetic properties of the infused amino acids and electrolytes are essentially the same as for amino acids and electrolytes supplied by ordinary food. However, the amino acids of dietary protein first enter the portal vein and then the systemic circulation, while intravenously infused amino acids reach the systemic circulation directly.
Lipid emulsion.
The individual triglycerides in combination lipid emulsions have different clearance rate Olive oil has the slowest clearance rate of the components (somewhat slower than LCT) and medium chain triglycerides (MCT) the fastest. Fish oil in a mixture with LCT has the same clearance rate as LCT alone.
5.3 Preclinical Safety Data
Genotoxicity.
No genotoxicity studies have been conducted with Finomel.
Carcinogenicity.
No carcinogenicity studies have been conducted with Finomel.6 Pharmaceutical Particulars
6.1 List of Excipients
The excipients used in Finomel are glycerol, egg lecithin, sodium oleate, dl-alpha-tocopherol, glacial acetic acid, hydrochloric acid (pH adjustment), sodium hydroxide (pH adjustment) and water for injections.
6.2 Incompatibilities
See Section 4.2 Dose and Method of Administration; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Storage condition.
Store below 25°C. Do not freeze. Store in overpouch.
After reconstitution.
It is recommended to use the product immediately after the non-permanent seals between the three chambers have been opened. However stability data of the reconstituted mixtures supports 7 days between 2°C and 8°C followed by 48 hours at 25°C.
After supplementation (electrolytes, trace elements, vitamins; see Section 4.2 Dose and Method of Administration).
For specific admixtures, in-use stability has been demonstrated for 7 days between 2°C and 8°C followed by 48 hours at 25°C.
To reduce microbiological hazard, use as soon as practicable after reconstitution. If storage is necessary, hold at 2°C - 8°C for not more than 24 hours.
6.5 Nature and Contents of Container
Presentation.
The three-chamber bag is a non-PVC multi-layer plastic bag, with 3 port tubes:
One additive site is on the glucose compartment, one infusion site on the amino acid compartment and one port tube on the lipid compartment which is sealed-off to prevent any addition to this chamber.
The product is available in pack of (see Table 7):
 Not all pack sizes may be marketed.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
Any unused product or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
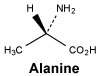
Chemical structure.
Alanine.
White or almost white, crystalline powder or colourless crystals. Freely soluble in water, very slightly soluble in ethanol (96%).
Molecular formula and molecular mass: C3H7NO2; 89.1.
CAS number.
56-41-7.
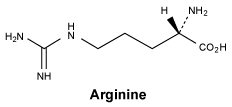
Arginine.
White or almost white, crystalline powder or colourless crystals, hygroscopic. Freely soluble in water, very slightly soluble in ethanol (96%).
Molecular formula and molecular mass: C6H14N4O2; 174.2.
CAS number.
74-79-3.
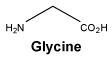
Glycine.
White or almost white, crystalline powder. Freely soluble in water, very slightly soluble in ethanol (96%).
Molecular formula and molecular mass: C2H5NO2; 75.1.
CAS number.
56-40-6.
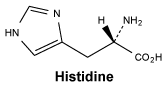
Histidine.
White or almost white, crystalline powder or colourless crystals. Soluble in water, very slightly soluble in ethanol (96 per cent).
Molecular formula and molecular mass: C6H9N3O2; 155.2.
CAS number.
71-00-1.
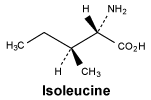
Isoleucine.
