What is in this leaflet
This leaflet answers some common questions about Spiriva capsules.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Spiriva against the expected benefits.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
This leaflet was last updated on the date at the end of this leaflet. More recent information may be available. The latest Consumer Medicine Information is available from your pharmacist, doctor or from www.medicines.org.au and may contain important information about the medicine and its use of which you should be aware.
Keep this leaflet with the medicine. You may need to read it again.
What Spiriva is used for
Spiriva is used to make breathing easier for people with chronic obstructive pulmonary disease (COPD).
This helps to improve your condition and to prevent exacerbations (periodic worsening of symptoms) from occurring.
Spiriva improves breathing by relaxing the air passages that carry air to and from the lungs. It begins to act within 30 minutes after use and the effect should last a full day.
Spiriva contains the active ingredient tiotropium. It belongs to a group of medicines called anticholinergics.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
Before you take Spiriva
When you must not take it
Do not take Spiriva if you have an allergy to:
- any medicine containing tiotropium
- any of the ingredients listed at the end of this leaflet
- any medicine containing atropine or its derivatives, e.g. ipratropium or oxitropium
- any other anticholinergic medicines.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not take this medicine to treat a sudden attack of breathlessness, wheezing or coughing. You will need a different type of medicine.
Do not give this medicine to children or adolescents (below the age of 18 years).
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have, or have had, any of the following medical conditions:
- high pressure in the eye (glaucoma)
- kidney or liver problems
- problems with your prostate gland
- problems with passing urine
- you have suffered from a heart attack during the last 6 months or from any unstable or life threatening irregular heart beat or severe heart failure within the past year.
Tell your doctor if you are pregnant or plan to become pregnant or are breast-feeding. Your doctor can discuss with you the risks and benefits involved. Spiriva is not generally recommended for use in pregnant women.
If you have not told your doctor about any of the above, tell him/her before you start taking Spiriva.
Do not allow the powder to enter into the eyes. Should this occur, immediately flush your eyes with cold tap water for several minutes and immediately consult your doctor for further advice. If the powder enters the eye, it may result in eye pain or discomfort, blurred vision, seeing halos around lights or coloured images in association with red eyes (i.e. narrow angle glaucoma).
Take this medicine only with the HandiHaler® device.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Spiriva may interfere with each other. These include other anticholinergic medicines used to treat COPD such as glycopyrronium, aclidiniuim, umeclidinium or ipratropium.
These medicines may be affected by Spiriva or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How to take Spiriva
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
The recommended dose for adults is 1 capsule to be inhaled, once a day.
How to take it
Inhale the powder in the capsules only using the HandiHaler device.
Do not swallow the capsules.
Do not open the capsules. If you open the capsules, the small amount of powder inside may be lost, or you may accidentally get the powder in your eyes.
Use each strip of 5 capsules within 5 days of first opening the strip.
Refer to the HandiHaler Instructions for Use.
Carefully read the Instructions for Use and follow the instructions on how to use Spiriva capsules with the HandiHaler device.
Pierce the capsule only once using the HandiHaler device. If you pierce it more than once, the capsule may break and pieces of gelatin from the capsule shell, small enough to pass the sieve, may be inhaled into your mouth or throat. Gelatin is not harmful and small particles swallowed or inhaled will be digested or absorbed and are not expected to cause undesirable effects.
Do not allow the powder to enter into the eyes. Should this occur, immediately flush your eyes with cold tap water for several minutes.
If you have any problems inhaling Spiriva using the HandiHaler device ask your doctor or pharmacist for advice.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
How long to take it
Continue taking your medicine for as long as your doctor tells you.
This medicine helps to control your condition, but does not cure it. It is important to keep taking your medicine even if you feel well.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (Australia 13 11 26) for advice, or go to Emergency at the nearest hospital, if you think that you or anyone else may have taken too much Spiriva. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Symptoms of an overdose may include fast or irregular heart beat, blurred vision, nausea, stomach pain, dry mouth, constipation and difficulty passing urine.
While you are taking Spiriva
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking Spiriva.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you become pregnant while taking this medicine, tell your doctor immediately.
Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
It is very important to follow the HandiHaler Instructions for Use carefully.
If your breathing becomes more difficult while you are taking this medicine, tell your doctor immediately.
If you are a smoker, your doctor or pharmacist can advise you on the steps to take to quit smoking.
Things you must not do
Do not take Spiriva to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not take Spiriva more frequently than once daily.
Things to be careful of
Be careful driving or operating machinery until you know how Spiriva affects you. This medicine may cause dizziness or blurred vision in some people. If you have any of these symptoms, do not drive, operate machinery, or do anything else that could be dangerous.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Spiriva.