White or almost white, crystalline powder or flakes. Sparingly soluble in water, slightly soluble in ethanol (96%). It dissolves in dilute mineral acids and in dilute solutions or alkali hydroxides.
Molecular formula and molecular mass: C6H13NO2; 131.2.
CAS number.
73-32-5.
Leucine.
White or almost white, crystalline powder or shiny flakes. Sparingly soluble in water, practically insoluble in ethanol (96 per cent). It dissolves in dilute mineral acids and in dilute solutions of alkali hydroxides.
Molecular formula and molecular mass: C6H13NO2; 131.2.
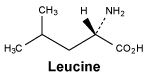
CAS number.
61-90-5.
Lysine hydrochloride.
White or almost white, crystalline powder or colourless crystals. Freely soluble in water, slightly soluble in ethanol (96%).
Molecular formula and molecular mass: C6H15ClN2O2; 182.7.
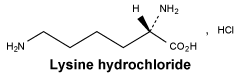
CAS number.
657-27-2.
Methionine.
White or almost white, crystalline powder or colourless crystals. Soluble in water, very slightly soluble in ethanol (96%).
Molecular formula and molecular mass: C5H11NO2S; 149.2.
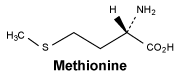
CAS number.
63-68-3.
Phenylalanine.
White or almost white, crystalline powder, or shiny, white flakes. Sparingly soluble in water, very slightly soluble in ethanol (96%). It dissolves in dilute mineral acids and in dilute solutions of alkali hydroxides.
Molecular formula and molecular mass: C9H11NO2; 165.2.
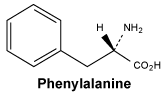
CAS number.
63-91-2.
Proline.
White or almost white, crystalline powder or colourless crystals. Very soluble in water, freely soluble in ethanol (96%).
Molecular formula and molecular mass: C5H9NO2; 115.1.
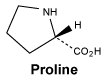
CAS number.
147-85-3.
Serine.
White or almost white, crystalline powder or colourless crystals. Freely soluble in water, practically insoluble in ethanol (96%).
Molecular formula and molecular mass: C3H7NO3; 105.1.
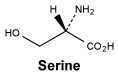
CAS number.
56-45-1.
Threonine.
White or almost white, crystalline powder or colourless crystals. Soluble in water, practically insoluble in ethanol (96%).
Molecular formula and molecular mass: C4H9NO3; 119.1.
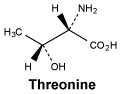
CAS number.
72-19-5.
Tryptophan.
White or almost white, crystalline or amorphous powder. Sparingly soluble in water, slightly soluble in ethanol (96%). It dissolves in dilute solutions of mineral acids and alkali hydroxides.
Molecular formula and molecular mass: C11H12N2O2; 204.2.
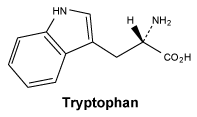
CAS number.
73-22-3.
Tyrosine.
White or almost white, crystalline powder or colourless crystals. Very slightly soluble in water, practically insoluble in ethanol (96%). It dissolves in dilute mineral acids in dilute solutions of alkali hydroxides.
Molecular formula and molecular mass: C9H11NO3; 181.2.
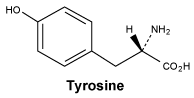
CAS number.
60-18-4.
Valine.
White or almost white, crystalline powder or colourless crystals. Soluble in water, very soluble in ethanol (96%).
Molecular formula and molecular mass: C5H11NO2; 117.1.
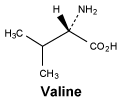
CAS number.
72-18-4.
Soya oil.
Clear, pale yellow liquid. Practically insoluble in ethanol (96%), miscible with light petroleum.
Molecular formula and molecular mass: Triacylglycerol (triglyceride) with fatty acid chains mainly C16:0, C18:0, C18:1, C18:2, C18:3.
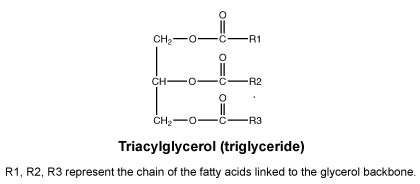
CAS number.
8001-22-7.
Olive oil.
Clear, colourless or greenish-yellow transparent liquid. Practically insoluble in ethanol (96%), miscible with light petroleum.
Molecular formula and molecular mass: Triacylglycerol (triglyceride) with fatty acid chains mainly C16:0, C18:1, C18:2.