This medicine helps most people with COPD, but it can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following list of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- dry mouth: this is usually mild
- sore mouth, gums, or throat; swollen, red, sore tongue
- oral thrush
- hoarse voice
- nose bleeds
- cough
- dizziness
- constipation
- trouble sleeping
- sinusitis, a feeling of tension or fullness in the nose, cheeks and behind your eyes, sometimes with a throbbing ache, fever, stuffy nose and loss of the sense of smell.
These are mild side effects of the medicine, and usually short-lived.
Tell your doctor as soon as possible if you notice any of the following:
- blurred vision
- seeing halos around lights or coloured images in association with red eyes; high pressure in the eye (glaucoma)
- difficulty in swallowing
- heartburn
- difficulty in passing urine
- pain while passing urine, urinary tract infection, increased need and frequency in passing urine
- a worsening of breathing problems (induced by the inhalation process). Inhaled medicines such as Spiriva may cause tightness of the chest, coughing, wheezing or breathlessness immediately after inhalation.
The above list includes serious side effects that may require medical attention.
If any of the following happen, tell your doctor immediately or go to Emergency at your nearest hospital:
- severe pain in the stomach with bloating, gut cramps and vomiting
- changes in heart rate (fast, slow or irregular), palpitations
- allergic reaction (shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body, rash, itching or hives on the skin).
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may occur in some people.
After taking Spiriva
Care and cleaning
Refer to the HandiHaler Instructions for Use on how to clean and take care of your Spiriva HandiHaler.
Storage
Keep your capsules in the pack until it is time to take them. If you take the capsules out of the pack they may not keep well.
Keep your capsules in a cool, dry place where the temperature stays below 25°C. Do not store your capsules in the freezer.
Do not store Spiriva or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product Description
What it looks like
Spiriva capsules are light green with the product code "TI 01" and the Boehringer Ingelheim logo printed in black ink on the capsules. Spiriva capsules contain a white powder.
Spiriva capsules contain only a small amount of powder, which means that the capsule is only partially filled. The amount of powder in each capsule is equivalent in size to the tip of a matchstick.
Spiriva is available in cartons containing 10*, 30 or 60* capsules and hospital packs containing 150* or 300* capsules. Combination packs of 10, 30* and 150* capsules with the HandiHaler device are also available.
*Not currently distributed in Australia.
The HandiHaler device is available separately from your pharmacist and should be replaced every twelve months.
Ingredients
Spiriva contains 18 micrograms of tiotropium (equivalent to tiotropium bromide monohydrate 22.5 micrograms) as the active ingredient.
It also contains 5.5 milligrams of lactose monohydrate (which contains milk protein).
Supplier
Spiriva is supplied in Australia by:
Boehringer Ingelheim Pty Limited
ABN 52 000 452 308
Sydney, Australia
www.boehringer-ingelheim.com.au
This Consumer Medicine Information was updated in May 2020.
Spiriva® is a registered trademark of Boehringer Ingelheim
© Boehringer Ingelheim Pty Limited 2020
Australian Registration Number:
AUST R 81525
HandiHaler®
How do you take your dose of Spiriva® using the HandiHaler®?
Taking your dose of Spiriva requires four main steps: Open the blister and the HandiHaler device, insert the Spiriva capsule, press the HandiHaler button, and inhale your medication.
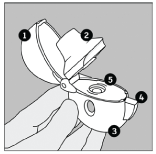
Become familiar with the components of the HandiHaler inhalation device:
- dust cap
- mouthpiece
- base
- piercing button
- centre chamber
Removing the Spiriva capsule from the blister
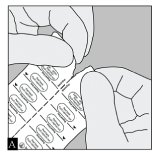
Spiriva capsules are packaged in a blister card. Each blister card consists of two blister strips, each containing 5 capsules and joined along a perforated line. Prior to removing the first capsule from the blister card, separate the blister strips by tearing along the perforation. (Figure A)
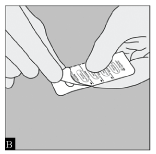
The blister should be carefully opened to expose only one capsule at a time. Immediately before you are ready to take your dose of Spiriva, peel back the aluminium foil using the tab at the rounded edge until one capsule is fully visible. The foil lidding should only be peeled back as far as the STOP line printed on the blister foil to prevent exposure of more than one capsule. (Figure B)
Turn the blister strip upside down and tip the capsule out, tapping the back of the blister pack, if necessary. DO NOT CUT THE FOIL OR USE SHARP INSTRUMENTS TO REMOVE THE CAPSULE FROM THE BLISTER.
Capsules should always be stored in the sealed blisters and only removed immediately before use. The medicine should be used immediately after the packaging over an individual capsule is opened, or else its effectiveness may be reduced.
If additional capsules are inadvertently exposed to air, they should not be used and should be discarded.
Do not store capsules in the HandiHaler.
Instructions for Use
The HandiHaler enables you to inhale the medicine contained in the Spiriva capsule that your doctor has prescribed for your breathing problems.
Remember to carefully follow your doctor's instructions for using Spiriva capsules.
The HandiHaler is especially designed for Spiriva capsules. It must not be used to take any other medication. Your HandiHaler can be used for up to twelve months to take your medication. You will need to replace your HandiHaler every twelve months.
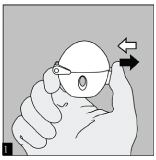
To release the dust cap press the piercing button completely in and let go.
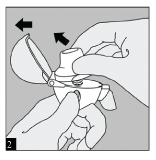
OPEN: Open the dust cap by pulling it upwards. Then open the mouthpiece by pulling it upwards.
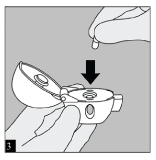
INSERT: Place the capsule in the centre chamber. It does not matter which end of the capsule is placed in the chamber.
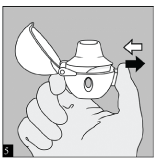
Close the mouthpiece firmly until you hear a click, leaving the dust cap open. Check to see that the mouthpiece is completely closed.
Be sure that the mouthpiece sits firmly against the base so that there is no gap between the mouthpiece and the base.
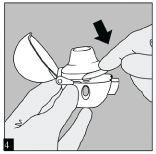
PRESS: Hold the HandiHaler with the mouthpiece upwards and press the piercing button completely in only once until it is flush against the base, and release.
This makes holes in the capsule and allows the medication to be released when you breathe in.
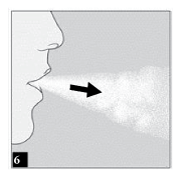
Breathe out completely.
Important:
Do not breathe (exhale) into the HandiHaler mouthpiece at any time.
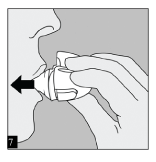
INHALE: Raise the HandiHaler to your mouth and close your lips tightly around the mouthpiece. Keep your head in an upright position and breathe in slowly and deeply but at a rate sufficient to hear or feel the capsule vibrate. Breathe in until your lungs are full; then hold your breath as long as is comfortable and at the same time take the HandiHaler out of your mouth. Resume normal breathing.
To ensure you get the full dose of Spiriva, you must repeat steps 6 and 7 once again.
If you do not hear or feel the capsule vibrate, tap the HandiHaler gently on a table, holding it in an upright position. Check to see that the mouthpiece is completely closed. Then breathe in again - slowly and deeply. If you still do not hear or feel the capsule vibrate after repeating the above steps please consult your doctor or pharmacist to ensure that you are using the HandiHaler correctly.
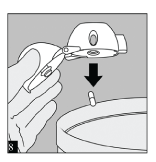
After you have finished taking your daily dose of Spiriva, open the mouthpiece again. Tip out the used capsule and discard.
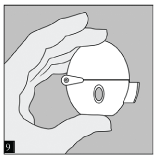
Close the mouthpiece and dust cap for storage of your HandiHaler device.
Cleaning your HandiHaler
Clean the HandiHaler once a month.
Open the dust cap and mouthpiece.
Then open the base by lifting the piercing button. Rinse the complete inhaler with warm water to remove any powder. Dry the HandiHaler thoroughly by tipping excess water out on a paper towel and air-dry afterwards, leaving the dust cap, mouthpiece and base open.
It takes 24 hours to air dry, so clean it right after you use it and it will be ready for your next dose.
If needed, the outside of the mouthpiece may be cleaned with a moist, but not wet, tissue.
Supplier
HandiHaler is supplied in Australia by:
Boehringer Ingelheim Pty Limited
78 Waterloo Road
North Ryde NSW 2113
Australia
Tel. 1800 226 315
HandiHaler is supplied in New Zealand by:
Boehringer Ingelheim (NZ) Limited
Auckland, New Zealand
Tel. 0800 802 461
Manufacturer
HandiHaler is manufactured by:
Boehringer Ingelheim Pharma GmbH & Co. KG
D-55216 Ingelheim
Germany
Published by MIMS August 2020




 A one year randomised, double blind, double dummy, parallel group trial compared the effect of treatment with 18 microgram of Spiriva once daily with that of 50 microgram of salmeterol HFA pMDI twice daily with the primary endpoint time to first moderate or severe exacerbation in 7,376 patients with COPD and a history of exacerbations in the preceding year (74.6% of treated patients were men, 99.6% white, and 48.1% current smokers; the mean age was 62.9 years and the mean FEV1 was 49.3% predicted). The treatment groups were balanced with respect to demographics, COPD characteristics, pulmonary medication use at baseline, and concomitant diagnoses. Patients were allowed to continue their usual medications for COPD, except for anticholinergic drugs and long acting beta2-agonists, during the double blind treatment phase. Short acting beta2-agonists were also permitted, as necessary, as rescue medications for acute relief of COPD symptoms. See Figures 2 and 3, and Table 3.