CAS number.
8001-25-0.
Medium chain triglycerides.
Colourless or slightly yellowish, oily liquid. Practically insoluble in water, miscible with ethanol (96%), with methylene chloride, with light petroleum and with fatty oils.
Molecular formula and molecular mass: Triacylglycerol (triglyceride) with fatty acid chains mainly C8:0, C10:0.

CAS number.
73398-61-5 and 65381-09-1.
Fish oil rich omega-3 acids.
Pale yellow liquid. Practically insoluble in water, very soluble in acetone and in heptane, slightly soluble in anhydrous ethanol.
Molecular formula and molecular mass: Triacylglycerol (triglyceride) fatty acids mainly C20:5, C22:6.

CAS number.
8016-13-5.
Glucose monohydrate.
White or almost white, crystalline powder. Freely soluble in water, very slightly soluble in ethanol (96%).
Molecular formula and molecular mass: C6H12O6, H2O; 198.2.

CAS number.
77938-63-7.
Sodium acetate trihydrate.
White or almost white, crystalline powder or colourless crystals. Very soluble in water, soluble in ethanol (96%).
Molecular formula and molecular mass: C2H3NaO2, 3H2O; 136.1.

CAS number.
6131-90-4.
Sodium glycerophosphate hydrate.
White or almost white, crystalline powder or crystals. Freely soluble in water, practically insoluble in acetone and in ethanol (96%).
Molecular formula and molecular mass: C3H7Na2O6P, H2O; 238.0.

CAS number.
1334-74-3.
Potassium chloride.
White or almost white crystalline powder or colourless crystals. Freely soluble in water, practically insoluble in anhydrous ethanol.
Molecular formula and molecular mass: KCl; 74.6.
CAS number.
7447-40-7.
Calcium chloride dihydrate.
White or almost white, crystalline powder, hygroscopic. Freely soluble in water, soluble in ethanol (96%).
Molecular formula and molecular mass: CaCl2, 2H2O; 147.0.
CAS number.
10035-04-8.
Magnesium sulfate heptahydrate.
White or almost white, crystalline powder or brilliant, colourless crystals. Freely soluble in water, very soluble in boiling water, practically insoluble in ethanol (96%).
Molecular formula and molecular mass: MgSO4, 7H2O; 246.5.
CAS number.
10034-99-8.
Zinc sulfate heptahydrate.
White or almost white, crystalline powder or colourless, transparent crystals, efflorescent. Very soluble in water, practically insoluble in ethanol (96%).
Molecular formula and molecular mass: ZnSO4,7H2O; 287.5.
CAS number.
7446-20-0.7 Medicine Schedule (Poisons Standard)
Not scheduled.
Summary Table of Changes


 The chamber volume for each bag sizes are detailed in Table 2:
The chamber volume for each bag sizes are detailed in Table 2: For the detailed formulations, see Table 4 and Table 5.
For the detailed formulations, see Table 4 and Table 5. The composition of the 3 in 1 admixture for each of the bag presentations are provided in Table 4 and Table 5.
The composition of the 3 in 1 admixture for each of the bag presentations are provided in Table 4 and Table 5.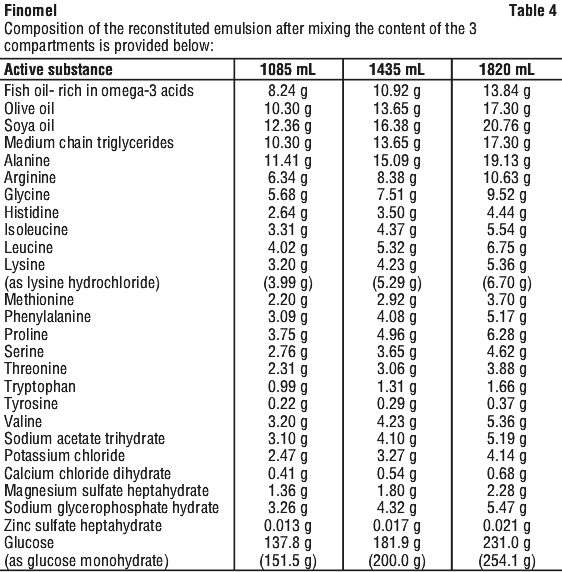

 Not all pack sizes may be marketed.
Not all pack sizes may be marketed.


