A one year randomised, double blind, double dummy, parallel group trial compared the effect of treatment with 18 microgram of Spiriva once daily with that of 50 microgram of salmeterol HFA pMDI twice daily with the primary endpoint time to first moderate or severe exacerbation in 7,376 patients with COPD and a history of exacerbations in the preceding year (74.6% of treated patients were men, 99.6% white, and 48.1% current smokers; the mean age was 62.9 years and the mean FEV1 was 49.3% predicted). The treatment groups were balanced with respect to demographics, COPD characteristics, pulmonary medication use at baseline, and concomitant diagnoses. Patients were allowed to continue their usual medications for COPD, except for anticholinergic drugs and long acting beta2-agonists, during the double blind treatment phase. Short acting beta2-agonists were also permitted, as necessary, as rescue medications for acute relief of COPD symptoms. See Figures 2 and 3, and Table 3.

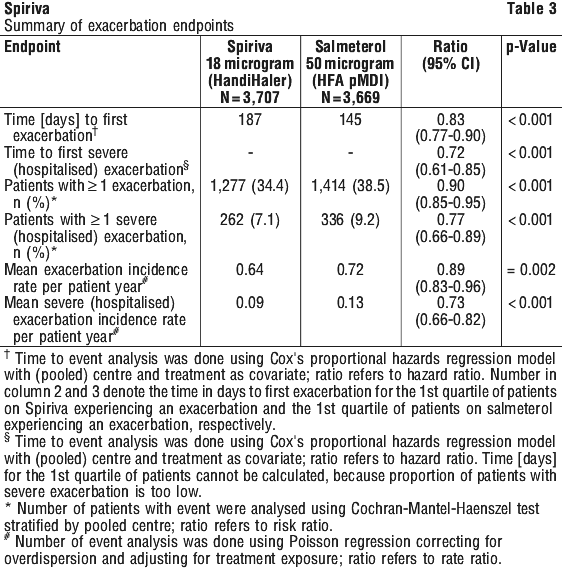 Compared with salmeterol, Spiriva increased the time to the first exacerbation (187 days vs. 145 days), with a 17% reduction in risk (hazard ratio, 0.83; 95% CI, 0.77 to 0.90; P < 0.001). Spiriva also increased the time to the first severe (hospitalised) exacerbation (hazard ratio, 0.72; 95% CI, 0.61 to 0.85; P < 0.001), reduced the annual number of moderate or severe (hospitalised) exacerbations (0.64 vs. 0.72; rate ratio, 0.89; 95% CI, 0.83 to 0.96; P = 0.002), and reduced the annual number of severe (hospitalised) exacerbations (0.09 vs. 0.13; rate ratio, 0.73; 95% CI, 0.66 to 0.82; P < 0.001).
Compared with salmeterol, Spiriva increased the time to the first exacerbation (187 days vs. 145 days), with a 17% reduction in risk (hazard ratio, 0.83; 95% CI, 0.77 to 0.90; P < 0.001). Spiriva also increased the time to the first severe (hospitalised) exacerbation (hazard ratio, 0.72; 95% CI, 0.61 to 0.85; P < 0.001), reduced the annual number of moderate or severe (hospitalised) exacerbations (0.64 vs. 0.72; rate ratio, 0.89; 95% CI, 0.83 to 0.96; P = 0.002), and reduced the annual number of severe (hospitalised) exacerbations (0.09 vs. 0.13; rate ratio, 0.73; 95% CI, 0.66 to 0.82; P < 0.001). A significantly higher proportion of patients in the tiotropium group than in the placebo group had an improvement of ≥ 4 units in the secondary endpoint of SGRQ total scores (i.e. exceeded the minimal clinically important difference) from baseline at 1 year (49% vs. 41%), 2 years (48% vs. 39%), 3 years (46% vs. 37%), and 4 years (45% vs. 36%) (p < 0.001 for all comparisons).
A significantly higher proportion of patients in the tiotropium group than in the placebo group had an improvement of ≥ 4 units in the secondary endpoint of SGRQ total scores (i.e. exceeded the minimal clinically important difference) from baseline at 1 year (49% vs. 41%), 2 years (48% vs. 39%), 3 years (46% vs. 37%), and 4 years (45% vs. 36%) (p < 0.001 for all comparisons).
